Clear Sky Science · en
Optical, luminescence and magnetic properties of braunite‒rhodonite nanocomposites synthesized by green aqueous sol‒gel route
Bright Materials from Gentle Chemistry
What if the tiny particles inside a medical scan or a future computer could both glow in vivid colors and respond smartly to magnetic fields—yet be made using water and simple, low‑impact chemistry? This study explores such a possibility using manganese and silicon, two common elements, to create nanometer‑sized particles that shine in green, yellow, and red light while also showing finely tunable magnetic behavior. These dual‑purpose materials could one day help power medical imaging, targeted therapies, and new generations of electronic devices.
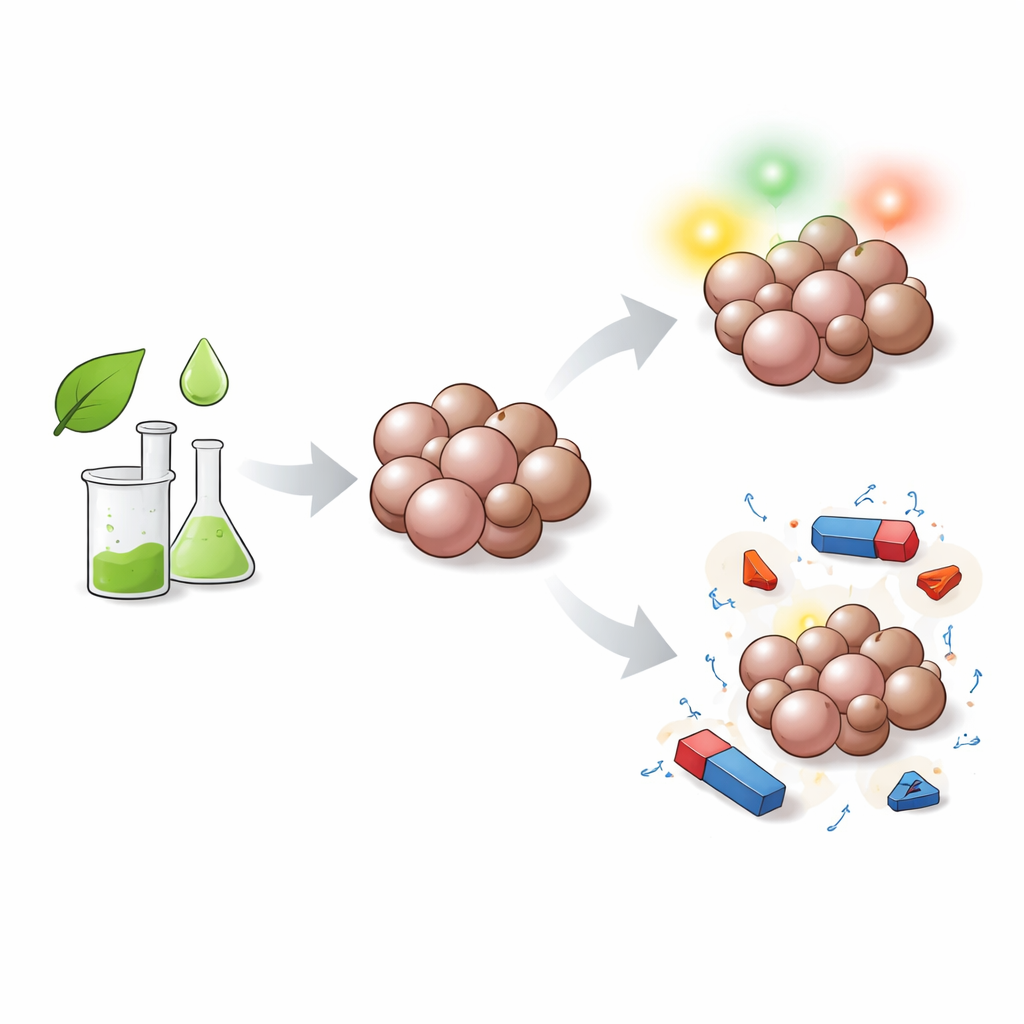
Why Tiny Particles Matter
At the scale of billionths of a meter, matter behaves in unusual ways. When particles get this small, their huge surface area and quantum effects can dramatically change how they absorb light, conduct electricity, or respond to magnets. Engineers and scientists exploit these quirks to design smarter drug carriers, better batteries, and more sensitive sensors. Instead of relying on a single substance, many cutting‑edge technologies now use nanocomposites—blends of more than one material at the nanoscale—to combine and amplify useful traits that no single ingredient can provide alone.
Building Nanoparticles the Gentle Way
The researchers focused on a mixture of two manganese‑silicate minerals, braunite and rhodonite, both rich in manganese and silicon. Rather than using extreme conditions or harsh chemicals, they adopted a "green" aqueous sol–gel route: liquid ingredients containing manganese and silicon were mixed in water with citric acid, slowly converted into a gel, dried, and then gently heated. By choosing three different heating temperatures—600, 750, and 900 degrees Celsius—they could steer how much of each mineral phase formed and how large the resulting nanoparticles became. X‑ray diffraction and high‑resolution electron microscopy confirmed that the final products were well‑crystallized nanocomposites, with particle sizes ranging from about 18 to 42 nanometers and an increasing share of the rhodonite‑like phase at higher temperatures.
Colorful Light from Manganese Centers
To understand how these particles interact with light, the team measured how they absorb and emit radiation from ultraviolet to near‑infrared wavelengths. The nanocomposites showed distinct absorption bands linked to manganese ions in two different charge states, which allowed the researchers to estimate the materials’ band gaps—the energy window that controls how easily electrons are excited. As the heating temperature, and with it the rhodonite content, increased, this band gap widened, indicating more pronounced semiconducting behavior. When excited with ultraviolet light, the particles emitted bright visible photoluminescence: tunable green emissions between 525 and 565 nanometers, a yellow glow near 584 nanometers, and red light around 619 nanometers. These colors mainly arise from manganese ions in different local environments inside the crystal lattice, with higher temperatures favoring green‑emitting sites.
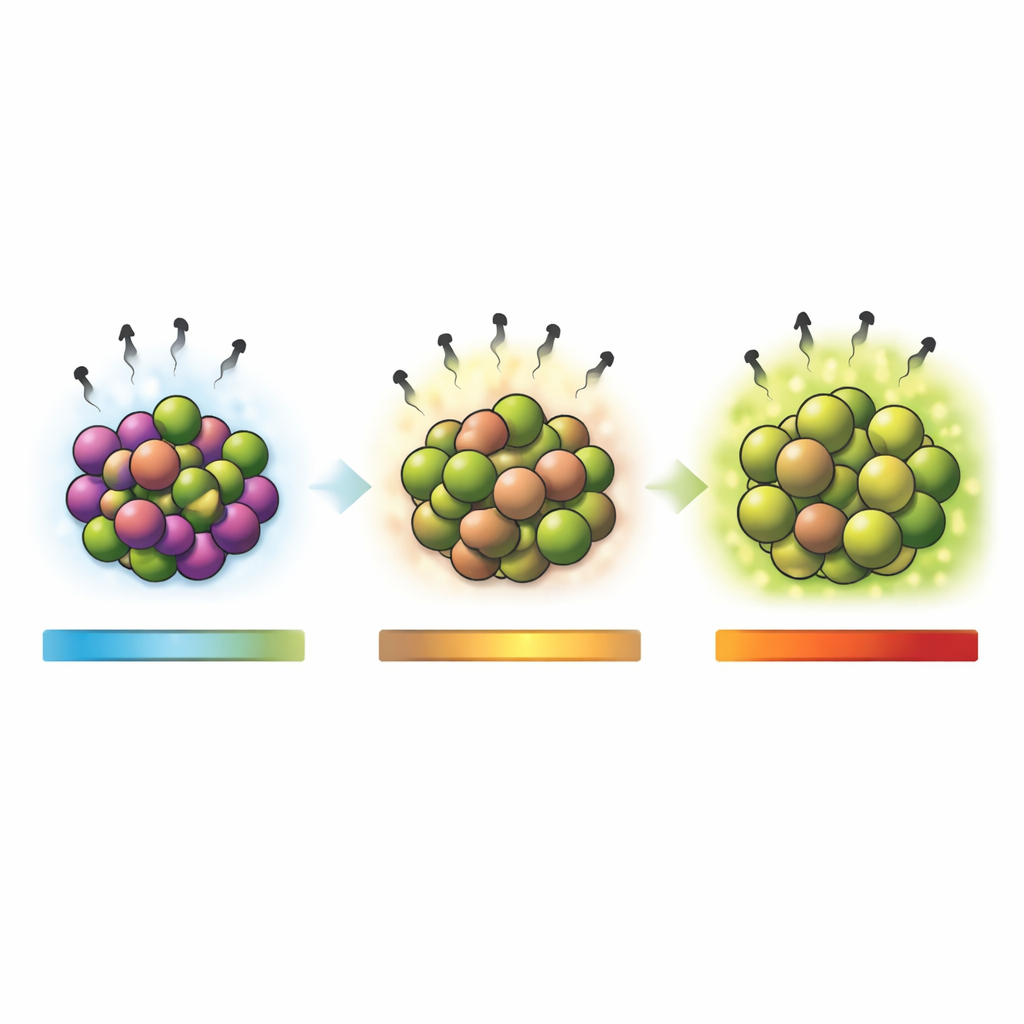
Hidden Magnetism in the Mix
The same manganese atoms that drive the light emission also give the nanocomposites intriguing magnetic properties. Measurements of how the particles respond to an applied magnetic field showed that all samples behave largely as antiferromagnets, where neighboring magnetic moments tend to cancel each other out. At the same time, a clear paramagnetic contribution—an additional, field‑aligned response—increased with rising rhodonite content and particle size. In practice, this means that by adjusting how hot the material is fired, one can fine‑tune the balance between ordered and more easily reoriented magnetic regions. Such control is valuable for emerging "spintronics" technologies that use magnetic moments, rather than electric charge alone, to store and process information, as well as for biomedical uses where magnetic particles can be guided, heated, or used as contrast agents.
Where These Dual‑Role Nanoparticles Could Lead
Taken together, the study shows that a simple, water‑based sol–gel method can yield manganese‑silicate nanocomposites that simultaneously offer tunable visible light emission and controllable magnetic behavior, all governed by the chosen heating temperature. For a layperson, this means that by "cooking" the same basic recipe a bit hotter or cooler, scientists can dial in different colors of light and different strengths of magnetic response without changing the core ingredients. Such versatile, relatively low‑toxicity particles are promising candidates for light‑emitting diodes, optoelectronic components, bio‑imaging probes, and advanced magnetoelectronic and spintronic devices that may one day underpin faster, denser, and more energy‑efficient technologies.
Citation: Nagy, M.G.Y., Ibrahim, F.A. & Abo-Naf, S.M. Optical, luminescence and magnetic properties of braunite‒rhodonite nanocomposites synthesized by green aqueous sol‒gel route. Sci Rep 16, 8945 (2026). https://doi.org/10.1038/s41598-026-39360-3
Keywords: manganese silicate nanocomposites, photoluminescence, antiferromagnetic nanoparticles, green sol–gel synthesis, optoelectronic biomedical materials