Clear Sky Science · en
Microbiological and rheological dynamics of mixed biofilms formed by bacteria and eukaryotic virus
Hidden communities on hospital devices
Many hospital infections begin not with a single germ, but with slimy, invisible communities called biofilms that coat catheters and other medical devices. This study asks an overlooked question with big implications for patient safety: what happens when not only bacteria, but also human-infecting viruses, move into these biofilms together? By watching how a common mammalian virus behaves inside bacterial biofilms, the researchers uncover how these sticky microbial cities might quietly shelter viruses and change their behavior.
Sticky cities of microbes
Biofilms are like microscopic cities built by microbes on wet surfaces—pipes, rocks, teeth, or plastic tubes placed in patients. Bacteria settle, multiply, and secrete a glue-like matrix that protects them from drying, chemicals, and the immune system. In hospitals, such biofilms commonly form on urinary and intravenous catheters, where they can seed bloodstream infections and urinary tract infections that are hard to treat. These biofilms usually contain many different species and even different kingdoms of life, but most research has focused on bacteria alone, leaving the role of viruses largely unexplored.
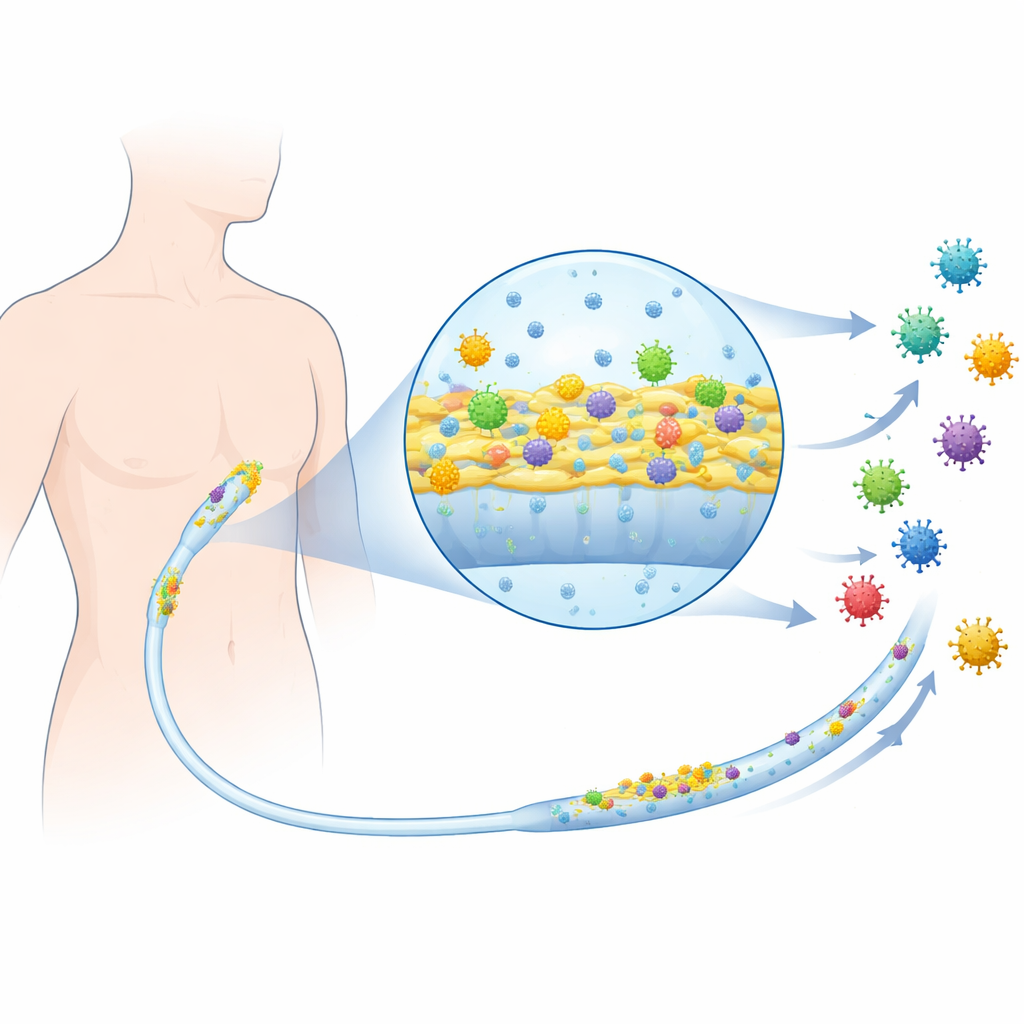
Bringing a mammalian virus into the mix
The team worked with clinical bacterial strains that are known to colonize catheters, including uropathogenic Escherichia coli from urinary infections and other bacteria frequently found in central line infections. They then introduced mammalian reovirus, a well-studied, non-enveloped virus that infects animal cells, as a stand-in for human-infecting viruses more generally. First they checked whether adding virus-containing material affected normal, free-floating bacterial growth. Across many strains, the bacteria grew and survived just as well with or without the virus, showing that the virus was not directly killing the bacteria or acting like an antibiotic.
Biofilms change feel, not size
When bacteria settle into biofilms, the story became more nuanced. For most strains, the total amount of biofilm—measured by a standard stain that quantifies how much material coats a surface—hardly changed in the presence of the virus. A few strains, however, produced slightly more or less biofilm depending on whether viral preparations were present, hinting at strain-specific effects. To go beyond simple quantity, the researchers borrowed tools from materials science and measured how “soft” or “stiff” each biofilm was, much like probing the texture of a gel. They found that every bacterial strain formed its own mechanical signature. Importantly, adding virus could either stiffen or soften these biofilms depending on the strain: in one E. coli strain, virus made the matrix more rigid, while in a Staphylococcus epidermidis strain, virus led to a weaker, more compliant structure.
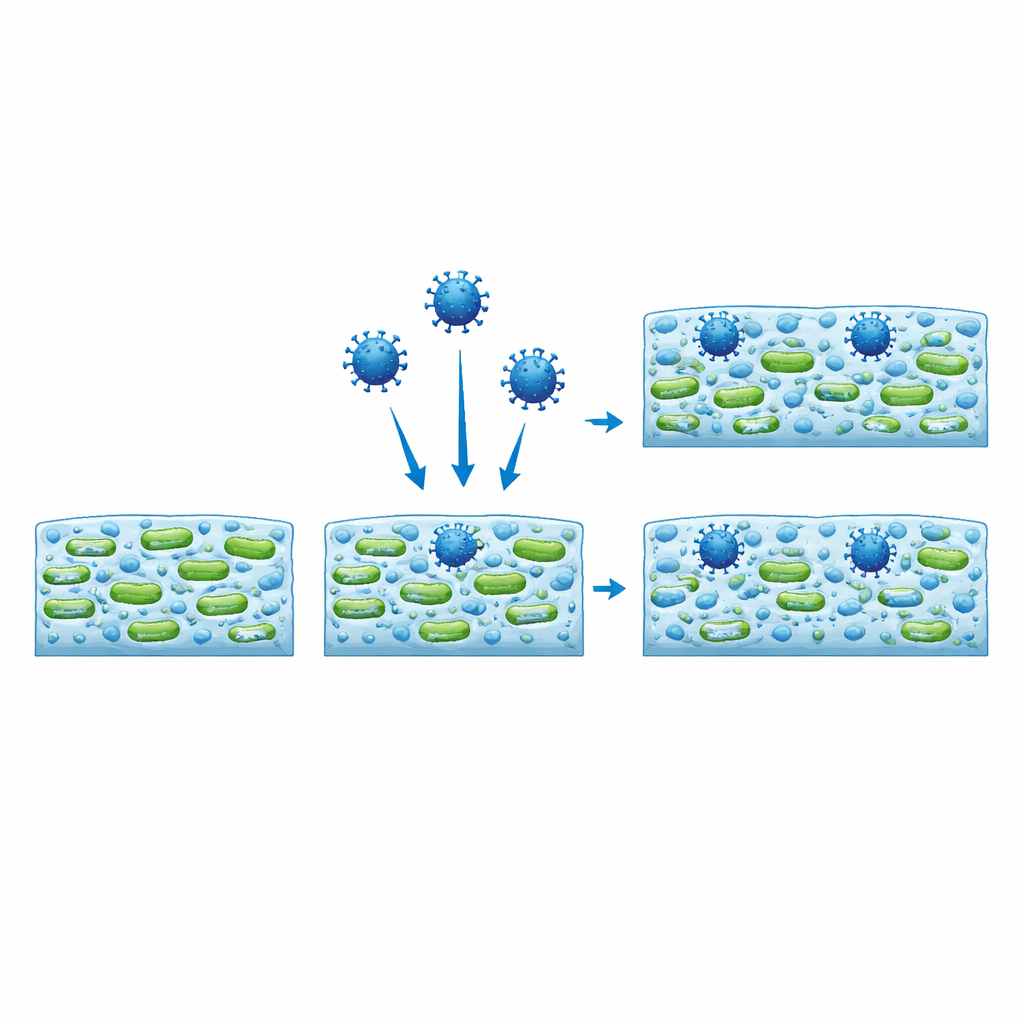
Viruses find shelter and keep their punch
Using fluorescence microscopy, the scientists labeled viral particles so they would glow and followed where they ended up inside the biofilm. Unlike inert, similarly sized plastic beads, which mostly stayed in sparse regions and were washed away, the viruses penetrated deep into the matrix and tended to cluster where bacterial cells were densest. In some cases, the presence of virus even changed the overall shape of the biofilm, turning compact clumps into more web-like networks or vice versa, again depending on the bacterial strain. To test whether these trapped viruses were still dangerous, the authors recovered them from the biofilms and exposed them to mammalian cells. The recovered viruses remained infectious, even though they had been embedded in the bacterial slime for two days.
Why this matters for patients and beyond
To a layperson, the key message is that biofilms on medical devices may not only protect bacteria from treatment—they can also hide and preserve viruses that infect human or animal cells. The study shows that viruses can lodge within the bacterial matrix, subtly altering the structure of the biofilm while staying fully capable of infecting new cells once they are released. This suggests that catheters and other surfaces coated in biofilms could serve as quiet reservoirs where viruses persist, shielded from drying, disinfectants, and immune attack. Understanding these mixed microbial communities may be crucial for designing better materials, cleaning strategies, and therapies that target both bacterial and viral partners in order to reduce stubborn, device-associated infections.
Citation: Gao, J., Abriat, C., Laekas-Hameder, M. et al. Microbiological and rheological dynamics of mixed biofilms formed by bacteria and eukaryotic virus. Sci Rep 16, 8315 (2026). https://doi.org/10.1038/s41598-026-39314-9
Keywords: biofilms, catheter infections, virus–bacteria interactions, reovirus, microbial communities