Clear Sky Science · en
Progression of peripheral blood mononuclear cell mitochondrial function during the early phase of sepsis in intensive care unit patients
Why our cells’ powerhouses matter in deadly infection
Sepsis, a life‑threatening reaction to infection, sends hundreds of thousands of people to intensive care units (ICUs) every year and kills millions worldwide. Doctors know that many organs falter during sepsis, but the hidden role of tiny “power plants” inside our immune cells—mitochondria—has been harder to pin down. This study followed ICU patients with sepsis through their first week in critical care to see how the energy production of key blood immune cells changed over time, and whether those changes were linked to survival.
Looking inside the blood during sepsis
The researchers focused on peripheral blood mononuclear cells, a major group of white blood cells that help drive inflammation and fight germs. Because they circulate throughout the body and are easy to sample, these cells are a useful window into how the immune system is coping. The team compared 40 adults with sepsis or septic shock in a Dutch ICU to 30 carefully matched hospital controls of similar age and sex who did not have sepsis and were otherwise metabolically healthy. Blood from sepsis patients was taken three times—within two days of ICU admission and again around days three to four and five to six—while blood from controls was sampled once. From each sample, the investigators isolated immune cells, placed them in a standardized nutrient solution, and measured how fast their mitochondria consumed oxygen under different conditions, a direct readout of how hard the cells’ powerhouses were working. 
Immune cell engines running in high gear
Contrary to the idea that sepsis simply “shuts down” cellular power production, the team found that mitochondrial activity in immune cells was consistently higher in sepsis patients than in controls. Across all time points, basal respiration (the basic oxygen use at rest) was elevated. Components tied to useful energy generation—ATP‑linked respiration—as well as the maximum breathing capacity of the mitochondria also tended to be higher, particularly later in the first week. Even proton leak, a form of “wasted” breathing where fuel is burned without making energy, was increased. Together, these findings suggest that, at least in circulating immune cells, the energy machinery is revved up rather than stalled during early sepsis, perhaps reflecting the intense demands of an activated immune response.
Gene signals that match the higher activity
To see whether this higher breathing rate was supported at the molecular level, the researchers examined activity of several genes that encode critical parts of the mitochondrial machinery. In a subset of 10 sepsis patients and 10 matched controls, they measured gene expression for pieces of the oxidative phosphorylation complexes—the protein assemblies that convert fuel into ATP. Two genes in particular, SDHB (part of complex II) and ATP5F1A (part of complex V, the ATP synthase), were significantly more active in sepsis patients at multiple time points, with increases of roughly 40–50% over controls. This molecular “upshift” in core machinery fits well with the functional data, reinforcing the idea that immune cell mitochondria are being pushed to work harder during sepsis rather than breaking down outright. 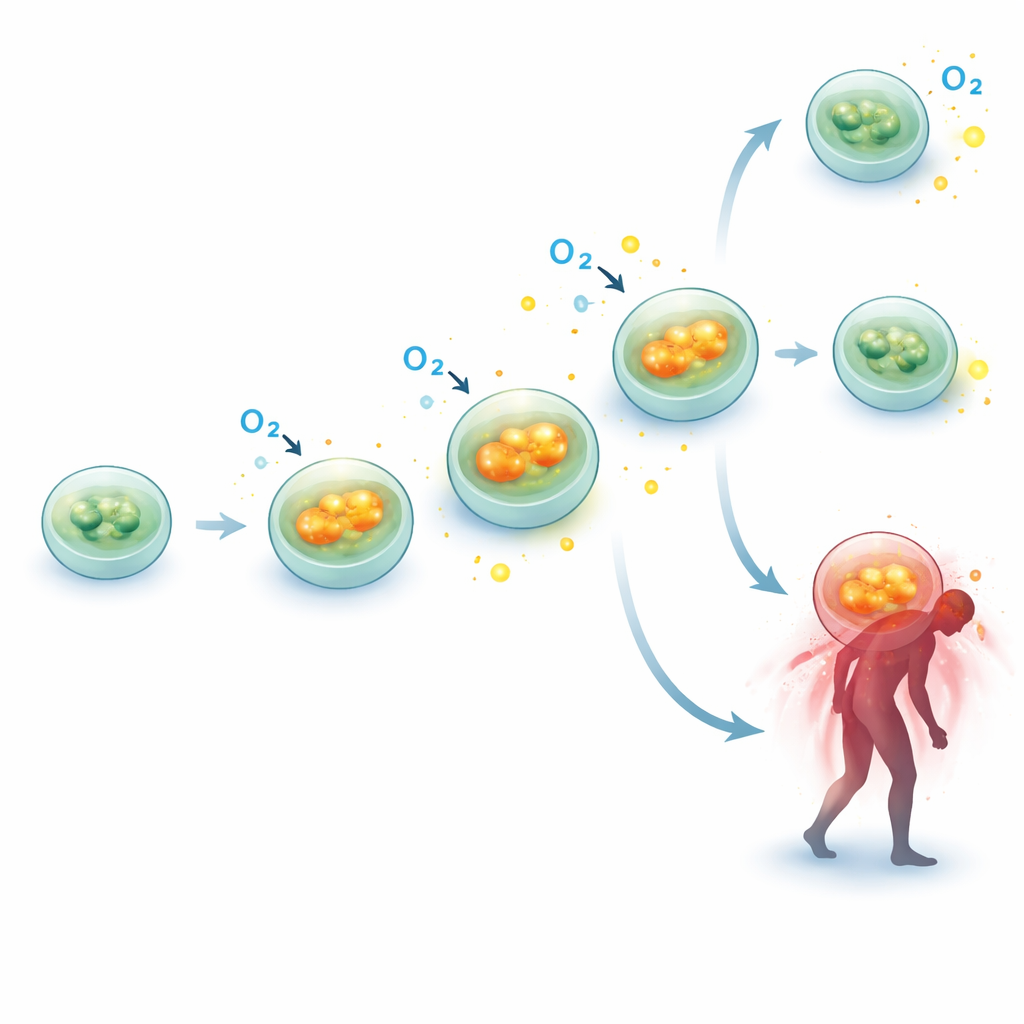
Energy changes tied to who lives and who dies
The most sobering finding was how these mitochondrial changes related to outcome. When the team compared survivors to non‑survivors over three months, they did not see big differences in mitochondrial function at the very first measurement. However, the direction of change over the first week told a different story. In survivors, mitochondrial respiration in immune cells tended to drift slightly downward or stay stable. In patients who later died, those same measures—basal, ATP‑linked, maximal respiration and spare capacity—rose further as the week went on. Statistical analysis suggested that a stronger increase in basal respiration between the first and last measurements was associated with a higher risk of dying within three months, even after accounting for illness severity scores. Because the number of deaths was modest, the authors stress that this link is exploratory and needs confirmation, but it hints that a persistently escalating metabolic drive in immune cells may be a warning sign.
What this means for patients with sepsis
For a non‑specialist, the core message is that in early sepsis, the powerhouses inside circulating immune cells do not simply fail; instead, they appear to switch into overdrive. This extra effort may help fuel the body’s early fight against infection, but in some patients it keeps climbing rather than settling down, and that persistent overactivity in blood immune cells was associated with poorer survival. The study does not yet show whether this mitochondrial push is helpful, harmful, or both at different stages, but it underscores that sepsis is as much a disease of the body’s energy economy as it is of infection and inflammation. In the future, carefully tracking and perhaps gently steering this cellular power usage could become part of how doctors gauge risk and tailor treatments for people battling sepsis in the ICU.
Citation: Moonen, H.P.F.X., Slingerland-Boot, R., de Jong, J.C.B.C. et al. Progression of peripheral blood mononuclear cell mitochondrial function during the early phase of sepsis in intensive care unit patients. Sci Rep 16, 8828 (2026). https://doi.org/10.1038/s41598-026-39202-2
Keywords: sepsis, mitochondria, immune cells, critical illness, cellular metabolism