Clear Sky Science · en
In vitro and in vivo validation of a novel 3D-printed vessel anastomosis device for microvascular surgery
Why tiny blood vessels matter in big surgeries
When surgeons rebuild a jaw after cancer, reattach a severed finger, or transplant tissue from the leg to the face, success ultimately hinges on sewing together hair‑thin blood vessels so the new tissue survives. This delicate step, called microvascular anastomosis, is slow, technically demanding, and vulnerable to clots and leaks that can doom an otherwise perfect operation. This study introduces a 3D‑printed connector designed to make these vessel joins faster, more reliable, and customizable to each patient, potentially improving outcomes while saving operating room time and costs.
The challenge of stitching tiny tubes
In current practice, surgeons reconnect small arteries and veins—often 1 to 3 millimeters wide—by hand, placing a ring of ultra‑fine stitches through the vessel wall. Mastering this skill takes years, and even in expert hands it lengthens the period during which the transplanted tissue has no blood supply, raising the risk of damage. Suture‑free devices already exist, but they struggle with arteries that have thicker, springier walls, may injure the inner vessel lining when edges are folded outward, and come only in a few standard sizes that may not match every patient. The result is a technology gap: surgeons need a quick, artery‑friendly system that can be tailored to individual anatomy without sacrificing strength or safety.
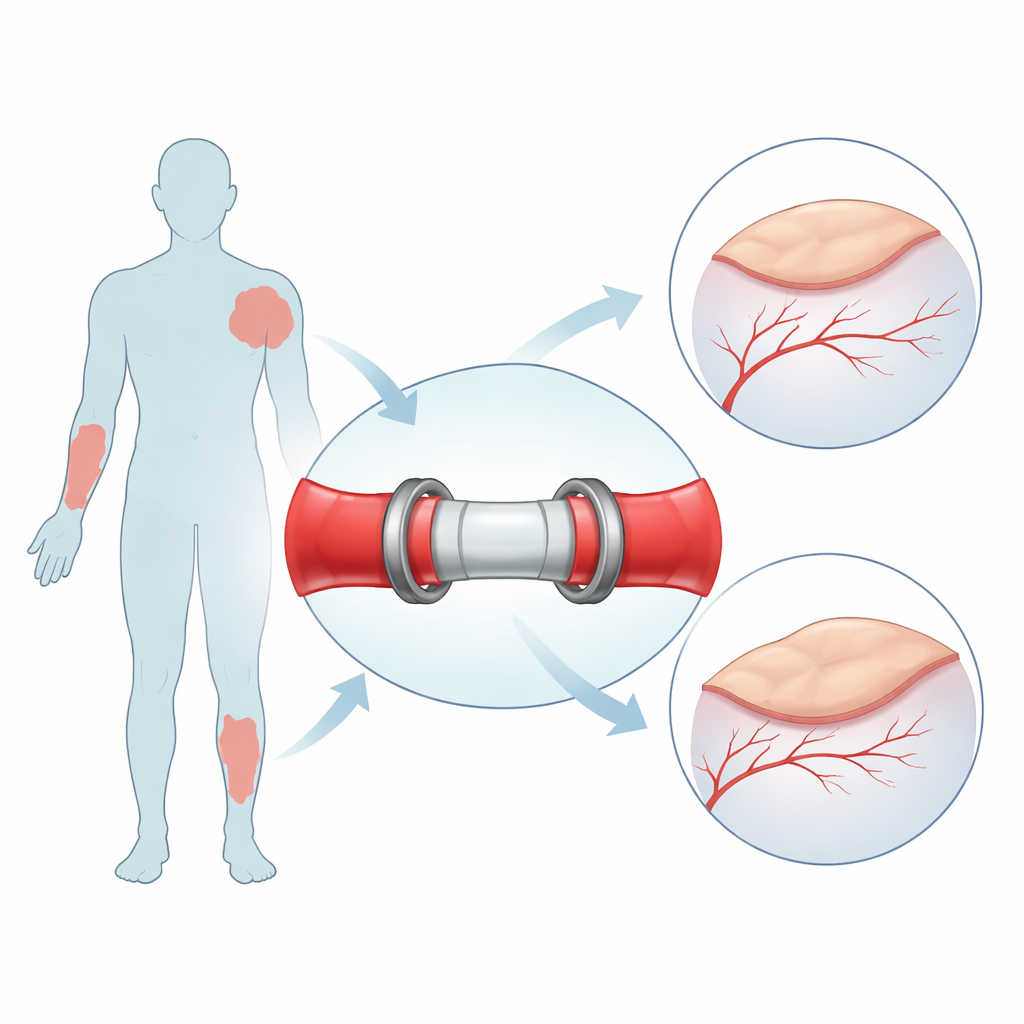
A snap‑together bridge for blood flow
The research team designed a small internal “bridge” that sits inside the vessel rather than folding the edges over an external ring. Each end of the device carries gentle ridges that grip the inside of the artery or vein, while a flexible outer clasp hugs the vessel from the outside like a cuff, holding it firmly in place. Two such halves then snap together with interlocking rings, creating a continuous channel for blood. Because the vessel ends are simply slid over the connector instead of turned inside‑out, precious length is preserved—crucial when every millimeter counts—and the join can be undone if surgeons need to inspect or revise it. The device is produced by high‑resolution 3D printing, allowing its diameter and geometry to be tuned to a patient’s specific vessel size using medical imaging data.
Putting the new connector to the test
To see whether this concept could withstand real‑world demands, the authors printed prototypes using two medical‑grade plastics commonly used in clinics. On the laboratory bench, they compared the new coupler to hand‑sewn joins using synthetic tubes and pig coronary arteries. In pressure tests, traditional sutured connections began to leak at around normal blood pressure, while the couplers held more than five times higher pressures before any fluid escaped. Stretching tests showed that the coupler assemblies tolerated similar forces to stitched vessels before failing, suggesting they are at least as mechanically robust as the standard method. In experiments with human blood vessel cells grown on flat samples of the same materials, the plastics supported cell survival but did not initially encourage strong attachment. A simple oxygen‑plasma surface treatment, which makes the surface more water‑friendly, dramatically improved how well the cells stuck and spread, hinting that modest surface tuning could make the device more welcoming to the vessel lining.

Trials in real blood vessels
The team then moved to pig tissues, first working on vessels removed from the heart and finally in a live animal model. In ex vivo tests, surgeons using the coupler completed a connection in roughly ten minutes—about half the time typically reported for hand sewing similar‑sized vessels. In the live pig, the device was used to bridge a carotid artery in the neck, a high‑pressure, high‑flow vessel chosen as a demanding test. Once the coupler was in place, blood flow resumed immediately with no visible leaks, and simple bedside tests suggested the artery remained open. Over four hours of monitoring, the connection stayed stable with no sign of clot formation or device dislodgement. The flexible outer clasp also acted as a protective sleeve, allowing forceps to pinch the vessel closed during placement without visibly damaging the fragile wall.
What this could mean for future surgery
For now, this 3D‑printed coupler is an experimental concept rather than a clinical product. The study shows that it can seal blood vessels securely, match the strength of traditional stitches, and be deployed quickly in a large animal model, while its surface can be modified to better host living cells. Longer‑term animal studies are still needed to prove that the device remains open for months, does not provoke clots or inflammation, and can be safely adapted to different vessel sizes and locations. If those hurdles are cleared, surgeons may one day replace some of their most painstaking stitches with a quick, snap‑fit connector customized for each patient—shortening operations, reducing complications, and making complex reconstructions more accessible.
Citation: Loh, J.S.P., Feng, KC., Yuan, Y. et al. In vitro and in vivo validation of a novel 3D-printed vessel anastomosis device for microvascular surgery. Sci Rep 16, 8772 (2026). https://doi.org/10.1038/s41598-026-39181-4
Keywords: microvascular surgery, 3D printed medical device, vascular coupler, sutureless anastomosis, reconstructive surgery