Clear Sky Science · en
Loss of mechanical stress induces synovitis, fibrosis and articular cartilage degeneration via distinct synovial cell subsets
Why resting joints can be risky
Many of us have heard that pounding our joints can wear them out, but this study shows that too little use can also harm them. Researchers used a mouse model to explore what happens inside knee joints when movement and weight bearing are drastically reduced for weeks, much like after long-term casting, paralysis, or prolonged bed rest. By tracking microscopic tissue changes and gene activity cell by cell, they uncovered how loss of mechanical stress sets off a chain reaction in the joint lining that ultimately damages the smooth cartilage needed for pain-free motion.
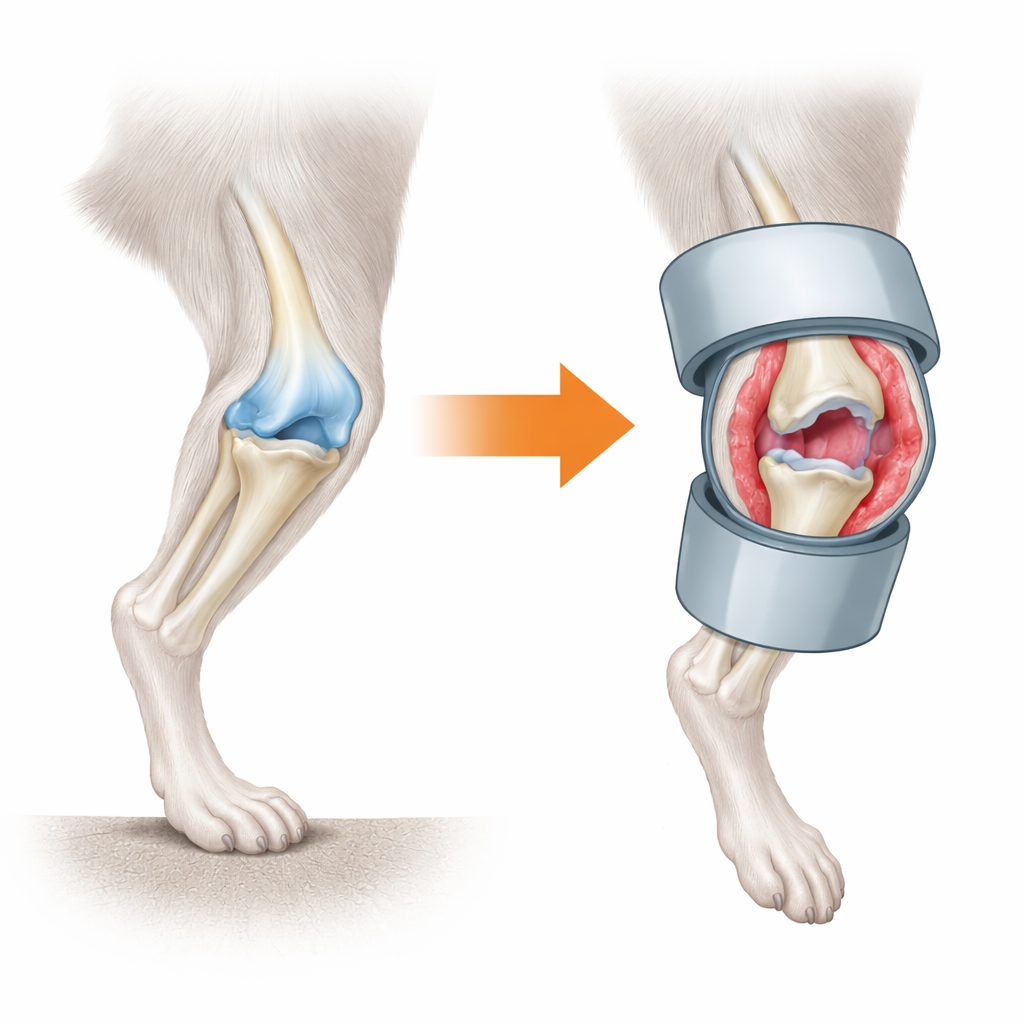
What happens when a knee stops moving
The team created a "minimized mechanical stress" model by immobilizing mouse knees with small plastic cylinders while suspending the animals’ tails so the hind limbs did not bear weight. Within just three days, the inner lining of the joint, called the synovium, became inflamed: its cell layers thickened, new blood vessels appeared, and immune cells crowded in. Over the next two weeks, that lining developed fibrous scar-like tissue and the joint grew stiff. Cartilage—the slippery tissue that coats the ends of bones—did not break down immediately. Instead, it began to show roughening and loss of surface material only after the synovial changes were well underway, and this damage worsened over six weeks.
Movement and pressure as joint medicine
The researchers then asked whether these changes could be reversed. After two weeks of immobilization and unloading, some mice had only joint motion restored while remaining unloaded, and others regained both motion and weight bearing. In both groups, synovial inflammation eased, and the lining thinned back toward normal, showing that simple movement strongly influences the health of the joint lining. Cartilage, however, recovered best when both motion and load were restored. At the molecular level, genes linked to healthy cartilage building were sharply reduced during disuse, but largely rebounded when the joints were again allowed to move and carry weight. This suggests that cartilage depends on a combination of gliding motion and gentle pressure to maintain its structure.
How joint lining cells drive cartilage damage
Digging deeper, the scientists examined which genes turned on in the synovium as damage progressed. They found rising activity of genes tied to fibrosis, tissue remodeling, and enzymes that can break down cartilage. Many of these signals came from immune-related messengers, such as inflammatory cytokines and growth factors, that can influence nearby cartilage cells. Single-cell RNA sequencing revealed that disuse reshaped the cellular cast inside the synovium. Two unusual cell groups expanded in immobilized joints: myofibroblast-like cells specialized in producing fibrous matrix, and a subset of macrophages that expressed high levels of inflammatory molecules and cartilage-degrading enzymes. Together, these cells formed a distinct, damage-promoting microenvironment around the joint.
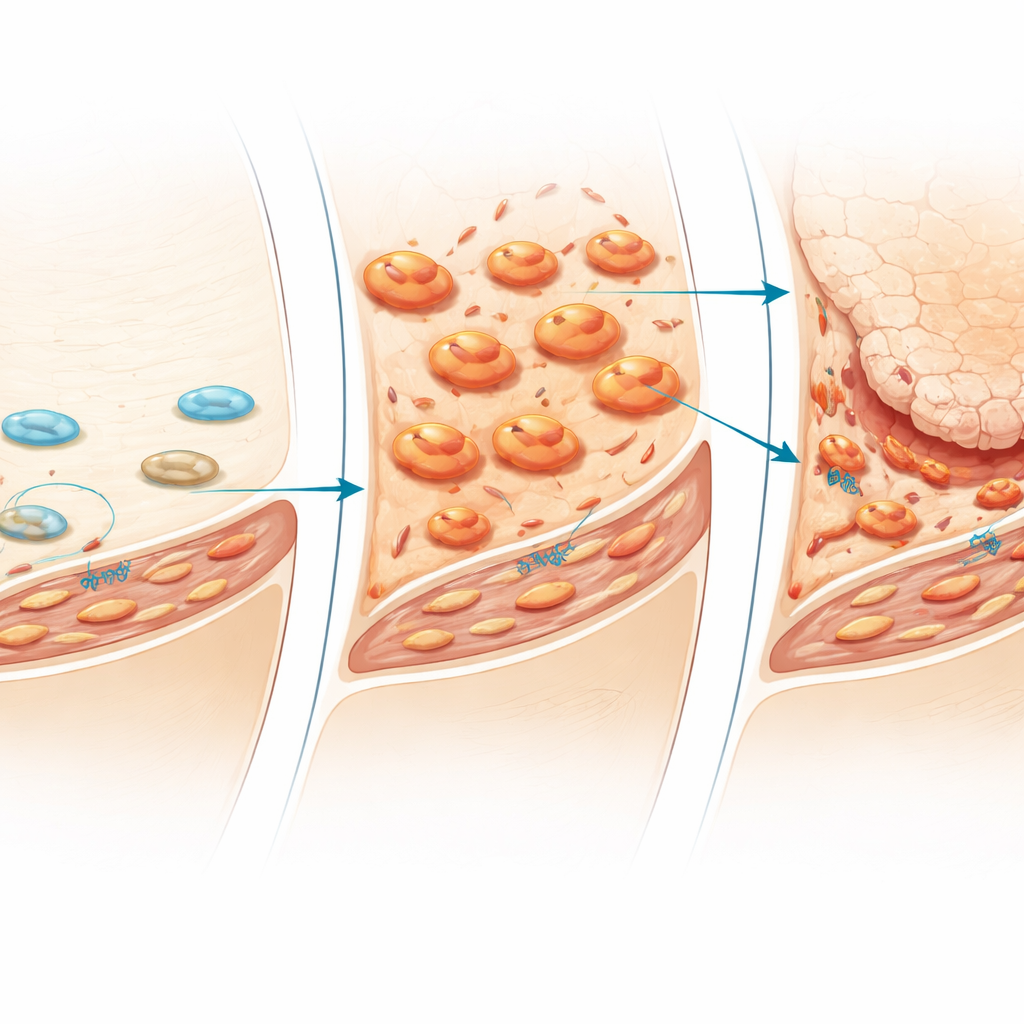
The early role of immune cells
Timing turned out to be crucial. In tissue sections taken at different stages, macrophages surged first in the thin synovial lining just days after mechanical stress was removed, then spread deeper into the tissue. Only afterward did fibroblasts proliferate and thick fibrotic tissue appear. When the team selectively depleted synovial macrophages by injecting liposomes that eliminate these cells, the cascade changed dramatically: inflammation and fibrosis in the joint lining were strongly reduced, and cartilage degeneration was markedly less severe. Markers of the myofibroblast subset also dropped. These findings point to macrophages as early organizers of the harmful response to disuse, activating fibroblasts and setting the stage for later cartilage breakdown.
What this means for protecting joints
Overall, the study shows that joints rely on regular motion and appropriate mechanical loading to keep the synovium and cartilage in balance. When that mechanical input disappears, specialized immune and fibroblast cells in the synovium shift into an inflammatory, fibrotic state that gradually erodes cartilage. Importantly, restoring movement and weight bearing can dial this process back, especially if done before damage is advanced. For people facing long periods of immobility—from casts, paralysis, or extended bed rest—these results underscore the value of early, guided joint motion and safe loading, as well as the possibility of future drugs that target the macrophage–fibroblast axis to prevent lasting joint injury.
Citation: Ishikura, H., Okada, H., Kin, Y. et al. Loss of mechanical stress induces synovitis, fibrosis and articular cartilage degeneration via distinct synovial cell subsets. Sci Rep 16, 8007 (2026). https://doi.org/10.1038/s41598-026-39416-4
Keywords: joint immobility, synovitis, cartilage degeneration, mechanical loading, macrophages