Clear Sky Science · en
Discovery of potential antiviral compounds and accelerating the therapeutic discovery against monkeypox virus
Why this research matters now
Monkeypox, now often called mpox, has jumped from being a rare tropical infection to a global concern, with outbreaks reported on several continents in recent years. Unlike COVID-19, there is still no drug made specifically to stop this virus. This study uses advanced computer tools to search for new small molecules that might disarm the virus where it hurts most: its ability to hide from our immune system. If confirmed in the lab, these candidate compounds could become the starting point for a new class of antiviral medicines.
How the virus hides from our defenses
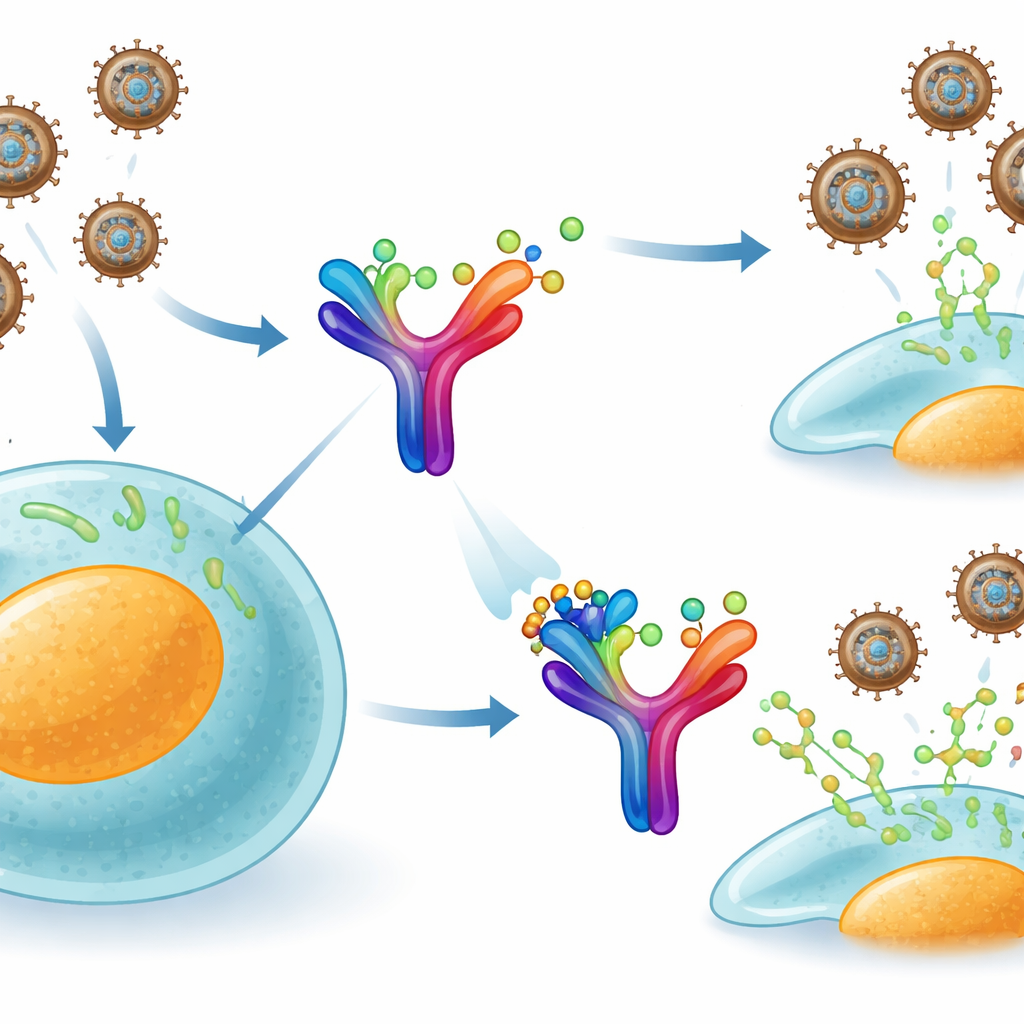
Monkeypox belongs to the same broader family as smallpox, and it uses a clever trick to dodge the immune system. Our cells release a signaling protein called interferon-gamma, which normally warns neighboring cells and switches on antiviral defenses. The virus, however, makes its own secreted protein, called B8R, that floats outside infected cells. B8R closely mimics our own interferon-gamma receptor and grabs these immune signals before they can reach their true target. In effect, it acts like a sponge, soaking up the body’s alarm messages and preventing a full antiviral response.
Using computers to design smarter treatments
Traditional drug discovery can take many years and enormous resources. Here, the researchers turn to computer-aided drug design to speed things up. Because no experimental 3D structure of the B8R protein exists, they first used AlphaFold-based tools to predict its atomic shape and carefully checked the quality of that model. With a reliable 3D structure in hand, they virtually screened a chemical library of 5000 small molecules. Rules that describe what usually makes a pill-like drug (such as size, shape, and solubility) were used to filter out poor candidates, leaving 2890 promising compounds for detailed testing against the B8R protein model.
Finding a promising chemical “key”
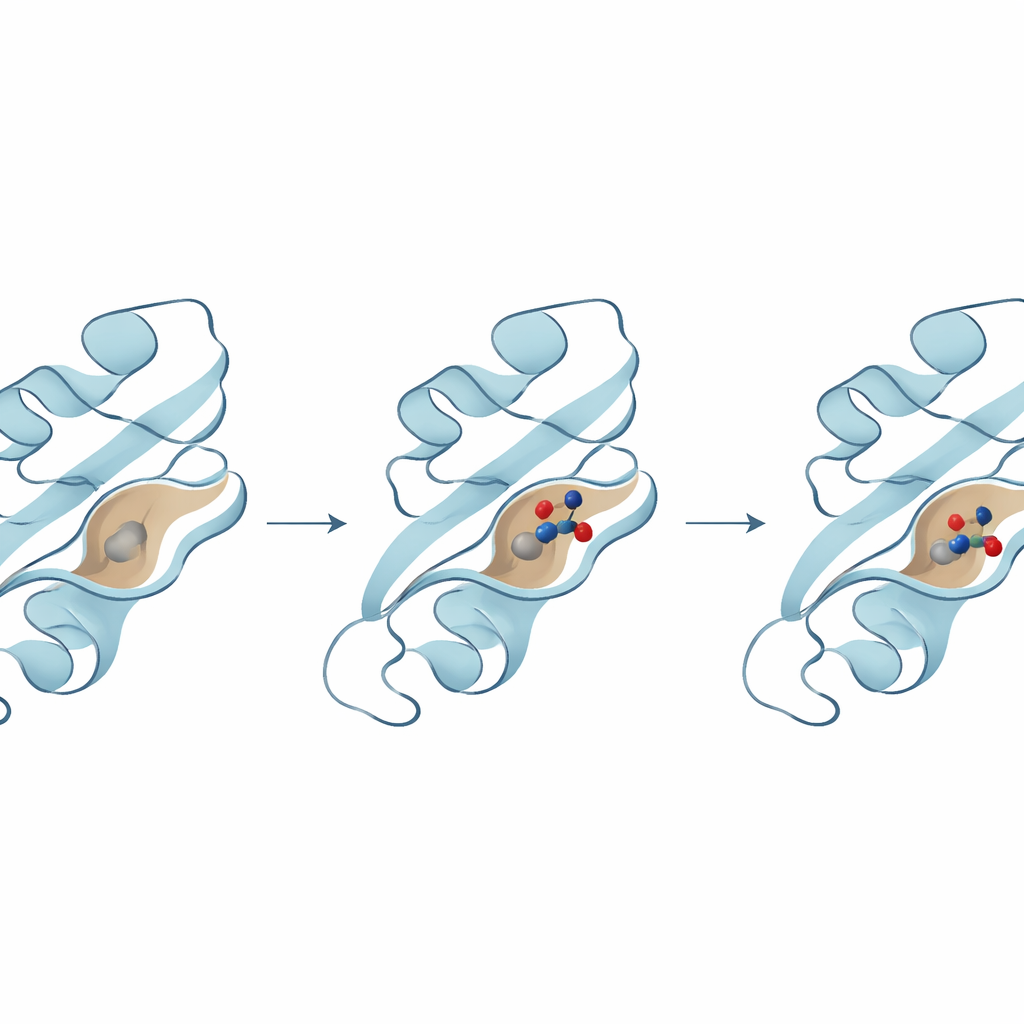
The next step was to see which molecules could fit snugly into an important pocket on the B8R surface, where it would normally interact with interferon-gamma. The team used molecular docking, a technique that tries out billions of possible poses for a molecule in the protein’s pocket and scores how tightly it would bind. Three top candidates emerged, each predicted to form strong interactions with key amino acids in the pocket. One compound in particular showed the strongest binding energy and made several stabilizing contacts with critical residues that help anchor it in place, suggesting it might be especially effective at blocking B8R’s activity.
Watching molecules move over time
Proteins and drug molecules are not rigid; they move, flex, and breathe in watery surroundings. To capture this, the researchers ran long molecular dynamics simulations—virtual movies lasting hundreds of nanoseconds—to see how each protein–compound pair behaved over time. These simulations revealed that the best-performing compound remained deeply buried in the B8R pocket, with the protein adjusting slightly around it while staying structurally stable. By contrast, the other two compounds wandered more, shifted into alternate pockets, or showed greater fluctuations, suggesting weaker or less reliable binding. Additional analyses of flexibility, energy, and large-scale motions all pointed to the same winner: one compound that consistently produced the most stable and energetically favorable complex.
What this could mean for future treatments
The study does not claim to have discovered a finished drug, and no experiments in cells or animals were performed yet. Instead, it delivers a strong, data-driven starting point: a specific small molecule that appears ideally shaped to lock into the monkeypox B8R protein and potentially restore the body’s interferon alarm system. By targeting the virus’s immune-evasion strategy rather than its replication machinery, this approach offers a fresh angle for antiviral therapy. The authors argue that their lead compound should now be tested in the laboratory, where its ability to block B8R and curb mpox infection can be measured directly. If those tests succeed, it could pave the way toward a targeted treatment that helps the immune system see and stop the virus more effectively.
Citation: Ahmad, F., Navid, A., Irfan, M. et al. Discovery of potential antiviral compounds and accelerating the therapeutic discovery against monkeypox virus. Sci Rep 16, 8306 (2026). https://doi.org/10.1038/s41598-026-39427-1
Keywords: monkeypox antiviral, immune evasion, drug discovery, computational modeling, interferon signaling