Clear Sky Science · en
A structured multi-day experimental framework integrating green chemistry for the extraction and characterization of Berberine hydrochloride in undergraduate education
Why this lab story matters
Chemistry students don’t just need to memorize formulas; they need to learn how real substances are obtained, purified, and tested—ideally without creating a lot of hazardous waste. This article describes a redesigned undergraduate laboratory experiment in which students extract a bright yellow medicine-like compound, berberine hydrochloride, from a traditional medicinal plant. The project shows how a common teaching lab can be reorganized to be clearer for students, more environmentally friendly, and less dependent on expensive instruments, while still building strong practical skills.
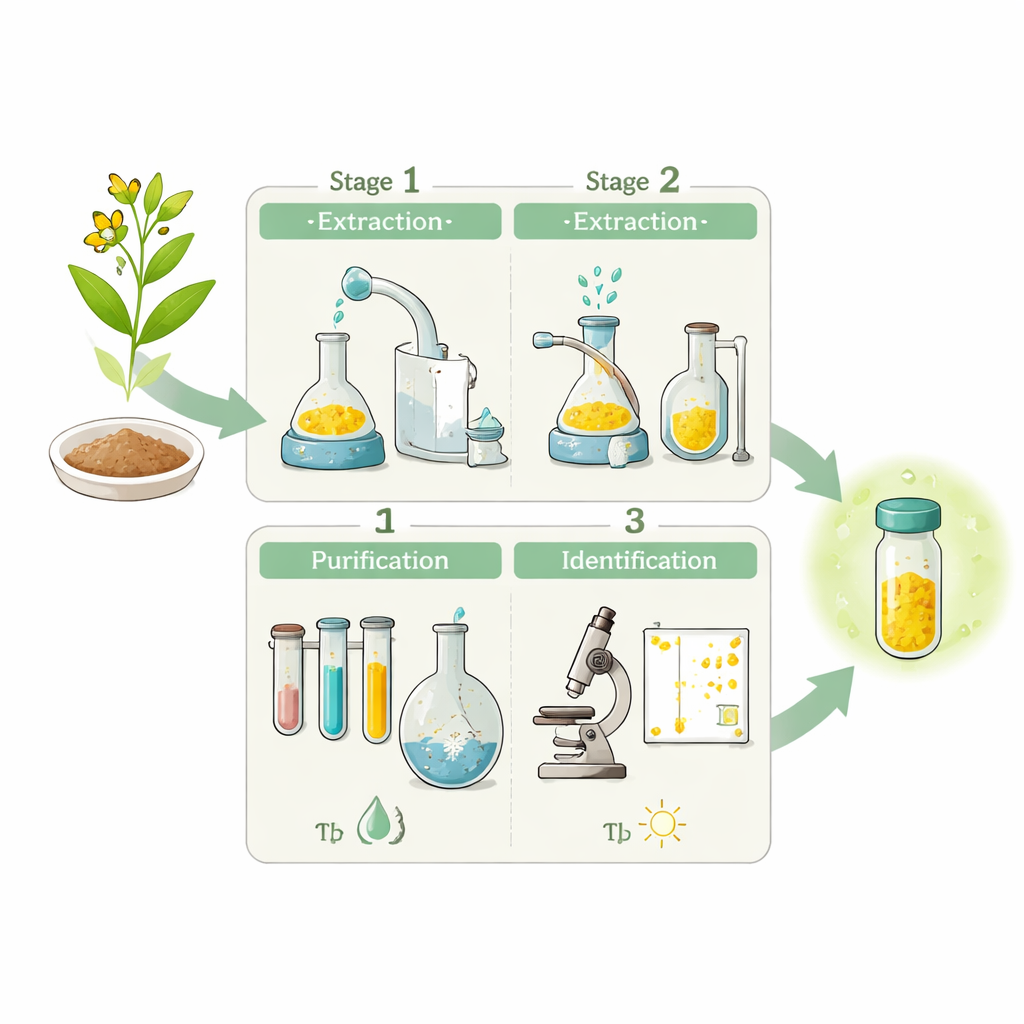
Turning a messy task into clear steps
Working with natural plant products can easily overwhelm beginners because the procedures involve many heating, cooling, and separation steps. The authors tackle this by breaking the experiment into three linked stages spread over several class sessions: extraction, purification, and identification. In the first stage, students boil powdered Coptis chinensis roots with a very dilute acid so that berberine moves from the plant material into water. They then adjust the acidity and salt content so that the compound separates out as a crude yellow solid. Each action—such as changing the pH or adding salt—is tied to a visible change, helping students connect what they do with what they see.
Polishing crude crystals into a pure product
In the second stage, students focus on turning the crude solid into cleaner, more uniform crystals. After allowing the mixture to sit for a week—so that crystals can slowly grow between classes—they filter, wash, and dry the solid, then dissolve it again in hot water. Careful control of temperature and acidity encourages the compound to crystallize out in a more orderly way, and a final recrystallization step produces high‑quality yellow crystals. This multi-step path is deliberately designed so that each student can measure how much material is gained or lost along the way, giving concrete practice with yield calculations and the trade‑off between quantity and purity.

Seeing and testing what was made
The third stage teaches students how to check whether they truly obtained the desired compound. Rather than rely on advanced machines, the course uses simple, widely available tools. Students perform chemical tests that cause striking color changes when berberine reacts with certain reagents, view the crystal shapes under a microscope, and run thin‑layer chromatography, in which small spots of the sample travel up a plate in a solvent and can be compared with a reference. Together, these tests create a web of evidence strong enough for teaching purposes and demonstrate how different methods support, but also limit, each other.
Building greener habits in the lab
A key innovation of this course is the built‑in attention to environmental impact. The authors introduce a framework called G‑RPWAM, which asks instructors to think systematically about reagents, procedures, waste, awareness, and methodology. In practice, this means using very dilute acid, replacing strong bases with safer lime, relying mostly on water instead of organic solvents, and letting crystals form at room temperature rather than forcing them with energy‑intensive cooling. The team tracks the amounts of chemicals, waste, and electricity used, showing sizable reductions in acid, salt, waste volume, and estimated carbon emissions compared with an older version of the experiment—all without sacrificing the amount or apparent purity of berberine produced.
What students gained from the redesign
To find out whether the new structure actually helped learning, the authors collected both performance data and student opinions from three class groups. Yields and purification results were consistent, suggesting the protocol is robust, and students reported that the clear staging and green focus made the lab easier to follow and more meaningful. Instructor ratings showed strong development of data analysis, problem‑solving, and creative thinking, though hands‑on technical skill still varied and may need extra practice or pre‑lab support. The authors stress that this is an intentionally “step‑by‑step” starter experience: by stabilizing the routine details now, students are better prepared later to design their own approaches in more open‑ended projects.
Big picture takeaway
This study demonstrates that undergraduate labs do not have to choose between solid skill training, clear teaching, and environmental responsibility. By thoughtfully dividing a complex natural‑product experiment into stages and weaving in green chemistry choices at every step, the authors created a laboratory module that is safer, more sustainable, and widely accessible, yet still rich in scientific reasoning. Their approach offers a practical template for other courses that want to teach real‑world chemistry while minimizing waste and confusion in the teaching lab.
Citation: Liu, Y., Huang, Q., Zhang, Z. et al. A structured multi-day experimental framework integrating green chemistry for the extraction and characterization of Berberine hydrochloride in undergraduate education. Sci Rep 16, 8092 (2026). https://doi.org/10.1038/s41598-026-39150-x
Keywords: green chemistry education, undergraduate laboratory, natural product extraction, berberine crystals, sustainable teaching labs