Clear Sky Science · en
The inner-shell ionization and fragmentation of selenophene at 120 eV
Breaking Molecules with Bright Light
When powerful X‑ray–like light hits a molecule, it can blast out tightly bound electrons deep inside an atom. What happens in the split second after that impact controls how the molecule shatters. This study looks at how a ring‑shaped molecule called selenophene, which contains the element selenium, flies apart after such a hit. Understanding these breakups is important for fields ranging from designing radiation‑resistant materials and drugs to interpreting images from cutting‑edge X‑ray lasers used to watch chemical reactions in real time. 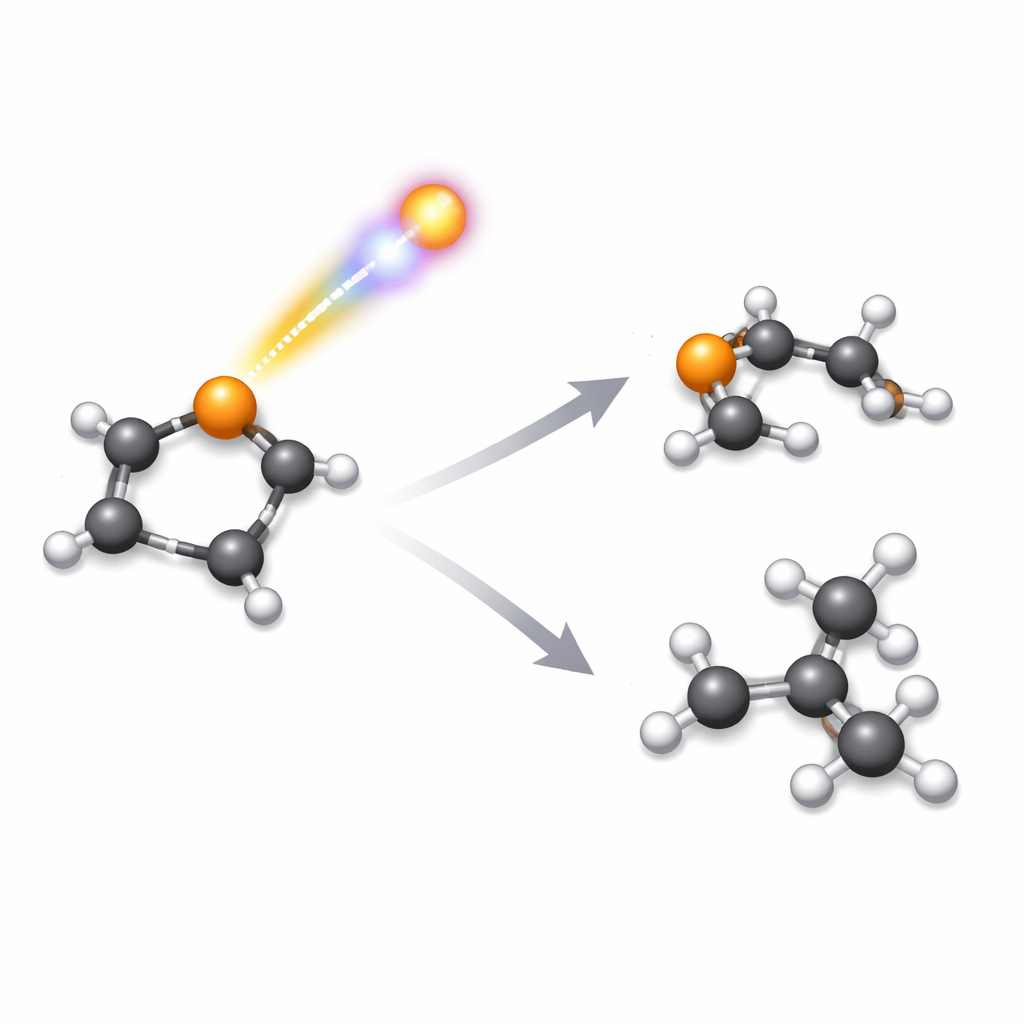
Zooming In on a Selenium Ring
Selenophene is a small, flat ring made of four carbon atoms and one selenium atom, decorated with four hydrogens. Similar rings that swap selenium for sulfur (thiophene) or oxygen (furan) show up in pharmaceuticals, natural products, and advanced electronics. The authors wanted to know how specifically targeting the selenium atom with high‑energy light changes the way this ring falls apart, compared with its sulfur and oxygen cousins. To do this, they used 120 eV photons from a free‑electron laser—bursts of intense, ultra‑short light that can knock out an “inner‑shell” electron from selenium and trigger a rapid cascade of events.
Watching a Charged Molecule Explode
When the inner electron is removed, another electron from higher up in the atom drops down to fill the hole, and in the process one or more additional electrons are kicked out. This chain reaction, known as Auger‑Meitner decay, leaves the entire selenophene molecule with two or three positive charges. Because like charges repel, the atoms are then yanked apart in a violent “Coulomb explosion.” The team recorded the three‑dimensional speeds and directions of the charged fragments using a velocity‑map imaging spectrometer, and then used a statistical method called covariance analysis to figure out which fragments were born in the same breakup events, even when their masses or compositions were very similar. 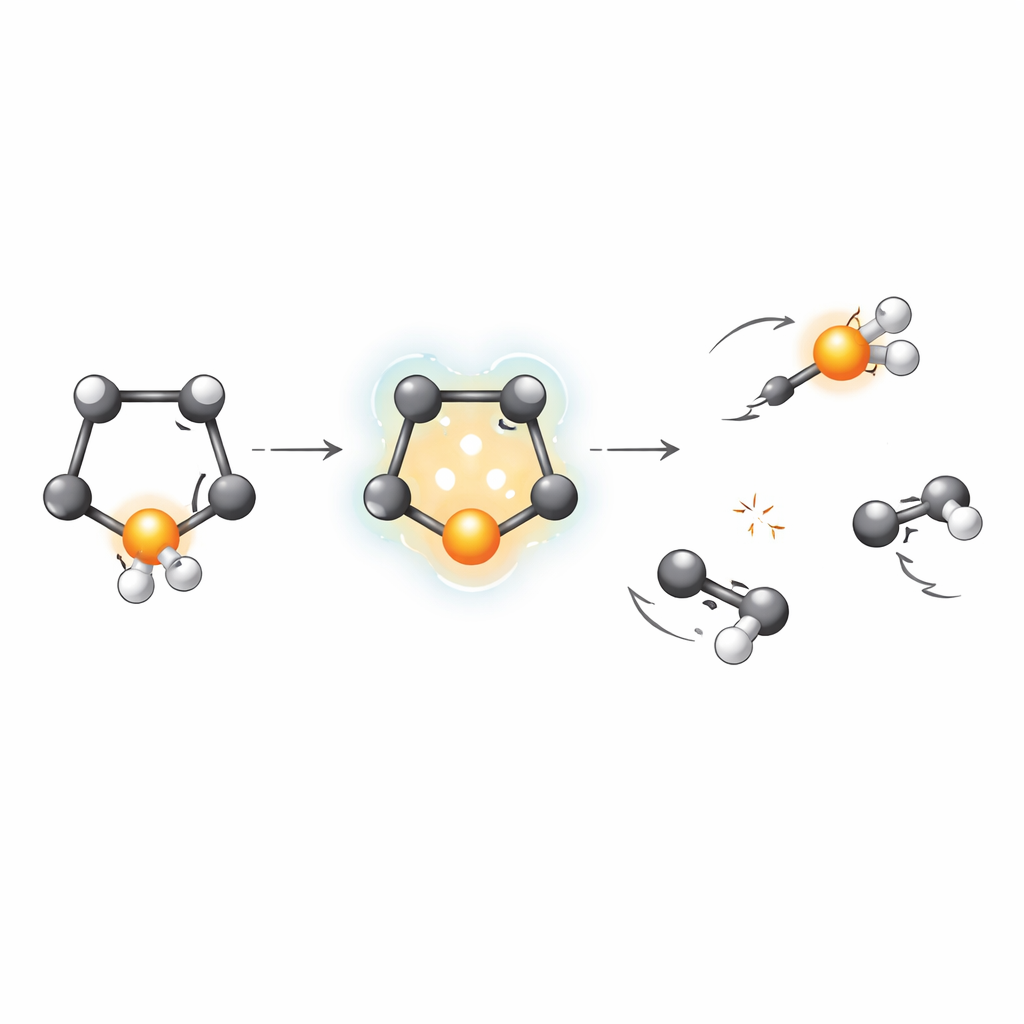
Untangling Dozens of Breakup Pathways
The measurements revealed more than fifty distinct ways that selenophene can fragment after inner‑shell ionization. Many of these involved two main heavy pieces containing carbon and selenium, sometimes with extra hydrogens lost along the way. Others involved three or more fragments, where a selenium‑containing ion flew off together with two separate carbon‑rich pieces. By carefully examining how the fragments recoiled from one another, and by taking advantage of selenium’s natural mix of isotopes, the authors could separate channels that would otherwise look identical in mass. They showed that most of the clearly identified two‑fragment breakups start from a doubly charged selenophene ring, and they quantified how often each route occurs.
Why Selenium Makes a Difference
One of the most striking findings is that selenophene tends to break both of its carbon–selenium bonds. More than half of all two‑fragment pathways involve a selenium‑containing ion separating from a four‑carbon fragment. In contrast, earlier work on thiophene and furan showed that those molecules more often break one bond between the ring and the heteroatom (sulfur or oxygen) and one carbon–carbon bond in the ring, producing different favored fragment pairs. The authors argue that this difference is linked partly to bond strengths: carbon–selenium bonds are weaker than carbon–sulfur or carbon–oxygen bonds, so it costs less energy to snap them both. At the same time, the way charge flows through the molecule after the Auger‑Meitner step appears to be less effective at redistributing charge away from selenium, leaving those weaker links especially vulnerable.
What This Means for Future X‑Ray Movies
To a non‑specialist, the key message is that swapping a single atom in a small ring molecule—from oxygen to sulfur to selenium—dramatically reshapes how that molecule responds when its inner electrons are disturbed by intense light. Here, selenium’s weaker bonding and different electronic structure steer selenophene toward breaking both links to selenium, rather than tearing the carbon ring itself in the same way seen for thiophene and furan. The study also shows that advanced fragment imaging and covariance analysis can reliably disentangle dozens of overlapping breakup routes, even when the pieces look almost identical by mass. These tools will be essential for turning ultrafast X‑ray experiments into clear, atom‑by‑atom “movies” of chemical change in more complex molecules and materials.
Citation: Walmsley, T., Allum, F., Harries, J.R. et al. The inner-shell ionization and fragmentation of selenophene at 120 eV. Sci Rep 16, 9442 (2026). https://doi.org/10.1038/s41598-026-39246-4
Keywords: inner-shell ionization, molecular fragmentation, selenophene, X-ray free-electron lasers, Auger-Meitner decay