Clear Sky Science · en
Impact of neoadjuvant chemotherapy on the functionality of adipose-derived mesenchymal stromal cells and their modulatory effects on fibroblasts in oncology patients
Why this matters for cancer survivors
Many people who undergo cancer treatment later struggle with slow-healing wounds and complications after surgery. At the same time, doctors are increasingly exploring ways to use a patient’s own fat-derived repair cells to help tissues recover. This study asks a practical and pressing question: after chemotherapy, are those repair cells still good enough to use, and if not, which cells are actually most affected?
The body’s hidden repair helpers
Our fat tissue is more than stored energy. It houses a rich population of versatile repair cells called adipose-derived mesenchymal stromal cells, or AD-MSCs. These cells don’t just sit quietly; they release a cocktail of signaling molecules that can calm inflammation, stimulate new blood vessels, and encourage nearby cells to grow and rebuild damaged tissue. Just above the fat, in the skin, live fibroblasts—workhorse cells that migrate into wounds and lay down collagen, the protein scaffold that gives new tissue strength. Together, AD-MSCs and fibroblasts help determine how well the skin bounces back after injury or surgery.
Testing fat repair cells before and after treatment
The researchers collected small samples of skin and fat from 66 patients undergoing surgery, some of whom had received chemotherapy beforehand and some who had not. From the fat, they isolated AD-MSCs and confirmed that these cells still looked and behaved like genuine repair cells: they carried the expected surface markers and could mature into bone-, cartilage-, and fat-like cells in the lab. From the skin, they isolated fibroblasts. The team then compared how AD-MSCs and fibroblasts from chemotherapy-exposed and non-exposed patients grew, cycled through cell division, released signaling molecules, and expressed genes linked to wound healing and regeneration.
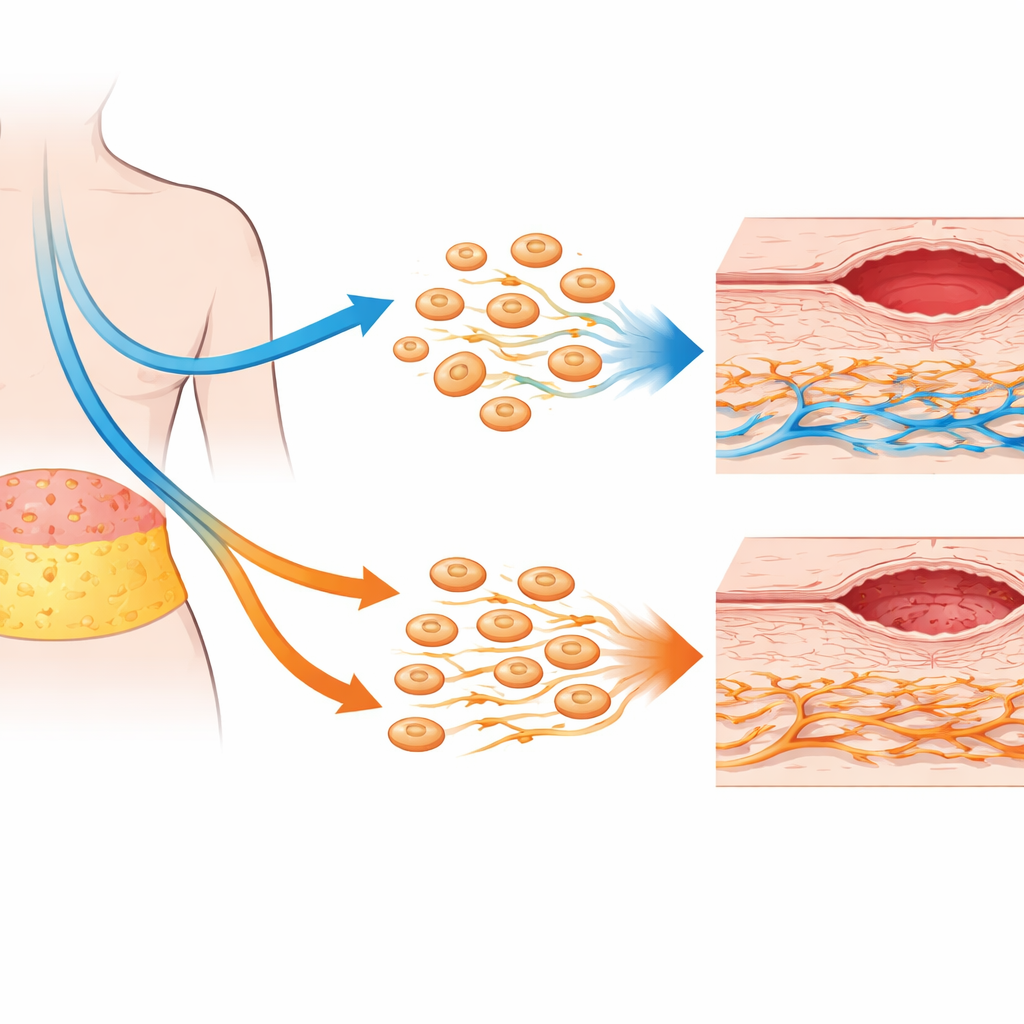
Fat repair cells stay surprisingly strong
Contrary to concerns that chemotherapy might blunt the power of AD-MSCs, the study found that these fat-derived repair cells were remarkably resilient. Their basic identity markers, growth pattern, and pattern of secreted signaling proteins were largely unchanged by prior chemotherapy. Gene activity linked to growth, self-renewal, and wound repair showed only small, non–meaningful shifts. In fact, AD-MSCs from chemotherapy-treated patients displayed slightly higher mitochondrial activity, hinting at more robust metabolism rather than damage. In co-culture experiments—where AD-MSCs and fibroblasts shared the same medium but not direct contact—AD-MSCs still boosted certain fibroblast behaviors, such as directed movement toward repair signals, especially in samples from patients who had not received chemotherapy.
Skin builders bear the brunt of chemotherapy
The picture was very different for fibroblasts. Cells taken from the skin of chemotherapy-treated patients showed clear signs of reduced biological vigor. Their ability to migrate into a scratch-like “wound” on a culture plate was significantly impaired, and their production of collagen tended to be lower. The pattern of signaling molecules they released shifted toward a more inflammatory, less regenerative profile, and many genes associated with growth signals, their receptors, and regenerative potential were dialed down. Even when these fibroblasts were co-cultured with their own AD-MSCs, most of their functions—movement, collagen production, and helpful gene activity—remained blunted compared to fibroblasts from patients who had not undergone chemotherapy.
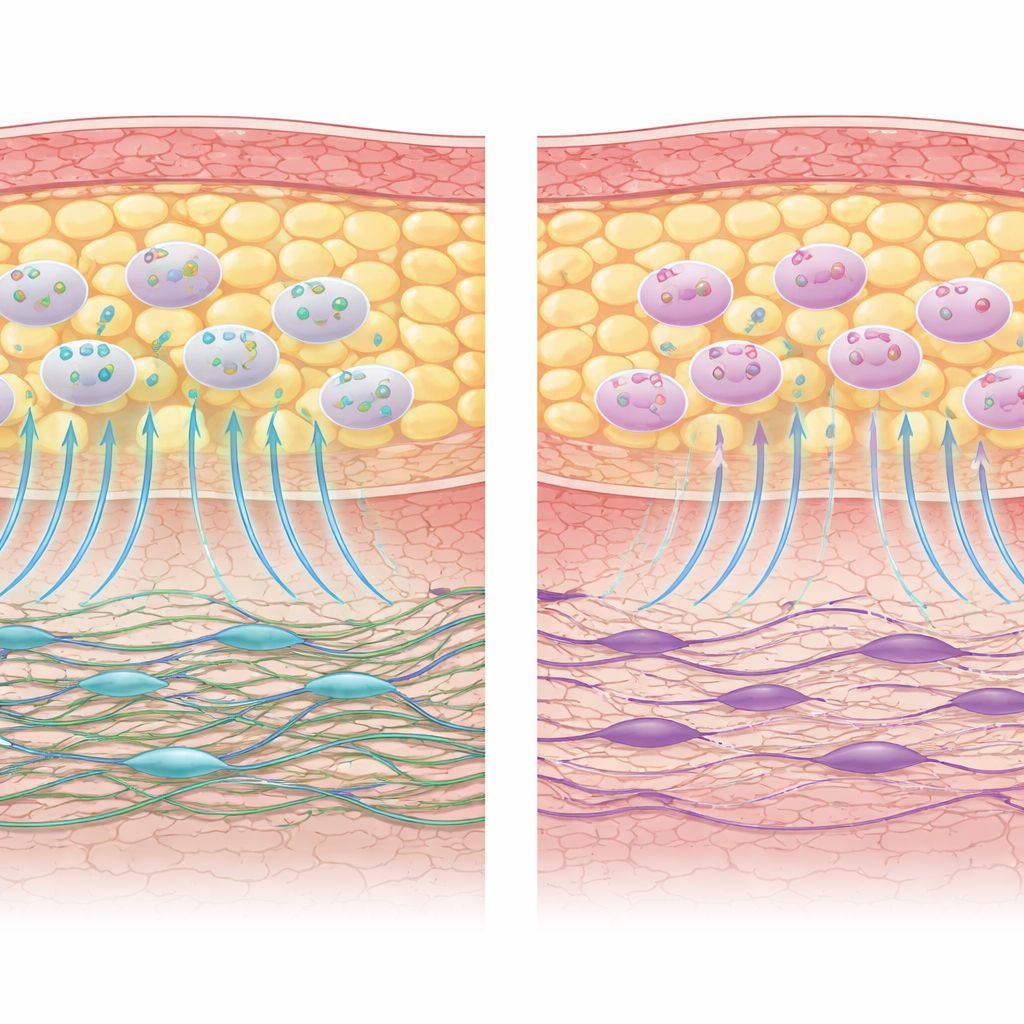
What this means for future healing therapies
To a layperson, the message of this work is that chemotherapy does not seem to ruin the body’s fat-based repair cell bank, but it does leave the skin’s frontline builders—fibroblasts—less able to rush into wounds and rebuild strong tissue. Because AD-MSCs stay functionally intact, they remain promising candidates for use in reconstructive procedures, wound care, and fat grafting after cancer treatment. However, the damaged state of fibroblasts may limit how much benefit these stem-like cells can deliver on their own. The authors suggest that future therapies may need to support both sides of the repair partnership: preserving or restoring fibroblast health while harnessing robust AD-MSCs, ideally in more complex, body-like models that better capture the real healing environment in cancer survivors.
Citation: Skoniecka, A., Słonimska, P., Tymińska, A. et al. Impact of neoadjuvant chemotherapy on the functionality of adipose-derived mesenchymal stromal cells and their modulatory effects on fibroblasts in oncology patients. Sci Rep 16, 8614 (2026). https://doi.org/10.1038/s41598-026-39457-9
Keywords: chemotherapy, wound healing, fat-derived stem cells, fibroblasts, regenerative medicine