Clear Sky Science · en
High-performance adsorption of sulfamethoxazole and phenol using graphene-like carbon derived from glucose
Why cleaning tiny pollutants matters
Many medicines and industrial chemicals that help us in daily life can quietly damage rivers, lakes, and even drinking water when they are not fully removed in treatment plants. This study focuses on two such troublemakers—an antibiotic called sulfamethoxazole and a basic chemical called phenol—and shows how a new, low-cost carbon material made from simple sugar (glucose) can pull these pollutants out of water with remarkable efficiency. 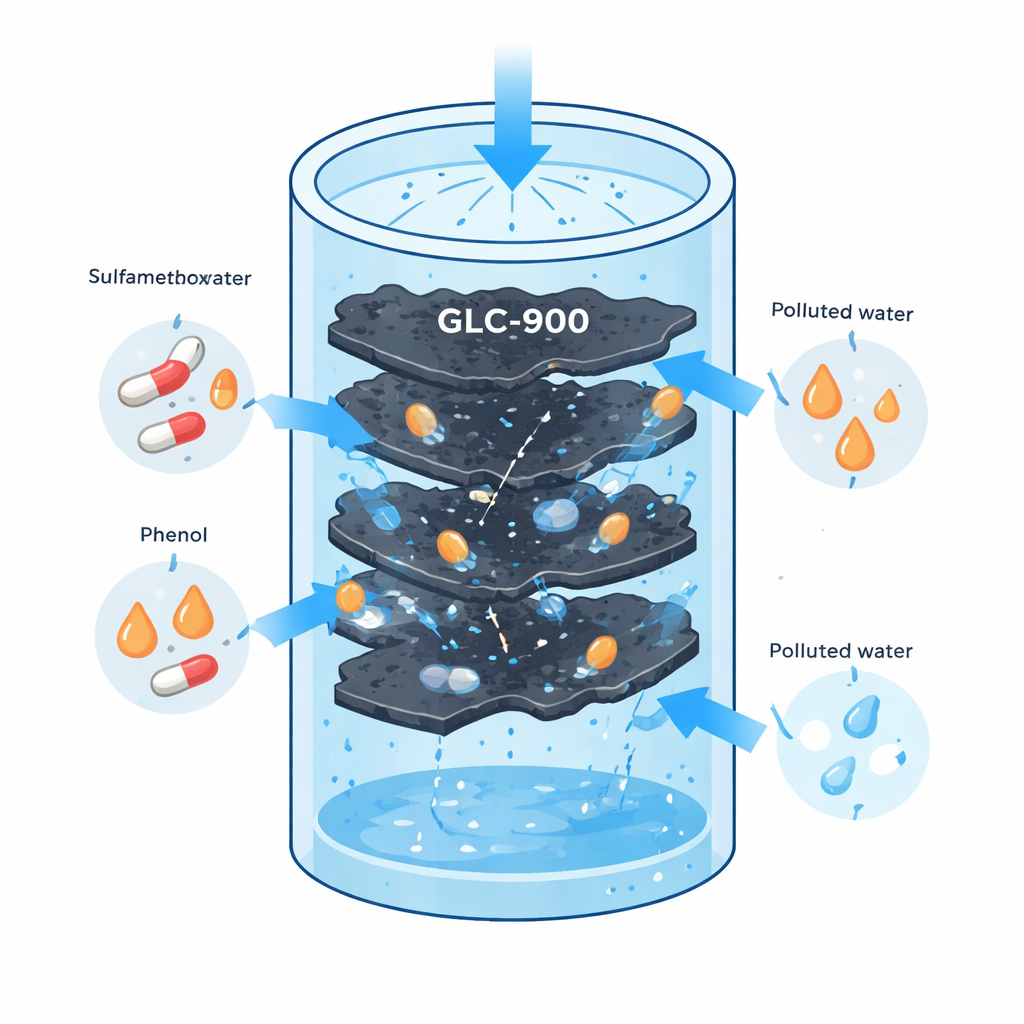
Everyday chemicals that linger in water
Sulfamethoxazole is a common antibiotic used to treat infections in humans and animals. Because our bodies do not fully break it down, much of it is excreted and ends up in wastewater. Treatment plants are not designed specifically for these kinds of drugs, so they can slip through into rivers, groundwater, and even drinking water. Over time, this low-level but constant exposure may help dangerous bacteria become resistant to antibiotics. Phenol is widely used in industries such as plastics, resins, and petroleum processing and is known to be toxic and potentially cancer-causing. Even at very low levels, phenol can harm aquatic life and pose risks to human health, which is why regulators strictly limit its presence in drinking water.
A sponge-like carbon made from sugar
The researchers created a material called graphene-like carbon, or GLC-900, starting from ordinary glucose. They heated glucose together with two helper chemicals: one that helps carve pores into the carbon and another that guides the carbon into thin, layered, graphite-like sheets. By heating to 900 °C in an oxygen-free environment and then washing out the metal, they obtained a black, foam-like solid packed with tiny, connected pores. Careful measurements showed that this material has an extremely large internal surface area—about 935 square meters per gram, roughly the area of several basketball courts squeezed into a teaspoon of powder. This combination of thin sheets and abundant pores makes GLC-900 act like a powerful sponge for dissolved pollutants.
How well the new carbon cleans water
To see how effective GLC-900 is, the team mixed a small amount of it into water containing either sulfamethoxazole or phenol at realistic pollution levels. Within about an hour, the concentrations of both chemicals dropped sharply, showing that the pollutants had been captured on the carbon surface. When the researchers increased the starting concentration, the material continued to perform strongly. Mathematical models that describe how molecules stick to surfaces indicated that the carbon forms a uniform, single layer of adsorbed molecules until its sites are filled, and the maximum capacities were very high: roughly 289 milligrams of sulfamethoxazole and 232 milligrams of phenol per gram of adsorbent. These values are generally better than those of many commercial activated carbons and biochars, meaning less material is needed to clean the same amount of water.
What happens at the microscopic level
Microscope images and surface analyses helped explain why GLC-900 works so well. The material consists of crumpled, interconnected sheets, forming a three-dimensional maze of pores that water and pollutants can easily enter. Chemical tests suggested that the pollutants are held mainly by gentle, non-permanent forces—similar to how water sticks to glass rather than forming a new compound. These include hydrogen bonds between the pollutants and oxygen-containing groups on the carbon, “stacking” interactions between their ring-shaped structures and the flat carbon layers, and the tendency of oily molecules to leave water and cling to less watery surfaces, known as hydrophobic effects. The process is energetically favorable and actually works better at slightly higher temperatures, which is consistent with this kind of physical adsorption. 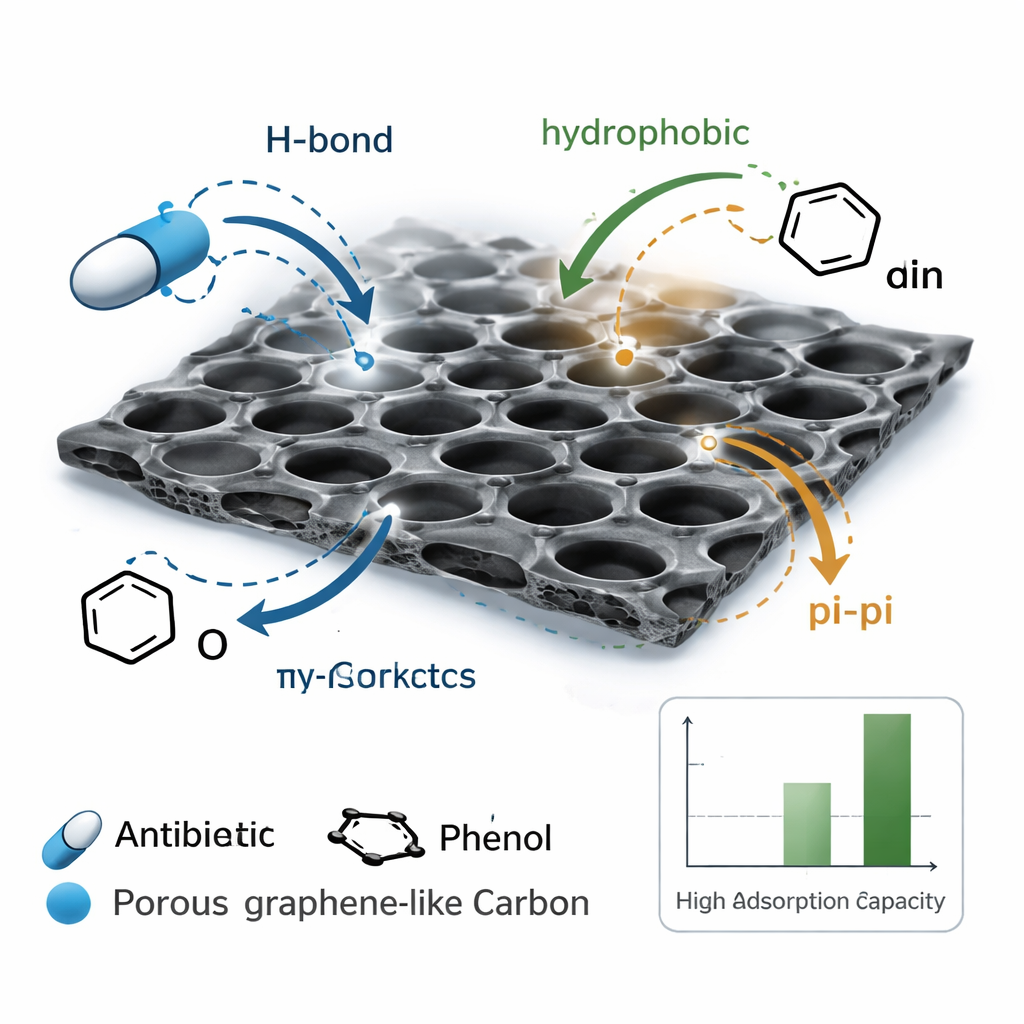
Real-world conditions and reuse
The team also asked how the material would behave in more realistic settings. Natural organic matter, represented here by humic acid—the brownish material that colors some surface waters—competed with the target pollutants for space on the carbon and reduced performance, a challenge shared by most adsorbents. Common dissolved salts, on the other hand, had little effect. When the used carbon was washed with ethanol, it could be reused for several cleaning cycles while still removing more than 90 percent of the pollutants in early rounds. The authors estimated that producing this sugar-derived carbon would cost less per kilogram than many high-grade activated carbons, while avoiding petroleum-based feedstocks and the generation of harmful by-products.
What this means for safer water
In simple terms, this work shows that an inexpensive, sugar-based carbon with a sponge-like structure can quickly and strongly trap both an antibiotic and an industrial chemical from water. Because it is efficient, reusable, and relatively cheap to make, GLC-900 could become a practical tool for treating wastewater from hospitals, farms, and factories before it reaches rivers and drinking-water sources. While more research is needed to test it in continuous flow systems and with mixtures of many pollutants, this study points toward a future where everyday materials like sugar can be transformed into powerful filters that help keep our water cleaner and our ecosystems healthier.
Citation: Lingamdinne, L.P., Angaru, G.K.R., Shrestha, B. et al. High-performance adsorption of sulfamethoxazole and phenol using graphene-like carbon derived from glucose. Sci Rep 16, 7794 (2026). https://doi.org/10.1038/s41598-026-39165-4
Keywords: water purification, antibiotic pollution, phenol removal, graphene-like carbon, wastewater treatment