Clear Sky Science · en
[¹¹C]Methionine PET uptake kinetics in corticotroph pituitary neuroendocrine tumors
Why tiny brain tumors matter
Cushing’s disease is caused by tiny, hormone‑secreting growths near the base of the brain. Although small, these tumors can disrupt the body’s stress hormone system and cause serious health problems, from weight gain to fragile bones and diabetes. Surgeons can often cure the disease if they know exactly where the tumor sits in the pea‑sized pituitary gland—but finding such minute lesions is challenging. This study explores whether a specialized medical scan, using a radioactive form of the natural amino acid methionine, can reveal subtle differences between tumor tissue and the normal pituitary by watching how quickly the tracer is taken up and cleared over time.
Looking beyond standard brain scans
Magnetic resonance imaging (MRI) is the usual tool for spotting pituitary tumors, but in Cushing’s disease it fails to clearly show the culprit growth in a substantial share of patients. Doctors have turned to positron emission tomography (PET) using radiolabeled amino acids like methionine to improve detection. Current PET protocols focus on images taken 20 to 40 minutes after injection of the tracer, essentially providing a detailed snapshot. However, in some other hormone‑related tumors, the tracer peaks much earlier. The researchers reasoned that these corticotroph pituitary tumors might also show distinguishing early‑time behavior that has been overlooked, and that following the tracer’s rise and fall—its “kinetics”—might sharpen the contrast between tumor and normal gland.
Watching the tracer over time
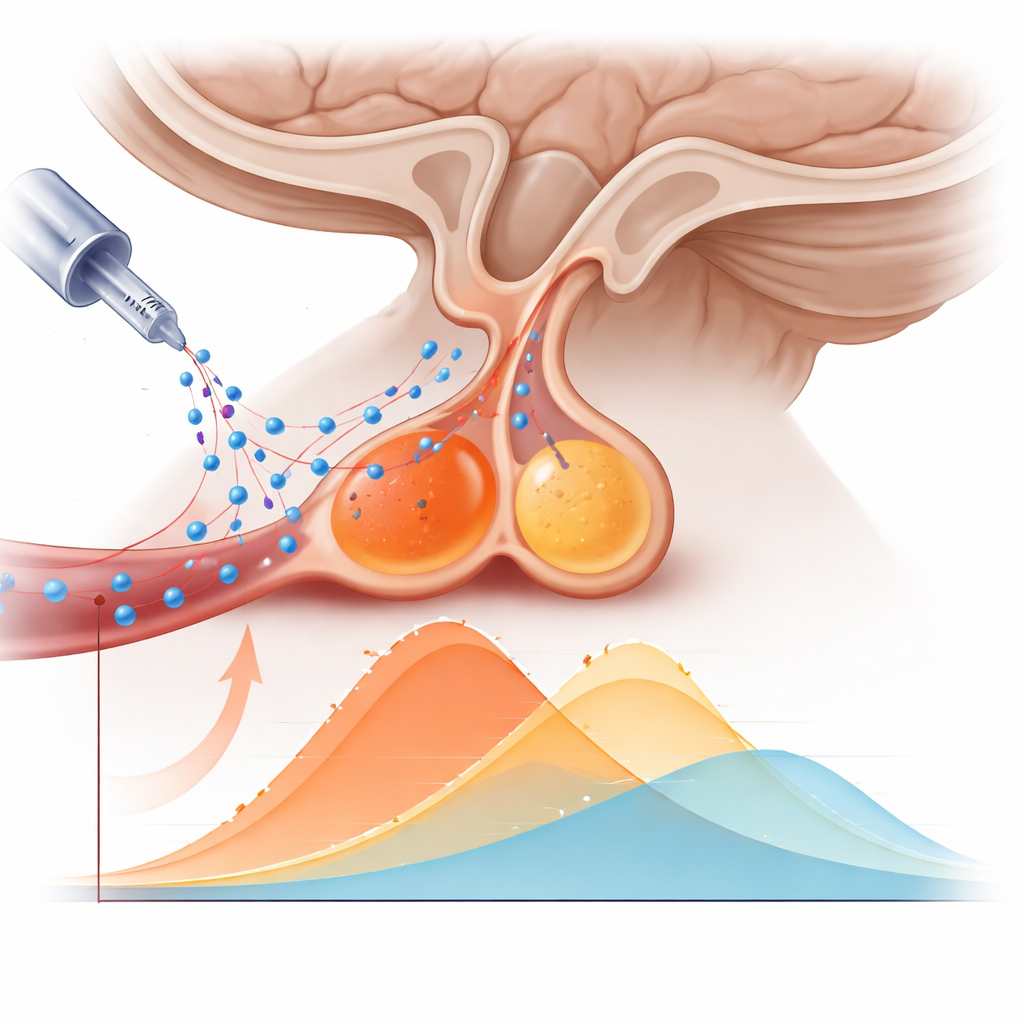
The team analyzed 15 adults with newly diagnosed Cushing’s disease whose pituitary tumors had already been confidently located on MRI and confirmed at surgery. Each patient underwent a combined PET/MRI exam with [¹¹C]methionine. Instead of reconstructing only late images, the investigators rebuilt the PET data into many short time frames covering the first 40 minutes after injection. For each person they outlined two regions: the tumor and the remaining normal pituitary. They then calculated how much tracer was present in each region at every time point, creating time‑activity curves. From these curves, they extracted simple numbers that could realistically be used in clinical practice: how quickly the signal rose at the beginning (early uptake slope), how high it peaked (peak uptake), and how long it took to reach that peak (time‑to‑peak).
How tumors behaved differently from normal tissue
Both tumor and normal pituitary tissue grabbed the methionine tracer quickly in the first minutes and then showed a gradual decline. Throughout the scan, however, the tumors consistently held more tracer than the surrounding gland. The early uptake slope was notably steeper in tumors, and their peak signal was clearly higher. When the researchers tested how well these two features could distinguish tumor from normal tissue, they found moderate‑to‑high discriminative power, suggesting that the numbers carry real diagnostic information. In contrast, the moment at which the signal peaked was quite similar between the two tissues and turned out to be unhelpful for telling them apart.
Does earlier imaging help find the tumor?
The study also asked a practical question: if radiologists look only at very early PET images, can they localize the tumor as well as, or better than, with the usual later images? Two independent readers reviewed early and late scans without clinical information. Early images correctly pointed to the tumor in about two‑thirds of patients, while late images did slightly better, at four‑fifths, but the difference was not statistically significant in this small group. Some tumors were visible only early, others only late, reflecting a trade‑off between rapid but noisy early signals and smoother, higher‑contrast late signals. Overall, simply moving the scan earlier did not clearly outperform the standard timing for everyday localization.
What this means for patients and future scans
For people with Cushing’s disease, the findings suggest that how a pituitary lesion takes up and releases methionine over time carries useful clues about whether it is truly tumor tissue. The steepness of the early rise and the height of the peak signal provided a more reliable fingerprint than timing alone. While early images did not replace conventional late scans for spotting the tumor, adding these kinetic measurements to standard PET may improve doctors’ confidence when surgery plans hinge on subtle imaging differences. The work lays the groundwork for larger studies and more sophisticated modeling approaches that could one day make PET not just a snapshot, but a moving picture that helps guide safer, more precise treatment.
Citation: Flaus, A., Pattée, A., Criton, G. et al. [¹¹C]Methionine PET uptake kinetics in corticotroph pituitary neuroendocrine tumors. Sci Rep 16, 8854 (2026). https://doi.org/10.1038/s41598-026-39219-7
Keywords: Cushing’s disease, pituitary tumor imaging, methionine PET, neuroendocrine tumors, dynamic PET kinetics