Clear Sky Science · en
Hydrothermal synthesis of ZnO nanoparticles from recycled ZnO obtained from electric Arc furnace dust: morphology control and applications
Turning Steel Dust into Useful Powder
Every year, steel factories generate tons of fine dust loaded with valuable metals that usually end up as hazardous waste. This study explores how to turn that dust into something far more useful: tiny particles of zinc oxide that can help in electronics, water purification, agriculture, and even fighting harmful bacteria. By carefully redesigning how the dust is treated, the researchers show that yesterday’s waste can become tomorrow’s high‑tech ingredient.
From Smokestack Dust to Clean Zinc
In electric arc furnace steelmaking, scrap metal is melted using powerful electrical arcs. This efficient process produces a fine dust that is captured in filters to protect the environment. The dust contains zinc in relatively high amounts, mixed with other metals such as iron, lead, sodium, and potassium. Instead of mining new zinc ore, the team started with zinc oxide already recovered from this dust and then used acid to selectively dissolve the zinc while leaving most of the lead behind. By choosing sulfuric acid at the right strength and solid‑to‑liquid ratio, they recovered over 90% of the zinc at room temperature, producing a clean, zinc‑rich solution that serves as the starting point for making new materials.
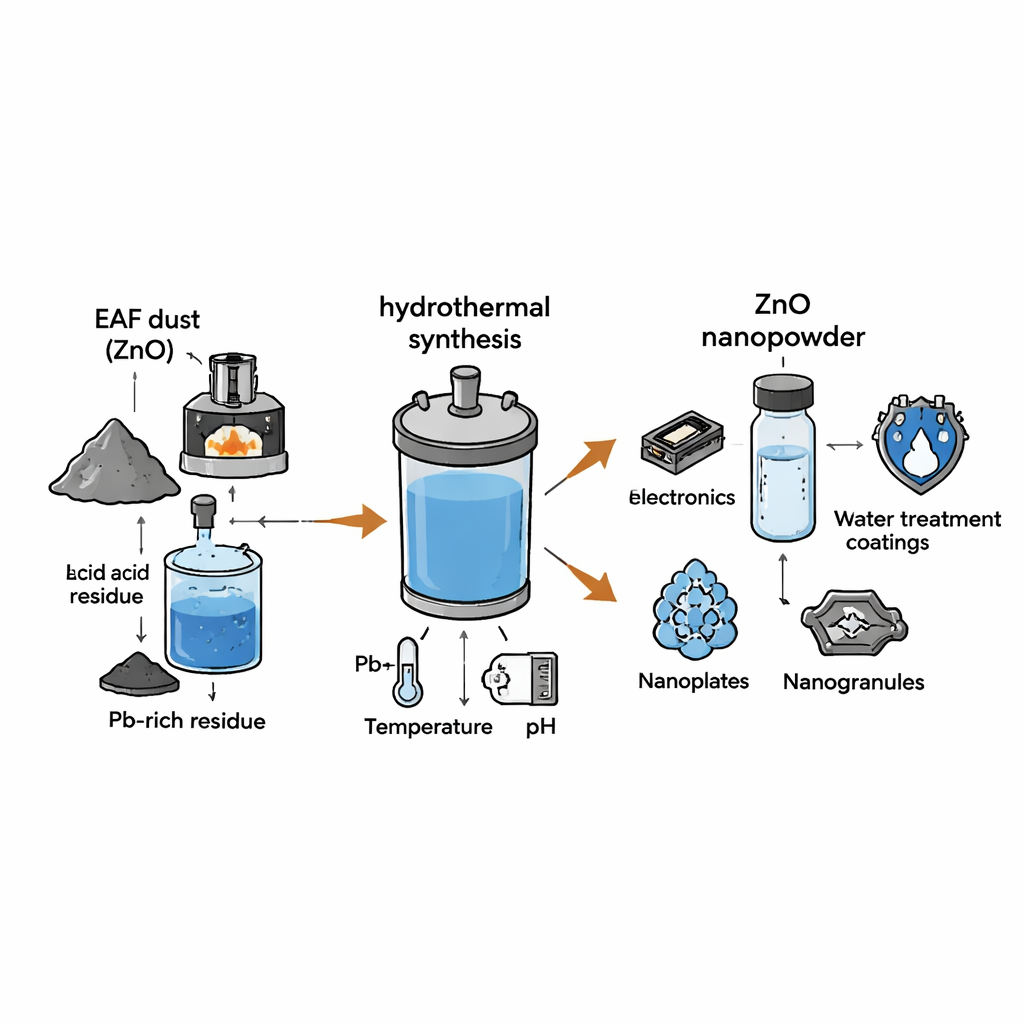
Cooking Nanoparticles under Pressure
To transform this purified solution into zinc oxide nanoparticles, the researchers used a method called hydrothermal treatment. In simple terms, they sealed the liquid in a small, pressure‑resistant vessel and heated it between 100 and 200 degrees Celsius while adjusting how basic (or alkaline) the mixture was. Under these hot, pressurized conditions, dissolved zinc combined with hydroxide ions to first form zinc hydroxide and then rearranged into crystalline zinc oxide. By varying the pH, reaction time, temperature, and the strength of the sodium hydroxide solution, they were able to “tune” how the particles grew—much like adjusting recipe settings in a pressure cooker to change texture.
Shaping the Tiny Building Blocks
The real breakthrough was controlling the shape and size of the zinc oxide particles despite starting from a complex, recycled source. At lower pH values, the particles formed poorly defined, impure structures. When the precursor solution was made strongly alkaline (around pH 11–12), the particles became highly crystalline and took on a uniform rod‑like form. Raising the synthesis temperature thinned these nanorods, while changing reaction time first sharpened their shape and then encouraged them to clump and flatten. Most dramatically, altering the concentration of sodium hydroxide at constant pH shifted the particles from large hexagonal blocks to neat nanorods, then to tiny granules, and finally to thin, plate‑like sheets. Standard lab tools such as X‑ray diffraction and electron microscopes confirmed that all of these shapes shared the same zinc oxide structure but differed in size and surface area.
Light and Germs: What Shape Can Do
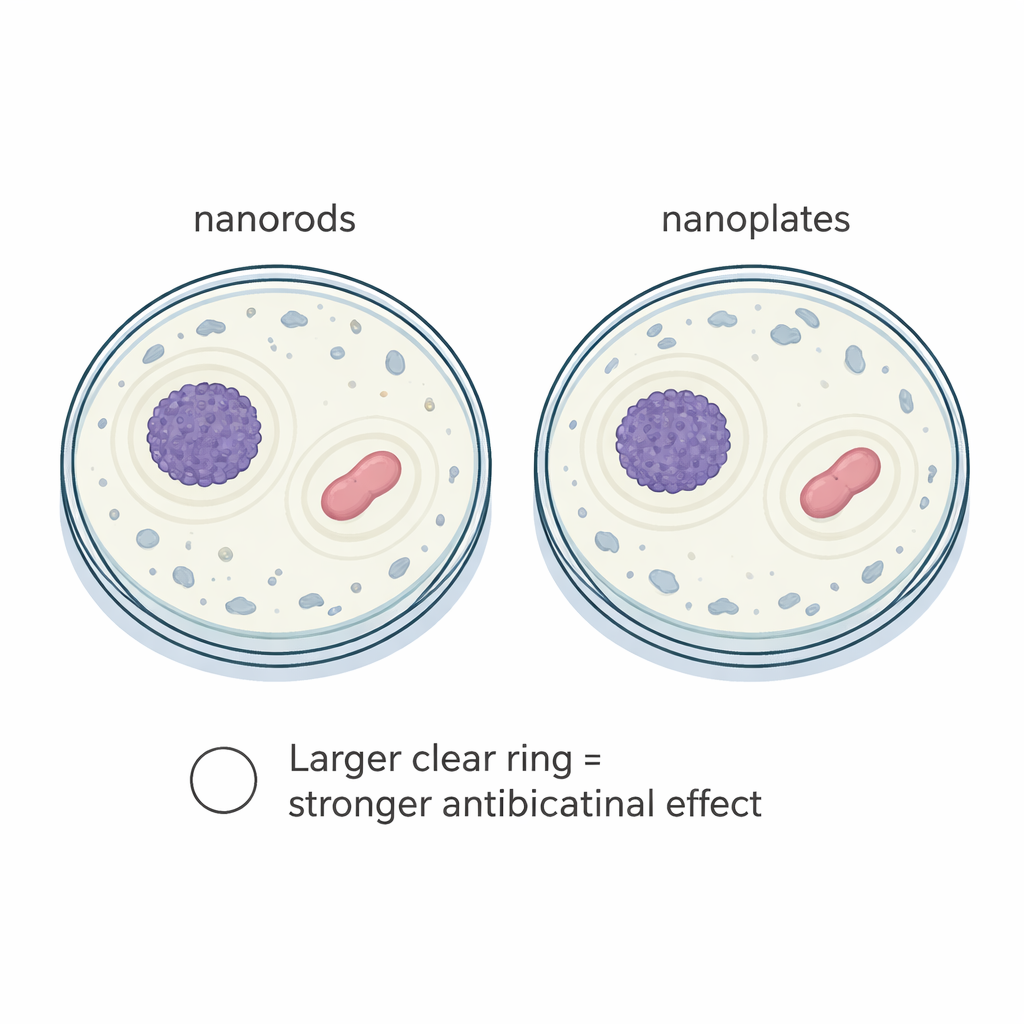
These different shapes are not just cosmetic. When the team shone ultraviolet and visible light on the materials, all of them strongly absorbed ultraviolet rays up to about 372 nanometers, with an energy gap of roughly 3.34 electron volts—ideal for UV‑blocking uses such as sunscreens, coatings, and sensors. The thinnest particles showed a slight shift in this absorption, consistent with quantum‑size effects seen when materials become very small. The researchers also tested how well the nanorods and nanoplates could slow the growth of two common bacteria, Staphylococcus aureus and Escherichia coli, by placing suspensions of the particles in wells on bacterial plates and measuring the clear “kill zones” around them. The nanoplate form consistently produced wider clear rings than the nanorods, especially against the Gram‑positive S. aureus, suggesting that its higher surface area and exposed crystal faces generate more reactive oxygen species that damage bacterial cells.
Waste as a Resource for Future Technologies
For a non‑specialist, the takeaway is straightforward: this work shows that industrial steel dust, usually treated as a disposal problem, can be transformed into carefully engineered, high‑performance zinc oxide nanoparticles. By fine‑tuning a two‑step process—gentle acid leaching followed by controlled high‑pressure heating—the researchers can dial in particle shapes that not only block ultraviolet light but also act as promising antibacterial agents. This approach supports a circular economy, where waste streams feed advanced technologies rather than landfills, and hints at a future in which cleaner factories and smarter materials development go hand in hand.
Citation: Somla, S., Yingnakorn, T., Chandakhiaw, T. et al. Hydrothermal synthesis of ZnO nanoparticles from recycled ZnO obtained from electric Arc furnace dust: morphology control and applications. Sci Rep 16, 7634 (2026). https://doi.org/10.1038/s41598-026-39138-7
Keywords: zinc oxide nanoparticles, industrial waste recycling, hydrothermal synthesis, ultraviolet protection, antibacterial materials