Clear Sky Science · en
Elucidating the mechanism of cefpodoxime-BSA interaction via a combination of multi-spectroscopic methods and molecular docking simulations
Why this antibiotic–blood interaction matters
When we swallow an antibiotic pill, we usually think only about killing germs. But before a drug can reach an infection, it must hitch a ride through the bloodstream. There it meets albumin, a major blood protein that ferries many medicines around the body. This study takes a close look at how the oral antibiotic cefpodoxime latches onto albumin, offering clues that help explain how long the drug stays in the body, how widely it spreads into tissues, and how safely it can be used in patients with different health conditions.
Meeting point in the bloodstream
Cefpodoxime is a third‑generation cephalosporin antibiotic used to treat common infections of the lungs, ears, throat, urinary tract, and skin. After being absorbed from the gut, the active drug enters the bloodstream, where only a small portion (about one fifth) usually attaches to albumin. The rest remains free to leave the blood and attack bacteria in tissues. Because this balance between bound and free drug strongly shapes both effectiveness and side effects, the researchers set out to pinpoint exactly how strongly cefpodoxime binds to albumin, where on the protein it settles, and what that partnership looks like at the molecular level.
Shining light on a hidden handshake
To watch the interaction without disturbing it, the team used a suite of light‑based methods. Standard ultraviolet absorption measurements showed that cefpodoxime and bovine serum albumin (a widely used stand‑in for human albumin) form a true complex rather than just overlapping signals. Fluorescence measurements, which track the glow of certain amino acids within the protein, revealed that this glow steadily dims as more drug is added. By analyzing how this dimming changes with concentration and temperature, the authors concluded that cefpodoxime and albumin form a stable pair in the ground state, rather than briefly bumping into each other in passing.
How tight and where the grip is
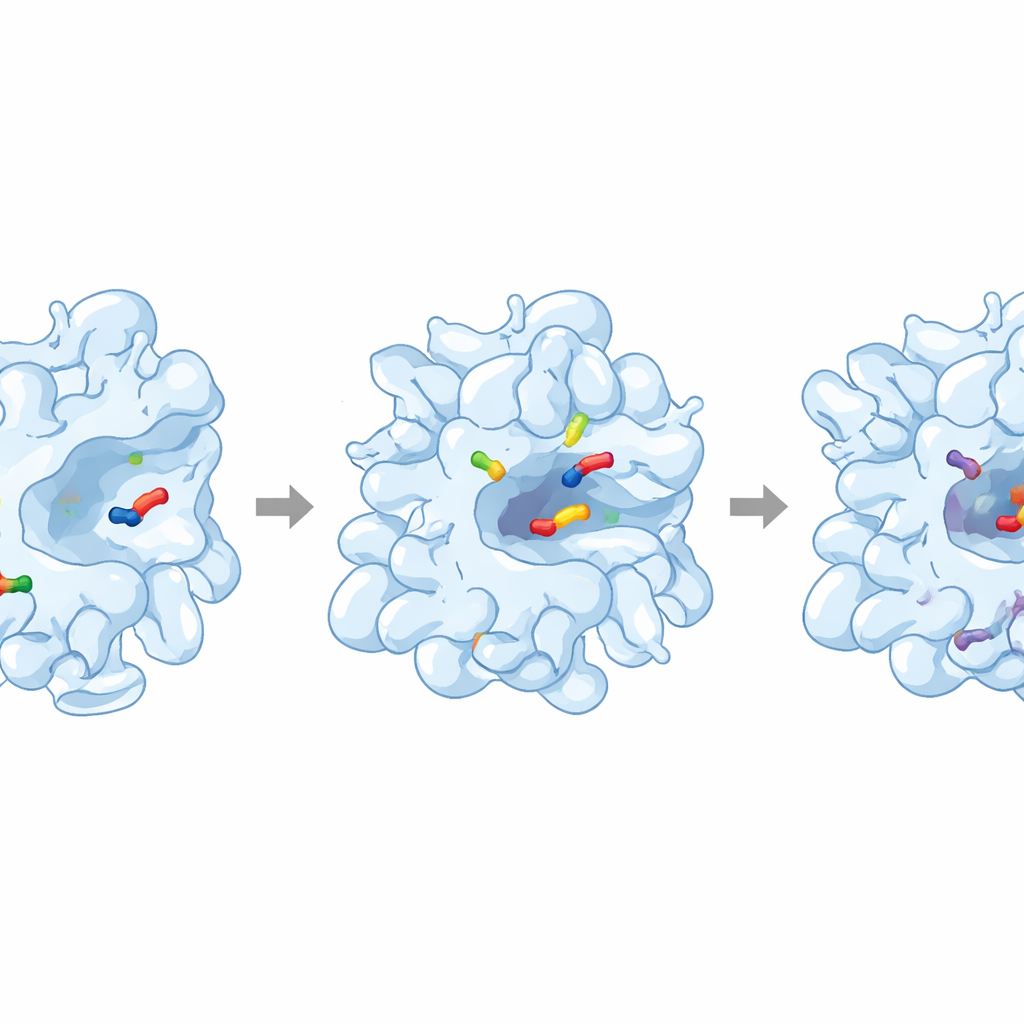
The data showed that cefpodoxime binds albumin with moderate strength: strong enough to form a clear complex, but weak enough that much of the drug remains free. Detailed analysis pointed to roughly one main binding site per albumin molecule. By comparing the behavior of cefpodoxime with that of two “marker” drugs known to claim specific pockets on albumin, the researchers found that cefpodoxime competes at what is called Site I, a deep cavity in one region of the protein. Computer docking simulations, which virtually fit the drug into a three‑dimensional model of albumin, backed this up and revealed that the drug nestles into a hydrophobic pocket, held in place by snug, water‑repelling contacts and a few supporting hydrogen bonds.
Subtle shape shifts and energy clues
Additional spectroscopic tools probed how this binding affects albumin’s architecture. Synchronous fluorescence scans, which are sensitive to the local environment around particular amino acids, hinted that one type of residue is pushed into a slightly more water‑repelling setting when the drug binds. Infrared measurements, which read out the vibrations of the protein backbone, showed small but clear shifts in bands associated with secondary structure, signaling a modest rearrangement of the protein’s fold. Thermodynamic calculations revealed that the process is spontaneous and takes in heat overall, with a large positive entropy change—an energetic fingerprint that points to hydrophobic interactions as the main driving force of binding.
What this means for treatment
Taken together, the experiments and simulations paint a coherent picture: cefpodoxime forms a one‑to‑one, moderately strong, hydrophobic partnership with albumin at a specific pocket, nudging the protein’s shape but not locking it rigidly. This behavior fits well with the drug’s known low binding in human plasma, its fairly short half‑life of around two hours, and its efficient clearance by the kidneys. In practical terms, the work helps explain why cefpodoxime can spread widely into tissues and why kidney function so strongly influences its levels. More broadly, the combined experimental–computational approach showcased here offers a blueprint for understanding how other antibiotics and drug candidates ride on albumin, knowledge that can guide dosing, predict interactions, and support the design of safer, more effective therapies.
Citation: El Gammal, R.N., Elmansi, H., El-Emam, A.A. et al. Elucidating the mechanism of cefpodoxime-BSA interaction via a combination of multi-spectroscopic methods and molecular docking simulations. Sci Rep 16, 7836 (2026). https://doi.org/10.1038/s41598-026-39137-8
Keywords: cefpodoxime, serum albumin, drug binding, pharmacokinetics, antibiotic transport