Clear Sky Science · en
Green DOE based RP-HPLC method for the simultaneous determination of Azelastine and Losartan in spiked human plasma samples
Why this research matters
Many people live with both heart-related conditions, such as high blood pressure, and allergic diseases. Two medicines often used in these situations are azelastine, an anti-allergy drug, and losartan, a blood-pressure medicine that also protects blood vessels. Doctors and researchers need reliable ways to measure how much of these drugs are present in a patient’s blood, both to fine‑tune treatment and to study new combination therapies. This study presents a highly sensitive laboratory test that can measure both drugs at once in human blood plasma, while also cutting chemical waste and making the process more environmentally friendly.
Two drugs, one shared health problem
Chronic low‑grade inflammation and damage to the inner lining of blood vessels lie at the heart of many modern illnesses, including diabetes, high blood pressure and allergic disease. Histamine released from immune cells contributes to allergic symptoms and may also promote artery disease. Azelastine helps by stabilizing these immune cells and blocking histamine’s effects, easing allergy symptoms. Losartan, originally developed to lower blood pressure, reduces strain on the heart and blood vessels and also dampens inflammation and blood platelet activity. Animal studies suggest that using these two drugs together can improve blood vessel function and blood sugar levels more than either drug alone, making the pair an attractive option for future combined therapies.
The challenge of tracking medicines in blood
To safely test and use this drug combination in people, scientists must be able to measure very small amounts of azelastine and losartan in blood samples. Traditional analytical methods often focus on a single drug, require many trial‑and‑error adjustments, and use large volumes of organic solvents that are costly and harmful to the environment. The authors set out to design a single, streamlined test that could separate and measure both drugs in human plasma with high accuracy and precision, and to do so in a way that saves time, solvent and energy.
A smarter way to build the test
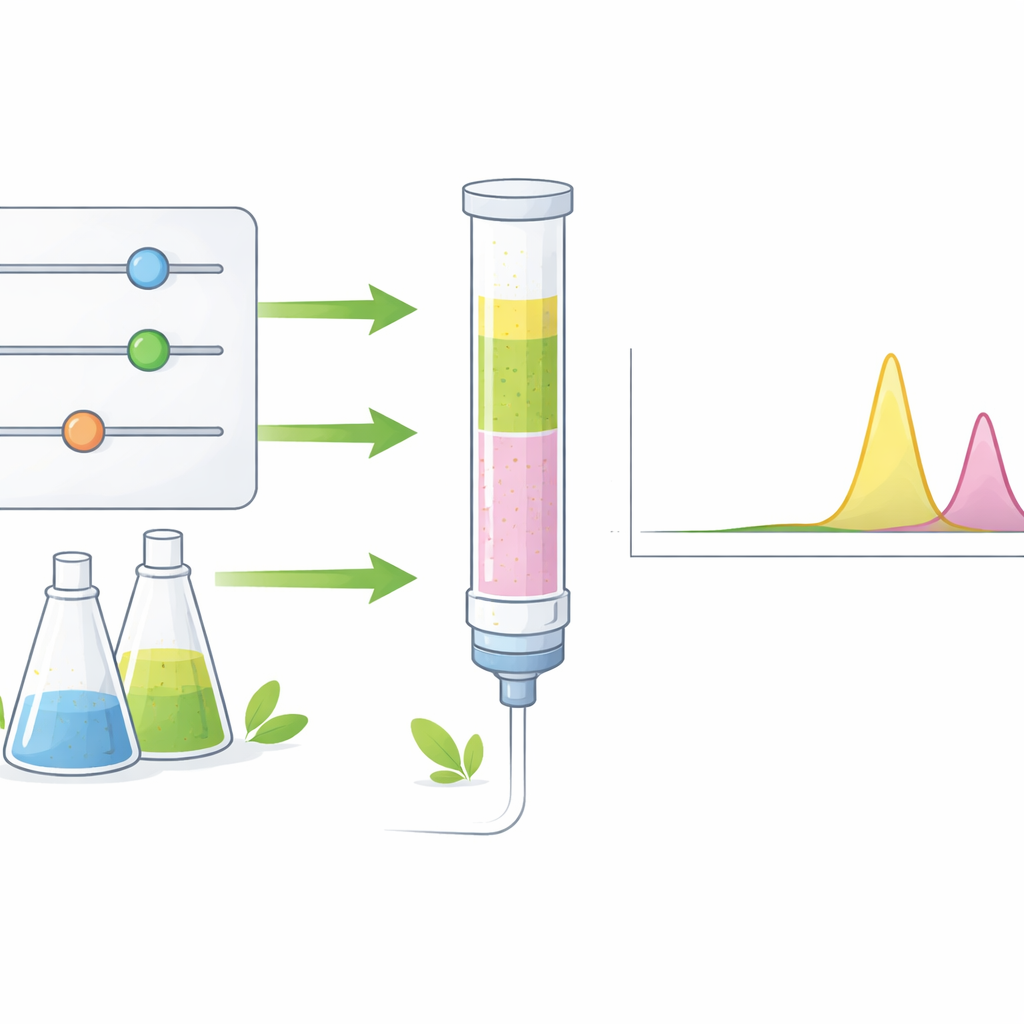
Instead of tuning the method by intuition alone, the researchers used a structured “design of experiments” strategy. They first explored many possible experimental factors, such as solvent type, flow rate, temperature and solution acidity, then identified three that had the greatest impact on performance: the percentage of methanol, the percentage of acetonitrile and the acidity of the water‑based buffer. They then applied a statistical design called a central composite design to test different combinations of these three factors in only 20 carefully chosen experiments. By analyzing how each setting affected peak shape, separation between the two drugs, measurement signal and total run time, they built mathematical models that pointed to the optimal conditions.
How the green test works in practice
Under the final conditions, a mixture of methanol, a small amount of acetonitrile and a mildly acidic phosphate buffer carries the drugs through a chromatography column, where they separate in time before reaching a sensitive fluorescence detector. This setup produces sharp, well‑resolved signals for both azelastine and losartan within about 14 minutes. The method can reliably measure extremely low concentrations of each drug in human plasma, well suited for therapeutic drug monitoring. Validation showed excellent linearity, accuracy, repeatability and robustness, meaning that small day‑to‑day variations do not harm its performance. Importantly, the efficient experimental design reduced the number of trials and the volume of solvent needed to develop and run the method.
Measuring performance with the environment in mind
To judge how environmentally friendly their approach was, the authors applied two modern assessment tools, Complex MoGAPI and AGREE, which score analytical methods on factors such as solvent toxicity, waste generation, energy use and overall sustainability. The new method scored better than conventional high‑performance liquid chromatography approaches that do not use structured experimental design, reflecting its reduced solvent use and fewer optimization runs. Additional tools that rate cost, practicality and day‑to‑day usability indicated that the method is not only green but also simple and affordable enough for routine laboratory work.
What this means for patients and laboratories
In plain terms, the study delivers a laboratory test that can accurately track two important cardiovascular and allergy medicines in human blood using less time, less solvent and fewer resources. Such a method can support clinical studies of azelastine and losartan combinations, help doctors monitor treatment in hospitalized patients and encourage wider adoption of greener analytical practices. By marrying smart experimental planning with environmental awareness, the work illustrates how laboratories can obtain better data while also reducing their ecological footprint.
Citation: Roshdy, A., Belal, F. & Marie, A.A. Green DOE based RP-HPLC method for the simultaneous determination of Azelastine and Losartan in spiked human plasma samples. Sci Rep 16, 8263 (2026). https://doi.org/10.1038/s41598-026-39426-2
Keywords: therapeutic drug monitoring, azelastine and losartan, green analytical chemistry, blood plasma analysis, high performance liquid chromatography