Clear Sky Science · en
DCTPP1 drives immunosuppression and poor prognosis in breast cancer by promoting M2 macrophage polarization
Why the Immune System Matters in Breast Cancer
Breast cancer is not just a cluster of rogue cells; it lives within a busy neighborhood of immune cells, blood vessels, and supporting tissue. This neighborhood can either help the body fight the tumor or quietly assist the cancer in growing and spreading. The study summarized here asks a practical question: can a single molecule inside breast cancer cells tip this neighborhood toward helping the tumor, and could that molecule become a new marker to guide treatment and prognosis?
A Cellular Gatekeeper with a Dark Side
The researchers focus on a protein called DCTPP1, an enzyme that helps keep the building blocks of DNA in balance in fast-growing cells. Using large public cancer databases and laboratory samples from breast tumors, they found that DCTPP1 is consistently higher in breast cancer tissue than in normal breast tissue, at both the gene and protein levels. Women whose tumors had more DCTPP1 tended to have worse clinical features, such as greater lymph node involvement and more advanced stage, and they were more likely to die sooner from their disease. These patterns held up even after the authors adjusted for other risk factors, suggesting that DCTPP1 itself is a strong indicator of poor prognosis.
How Tumor Neighbors Get Swayed
A central theme of the study is how DCTPP1 relates to the immune cells surrounding the tumor. In particular, the authors looked at macrophages, a type of immune cell that can either attack cancer (often called M1-like) or help it grow and hide (M2-like). By using computational tools to deconstruct bulk tumor data, they showed that tumors with high DCTPP1 were rich in M2-like macrophage signatures but poorer in M1-like macrophages and killer T cells. A separate analysis of breast tumor microarrays stained for multiple markers confirmed that tumors with more DCTPP1 also contained more M2-like macrophages nestled within the cancer tissue.
Putting Tumor and Immune Cells in a Shared Space
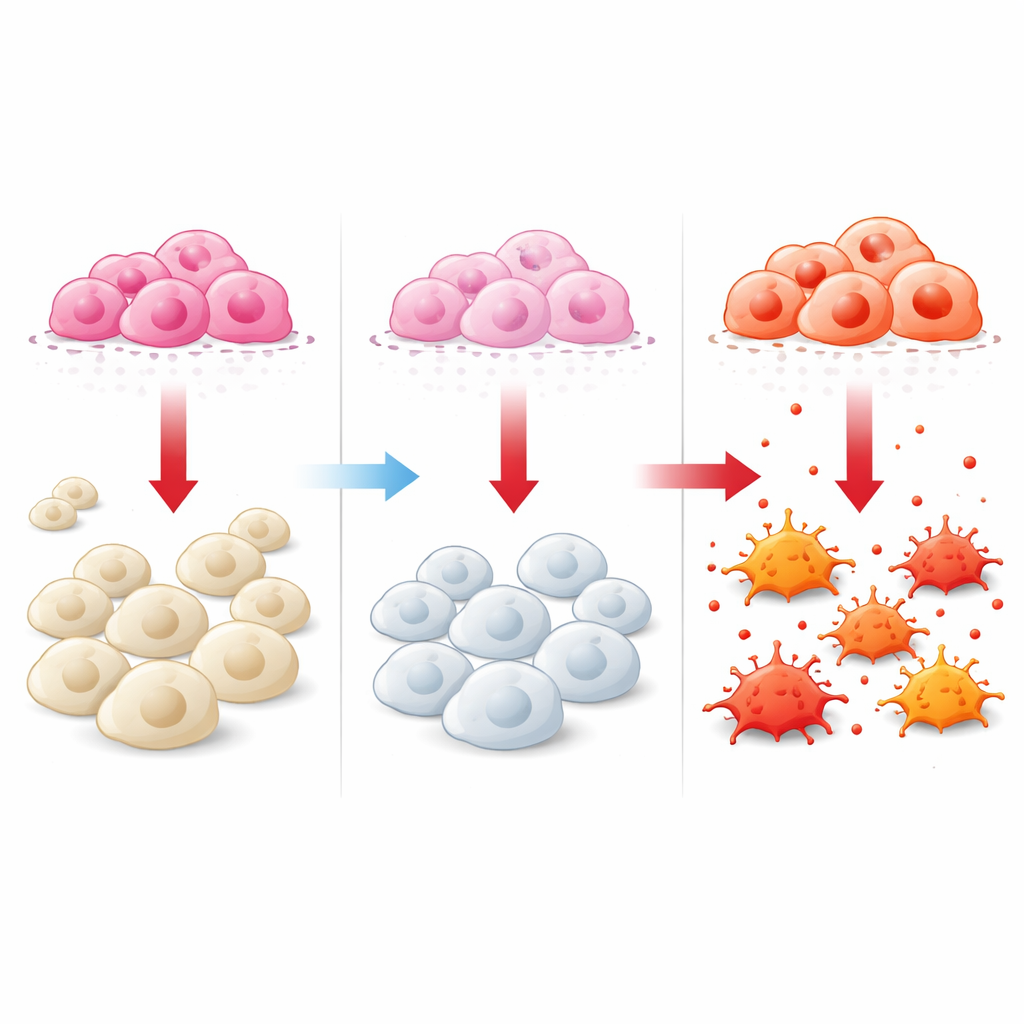
To move beyond correlations, the team set up a co-culture system where human breast cancer cells and human macrophage precursors were grown in neighboring chambers separated by a porous membrane. When cancer cells carried normal levels of DCTPP1, the macrophages below tended to adopt an M2-like, tumor-supporting profile. But when DCTPP1 was knocked down in the cancer cells, the macrophages shifted: genes and surface markers associated with the attacking M1-like state increased, while those linked to the suppressive M2-like state decreased. Flow cytometry, a method that counts and categorizes single cells, confirmed that the balance of macrophage types moved toward more M1-like and fewer M2-like cells when DCTPP1 was reduced in the tumor cells.
Clues to the Underlying Signaling and Treatment Response
Digging into the broader signaling landscape, the authors found that genes tied to immune activity and inflammation changed alongside DCTPP1 levels. High DCTPP1 was linked to gene programs involving DNA repair and to pathways, such as Wnt/β-catenin and MYC, that are known to encourage cancer growth and to blunt immune attack. Tumors with lower DCTPP1 tended to express more immune checkpoint molecules and scored better on a composite measure that predicts success with checkpoint-blocking drugs like anti-PD-1 and anti-CTLA-4 antibodies. This suggests that patients whose tumors have less DCTPP1 might be better candidates for modern immunotherapies.
What This Means for Patients and Future Therapies
In straightforward terms, this work points to DCTPP1 as both a warning light and a possible lever in breast cancer. High levels of this enzyme mark tumors that are more aggressive and that sit in an immune landscape dominated by macrophages that help rather than fight the cancer. Lowering DCTPP1 in cancer cells in the lab nudged nearby immune cells into a more hostile stance against the tumor. While more studies, especially in animals and clinical settings, are needed, these findings suggest that measuring DCTPP1 could refine prognosis and that targeting it might one day help rearm the immune system and improve responses to immunotherapy.
Citation: Chi, J., Liu, W., Zhai, Z. et al. DCTPP1 drives immunosuppression and poor prognosis in breast cancer by promoting M2 macrophage polarization. Sci Rep 16, 8767 (2026). https://doi.org/10.1038/s41598-026-39407-5
Keywords: breast cancer, tumor microenvironment, macrophage polarization, immunotherapy, biomarkers