Clear Sky Science · en
Role of atomic force microscopy in characterization of heterotypic cancer spheroids and their interaction with microplastic particles
Why the feel of cancer tissue matters
Cancer is usually described in terms of genes and rogue cells, but its physical feel—how soft or stiff the tissue is—also shapes how a tumor grows and responds to its surroundings. This study looks at tiny three‑dimensional clusters of lung cancer cells, called spheroids, and asks two practical questions: can we use a high‑resolution touch‑based microscope to reliably measure how stiff these mini‑tumors are, and does that stiffness influence how they interact with airborne microplastic particles that may reach the lungs?
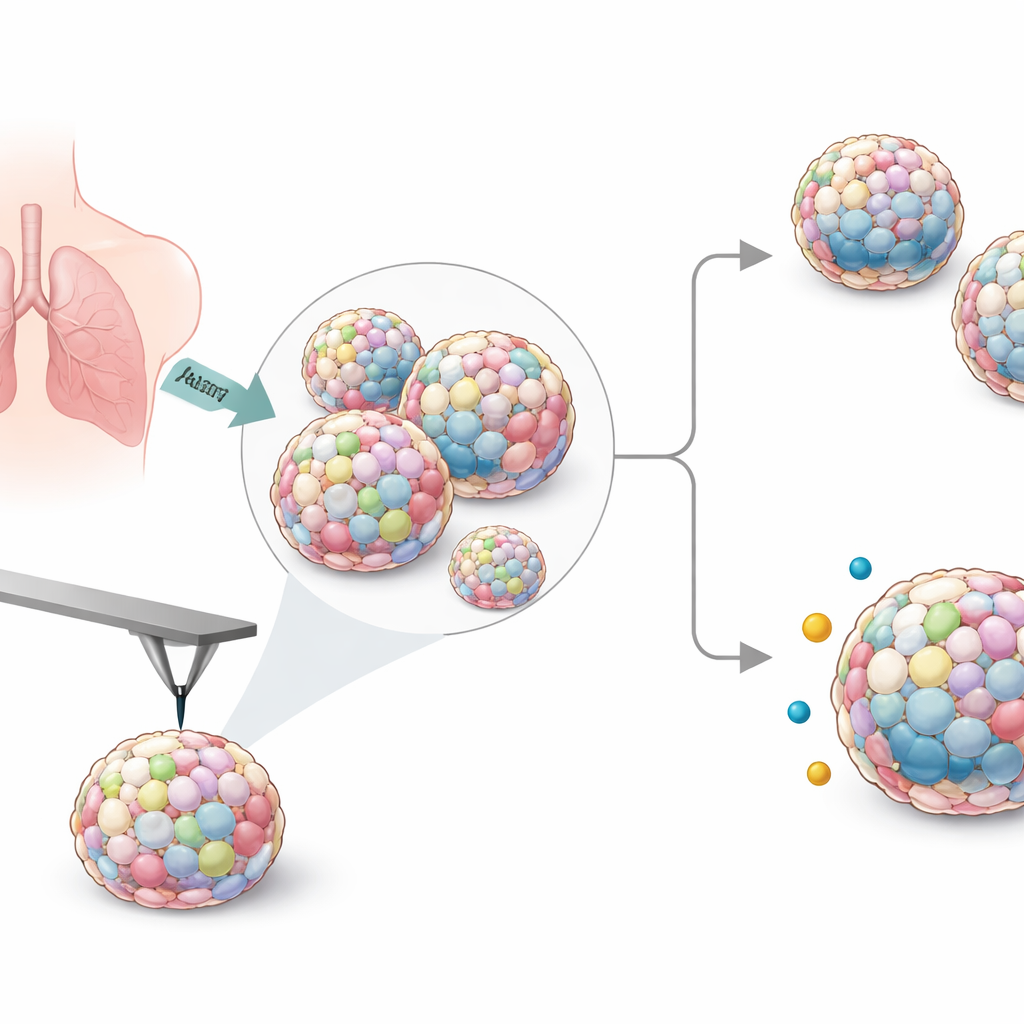
Building mini lung tumors in the lab
To mimic the complex makeup of real tumors, the researchers grew mixed spheroids from six human lung cancer cell lines together with supporting cells called fibroblasts, and sometimes immune‑like macrophages. They tested several ways of forming these clusters and settled on special low‑adhesion plates that reliably produced round, stable spheroids large enough to handle. By adjusting cell numbers and ratios, they created both small and larger spheroids, then chose a standard recipe that worked consistently across all cancer cell types.
Peeking inside and checking cell health
Once the spheroids were formed, the team examined their inner structure and cell behavior using various stains and sectioning methods. Thin slices showed that some cancer cell combinations formed very compact balls, while others were looser with more open spaces. Fibroblasts tended to settle toward the center, especially in most cell line combinations, whereas the cancer cells concentrated near the outside. Live‑dead staining revealed a familiar pattern seen in real tumors: well‑nourished living cells at the rim, and more damaged or dying cells in the low‑oxygen core. A DNA‑labeling test for cell division showed that, for most spheroids, cells across the whole structure were still able to proliferate, though one cancer cell line (Calu‑3) mainly divided at the outer edge.
Measuring stiffness with a nanoscale probe
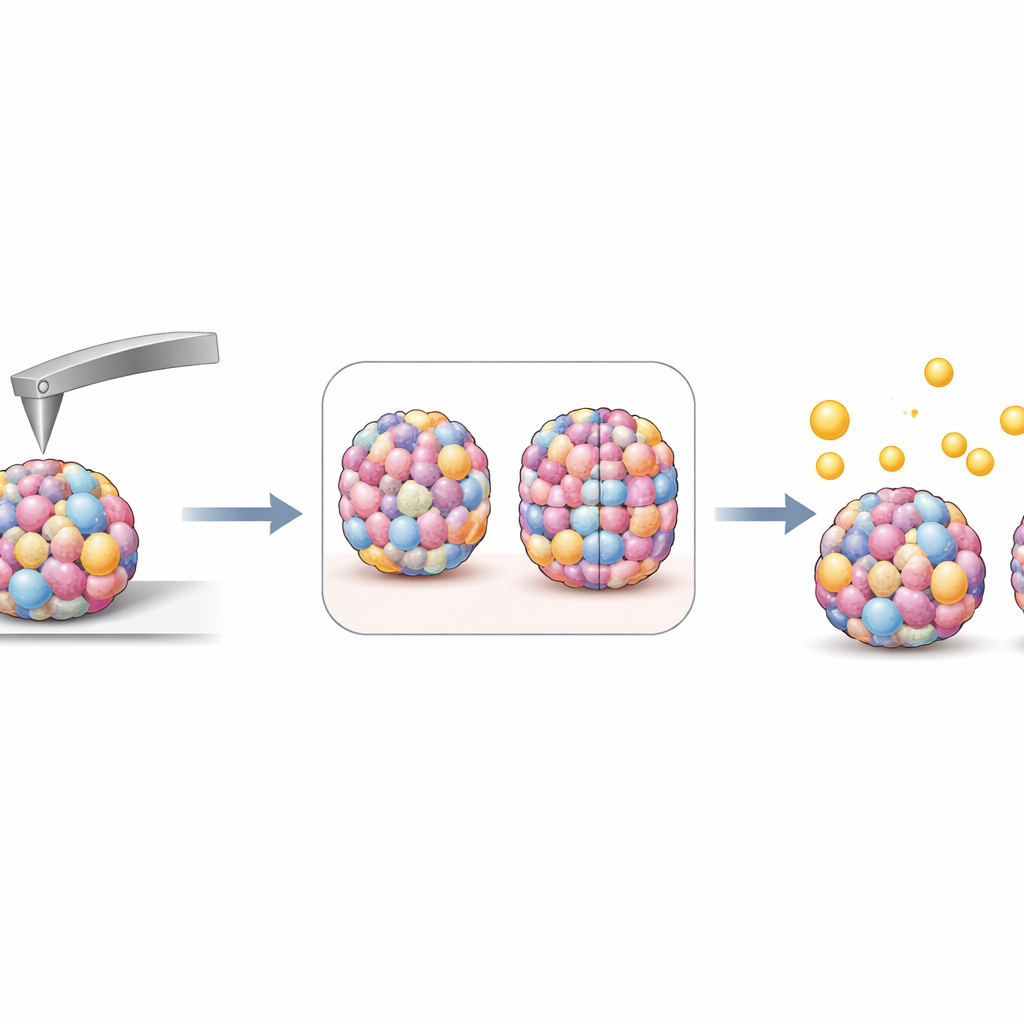
To turn texture into numbers, the scientists used atomic force microscopy, in which a tiny, sharp probe gently presses on the spheroid surface and records how much it indents. From this they calculated Young’s modulus, a standard measure of stiffness. Even though the spheroids were similar in size, their stiffness varied substantially depending on which lung cancer line they contained. Spheroids with A549 cells were among the softest, while those containing H23 or HCC827 cells were noticeably stiffer. Adding macrophages generally increased stiffness for several cancer types. When the team compared these values with how fast the original cancer cells grew in flat culture, they found that slower‑dividing cells tended to form stiffer spheroids, linking growth behavior to mechanical properties.
Testing contact with microplastics
Because microscopic plastic fragments have been detected in lung tumors, the researchers next exposed their spheroids to fluorescent polystyrene particles about one micrometer across, at concentrations similar to those measured in human blood. Under still conditions, the particles clumped and stuck unevenly to one side of the spheroids, so the group switched to gently rocking the samples to better mimic moving body fluids. Under these dynamic conditions, only a few individual particles attached to or entered the outer cell layers, too few for precise counting. Instead, the team again turned to stiffness measurements. After exposure, many spheroid types became slightly stiffer, consistent with rigid plastic beads attaching to or lodging in the surface, but this change did not show a clear, simple dependence on how stiff the spheroid had been to begin with.
What this means for future cancer and pollution studies
The work shows that stiffness is an informative extra parameter for describing three‑dimensional cancer models that otherwise look similar in size and shape. The way cells arrange themselves inside the spheroid and how quickly they divide only partly explains these mechanical differences, suggesting that stiffness captures hidden aspects of tumor biology. Although this study did not find a straightforward rule linking spheroid stiffness to microplastic uptake at realistic particle levels, it highlights that under gentle, moving conditions only a small number of such particles are likely to adhere to tumor‑like tissues. In the long term, combining detailed stiffness measurements with more sensitive particle‑tracking methods could help researchers better predict how tumors respond both to anticancer drugs and to environmental contaminants that reach the lungs.
Citation: Kolesnik, T., Öhlinger, K., Absenger-Novak, M. et al. Role of atomic force microscopy in characterization of heterotypic cancer spheroids and their interaction with microplastic particles. Sci Rep 16, 8303 (2026). https://doi.org/10.1038/s41598-026-39445-z
Keywords: lung cancer spheroids, atomic force microscopy, cell stiffness, microplastics, tumor microenvironment