Clear Sky Science · en
Optimization of THP-1-CAR monocytes utilizing CD32a signaling phagocytosis for antigen-specific T cell activation
Teaching the Immune System New Tricks
Modern cell therapies already reprogram immune cells to hunt down cancer, but most rely on T cells, which can struggle in the harsh environment of solid tumors. This study turns instead to another powerful defender—macrophages, the body’s professional “eaters”—and asks how to rewire them so they not only swallow harmful cells or viruses more efficiently, but also call in T cells for a more lasting, targeted attack. The work reveals design rules for building better chimeric antigen receptor (CAR) macrophages that could be applied to both infections like COVID-19 and hard‑to‑treat solid tumors.
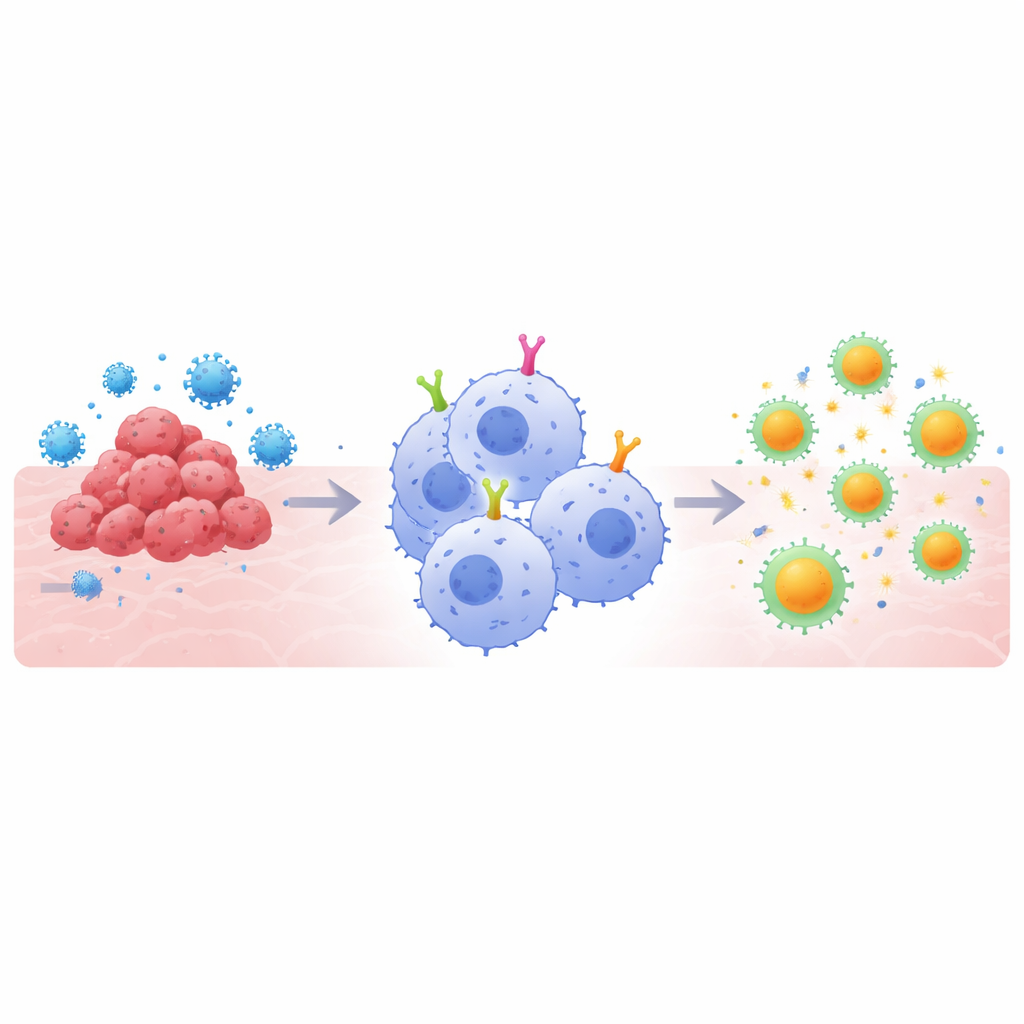
Why Super-Eater Cells Matter
CAR-based therapies work by outfitting immune cells with custom-made surface receptors that recognize a chosen target, such as a tumor protein or a viral spike. T-cell versions have transformed treatment for certain blood cancers, but they face obstacles against solid tumors and in some infections. Macrophages offer a different angle: they naturally engulf threats and chop them into pieces that can be shown to T cells, bridging the “innate” and “adaptive” arms of immunity. However, most CAR designs were originally built for T cells, not macrophages. The authors reasoned that to fully exploit macrophages’ strengths, the internal signaling parts of CARs—the segments that tell the cell what to do once a target is bound—would need to be tailored specifically for these eater cells.
Finding the Best Internal Wiring
The team systematically compared several internal signaling domains inside CARs introduced into a human monocyte cell line (THP‑1) that can be turned into macrophages. They focused on domains drawn from receptors that normally drive phagocytosis and inflammation: CD3ζ (the classic T-cell module), CD32a (a receptor that binds antibody‑coated targets), CR3 components CD11b and CD18 (key complement receptors), and TLR4 (a sensor of microbial danger). Using beads and pseudoviruses coated with the SARS‑CoV‑2 spike protein, they measured how well each CAR design enabled cells to engulf targets, produce inflammatory molecules, and subsequently activate human CD8 T cells that had previously seen the virus. They then transferred the most promising designs into CARs targeting mesothelin, a protein overproduced in many solid tumors, to see whether the same rules held in a cancer setting.
Building Better Eaters and Signal Boosters
A clear pattern emerged: CARs based on CD32a consistently drove stronger and faster phagocytosis than those using the conventional CD3ζ domain, despite CD3ζ carrying more of the usual activation motifs. In other words, the “fit” between the signal and macrophage machinery mattered more than sheer signal strength. Adding CR3-derived pieces (CD11b or CD18) on top of CD32a did not further increase eating capacity in most conditions, but it did raise the levels of key inflammatory cytokines such as TNF‑α and IL‑1β. TLR4‑containing combinations went even further in boosting inflammatory signals and the surface marker CD86, which is associated with T‑cell‑stimulating capacity, though this came with trade‑offs: lower CAR levels on the cell surface and somewhat reduced phagocytosis.
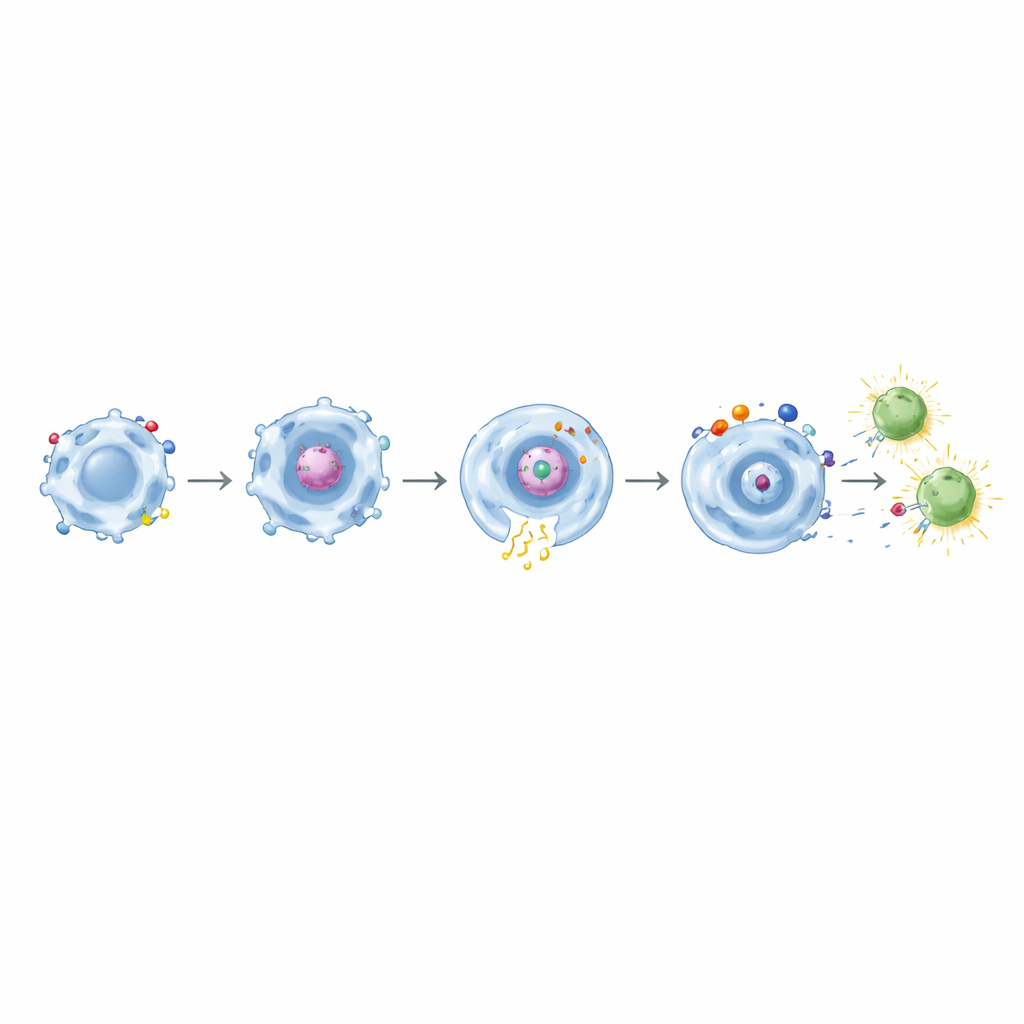
From Eating to Teaching T Cells
Crucially, after macrophages bearing CD32a‑based CARs engulfed spike protein, they were able to activate memory CD8 T cells from people previously vaccinated against COVID‑19. These T cells turned on early activation markers and produced effector molecules such as interferon‑γ and TNF‑α. The effect became even stronger with dual‑domain CARs that paired CD32a with CD11b, CD18, or TLR4. When similar designs targeting mesothelin were tested against engineered tumor cells, they again showed precise, target‑dependent phagocytosis combined with heightened inflammatory signaling. Among these, the CD32a+TLR4 combination stood out as a powerful “tissue alarm,” most effectively boosting cytokine secretion and CD86 while still maintaining specific tumor eating.
What This Could Mean for Future Treatments
To a lay observer, this study shows that engineering macrophages is not a one‑size‑fits‑all problem. By swapping and combining internal signaling modules, scientists can sculpt cells that either focus on efficient cleanup of diseased cells or act more like immune amplifiers that reshape a tumor’s suppressive environment and recruit T cells. CD32a‑based CARs emerged as a strong foundation for such designs, outperforming traditional T‑cell modules in macrophages and working across both viral and tumor targets. Although these results come from lab models and will need to be confirmed in primary human cells and animals, they provide a blueprint for next‑generation CAR macrophage therapies that could better tackle solid tumors and persistent infections by uniting rapid engulfment with precise T‑cell activation.
Citation: Hong, J., Lee, S., Kim, Y. et al. Optimization of THP-1-CAR monocytes utilizing CD32a signaling phagocytosis for antigen-specific T cell activation. Sci Rep 16, 8175 (2026). https://doi.org/10.1038/s41598-026-39406-6
Keywords: CAR macrophages, CD32a signaling, phagocytosis, solid tumor immunotherapy, T cell activation