Clear Sky Science · en
Analysis of pressure flotation mechanisms and their practical application in the treatment of metal-containing wastewater
Cleaning Dirty Water for a Metal-Hungry World
Industrial plants that plate, etch, or machine metal parts often leave behind wastewater laced with toxic metals. Letting this water flow untreated into rivers and lakes is not an option, yet current treatment methods can be slow, bulky, and energy-intensive. This study explores a faster, more compact way to strip metals from wastewater using tiny bubbles and a clever multi-chamber device that not only protects the environment but can also recover metals as a usable resource.
How Tiny Bubbles Lift Heavy Metals
At the heart of the method is pressure flotation, a process that relies on dissolved air. Wastewater is first adjusted with alkali so that dissolved metal ions, such as iron, zinc, nickel, and chromium, turn into solid, but fluffy, particles called hydroxide flocs. A portion of already treated water is then pressurized and saturated with air. When this air-rich stream is released back into the main tank, the sudden drop in pressure produces countless microscopic bubbles. These bubbles attach to the metal flocs, making them buoyant so they rise to the surface and form a floating sludge that can be skimmed off, leaving cleaner water below. 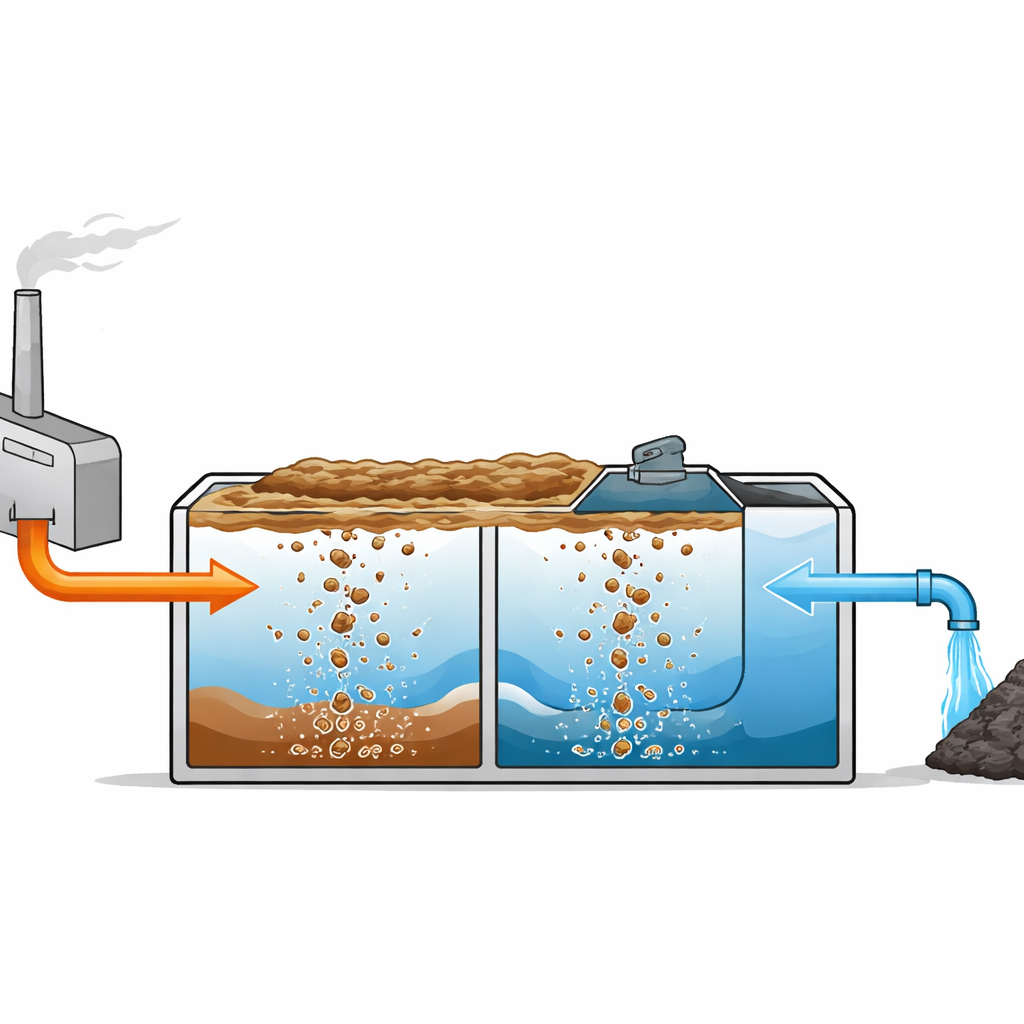
Balancing Bubbles, Particles, and Energy Use
The authors focus on the delicate balance between how much gas is dissolved in the water and how many solid particles need to be removed. Using physical laws for gas solubility and their own equations, they show how pressure, temperature, and recycling of treated water determine the number and size of bubbles. They then compare this with the size, density, and amount of metal flocs. Because these flocs are loose and full of trapped water, their overall density is only slightly higher than that of water. The analysis reveals that in realistic conditions, even a small number of bubbles per floc is enough to make the flocs float. This means that the process can work efficiently at relatively low air input, as long as the flocs are formed under conditions that encourage a porous, “snowflake-like” structure.
A Smarter Multi-Chamber Flotation Unit
Building on this theory, the researchers designed a two-stage flotation unit in which all key components—mixers, air saturator, pumps, pH control, and sludge removal—are integrated into a single compact module. The stages operate at different pH ranges so that groups of metals that form hydroxides at different conditions can be removed separately. In the first chamber, metals such as iron(III), tin, chromium(III), aluminum, and zinc are precipitated and floated; in the second, iron(II), nickel, and cadmium are targeted at higher pH. Tests on real electroplating wastewater showed that metal concentrations dropped by about 98–99% across the two stages, even though the amount of air used per unit of solid (the gas-to-solid ratio) was notably lower than in typical single-stage systems. 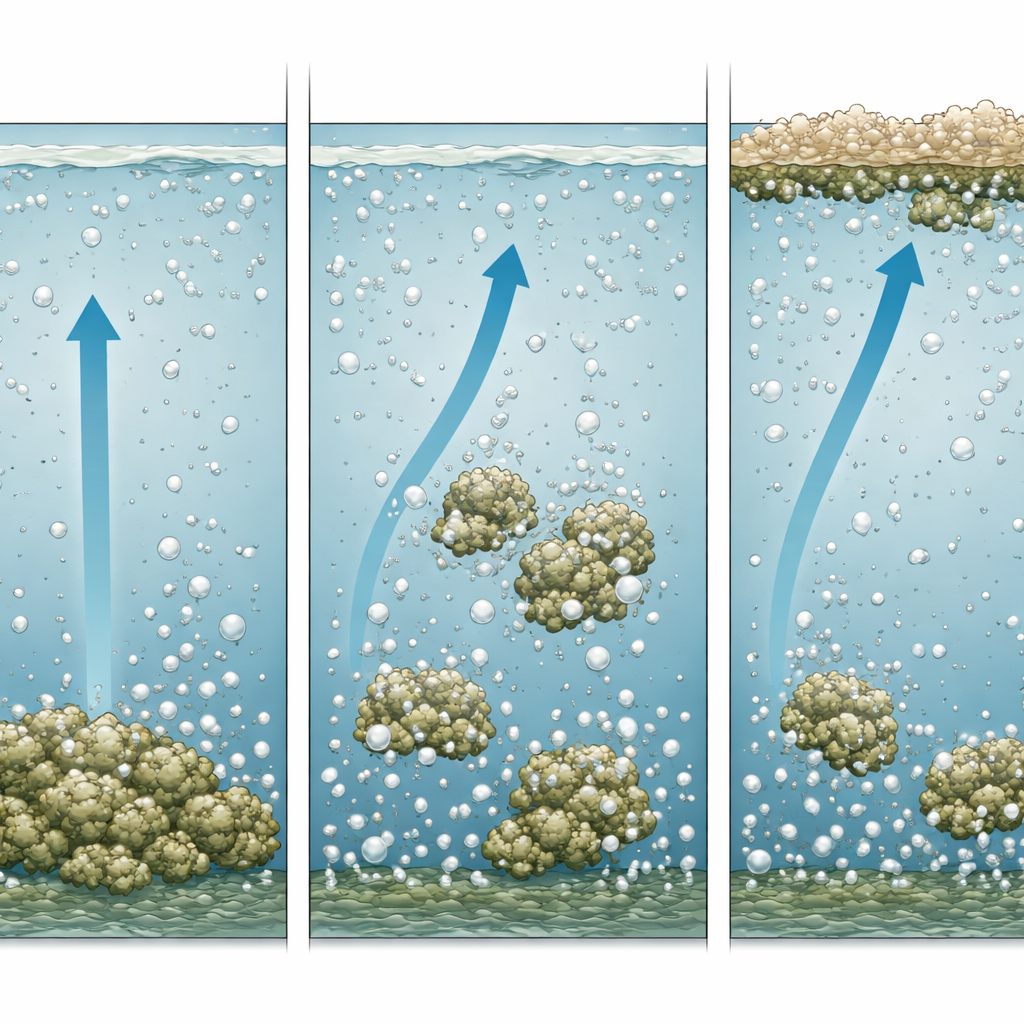
Using Fewer Resources to Do More
One of the most important findings is that it is more efficient to increase the air pressure in the saturator than to simply recycle more water through the system. Higher pressure packs more air into a given volume, yielding more bubbles without the cost of pumping much larger flows. For the tested wastewater, the optimal operating point was a pressure of 0.4 megapascals and a moderate recirculation ratio of 0.3, which together produced enough bubbles to float the metal flocs while minimizing energy use. Under these conditions, the gas-to-solid ratio was only 0.014, substantially below the values usually cited for dissolved air flotation, yet treatment performance remained excellent. The multi-chamber layout improved the chances of bubble–particle contact without needing extra power or chemicals.
Turning Wastewater into a Metal Resource
Because the process produces a dense, metal-rich sludge with less water than conventional settling, it is easier to dewater and can be stored more safely or even processed for metal recovery. For a unit treating 15 cubic meters of wastewater per hour with high metal concentrations, the authors estimate that more than 60 tonnes of metals could be recovered annually instead of being landfilled. In simple terms, the study shows that by understanding how bubbles and flocs interact, engineers can design compact, multi-chamber flotation systems that clean metal-laden wastewater more efficiently, use less energy, and turn a hazardous waste stream into a valuable resource.
Citation: Fylypchuk, V., Kalda, G., Anopolskyi, V. et al. Analysis of pressure flotation mechanisms and their practical application in the treatment of metal-containing wastewater. Sci Rep 16, 8805 (2026). https://doi.org/10.1038/s41598-026-39418-2
Keywords: wastewater treatment, dissolved air flotation, heavy metal removal, water purification technology, resource recovery