Clear Sky Science · en
Increased atherosclerosis and expression of inflammarafts in macrophage foam cells in AIBP-deficient mice.
Why clogged arteries are more than just extra fat
Atherosclerosis—the slow clogging and stiffening of our arteries—is a major cause of heart attacks and strokes. We often picture it simply as fat building up in blood vessels, but the story is more complicated: immune cells inside the vessel wall can either calm things down or fan the flames of inflammation. This study explores why, in some circumstances, these cells flip from fat-storing "cleanup crews" into dangerous instigators that help push plaques toward a more lethal stage.
Immune cells that hoard fat
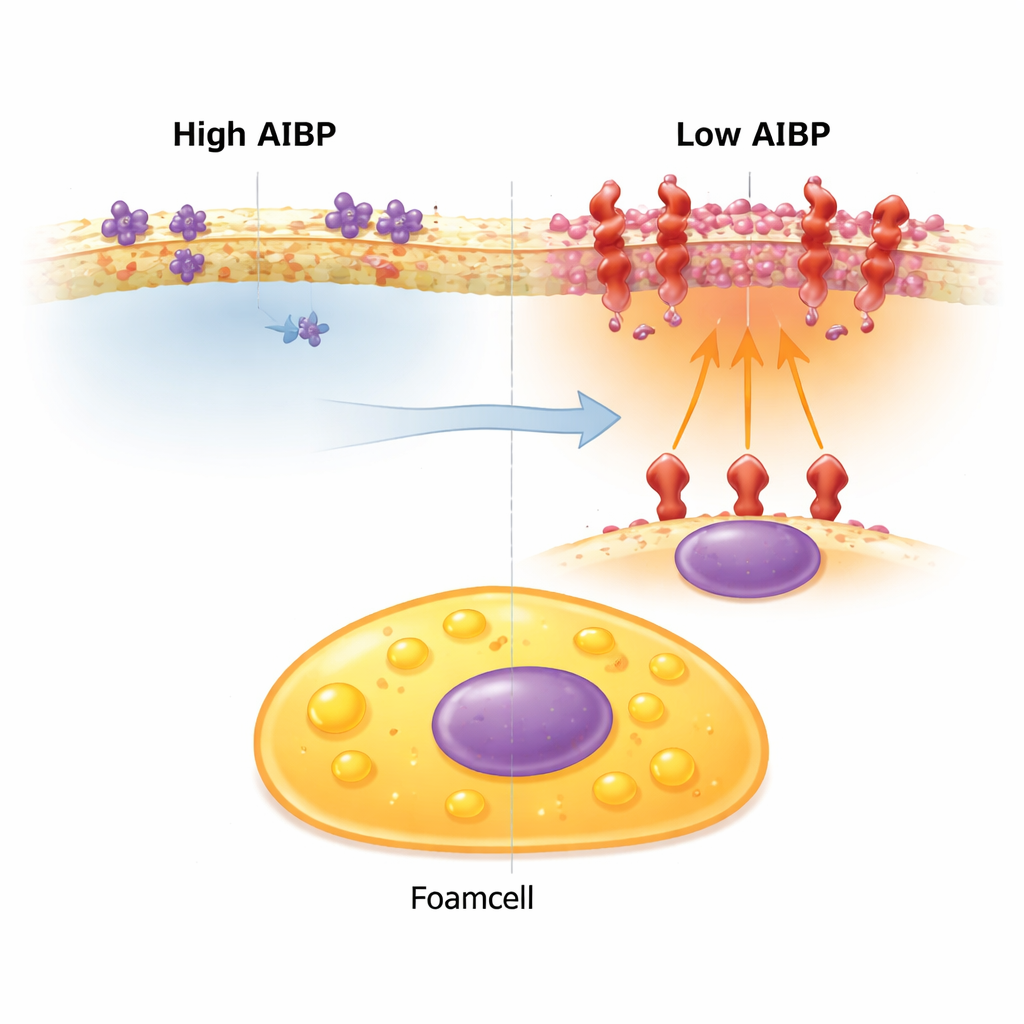
Inside developing artery plaques, a type of white blood cell called a macrophage takes up large amounts of cholesterol and turns into a so‑called foam cell—a bloated cell packed with oily droplets. Earlier work in mice suggested that these foam cells, despite their unhealthy appearance, are surprisingly quiet on the inflammation front, while leaner macrophages nearby tend to produce more inflammatory signals. The new research asks what happens if the body lacks a protein called AIBP, which normally helps remove cholesterol from cell surfaces and keeps certain membrane “hot spots” in check.
Membrane hot spots that amplify alarm signals
The cell surface is not uniform. Cholesterol and certain fats cluster into tiny platforms where receptors that sense danger can huddle together. When these platforms enlarge and pack in inflammatory receptors, the authors call them “inflammarafts.” One such receptor, TLR4, helps cells detect threats but can drive chronic inflammation if overactivated. In normal mice prone to high cholesterol, non‑foamy macrophages usually carry more of these inflammarafts, while foam cells remain relatively low in them—one reason foam cells have been viewed as less inflammatory.
When fat‑laden cells turn fiery
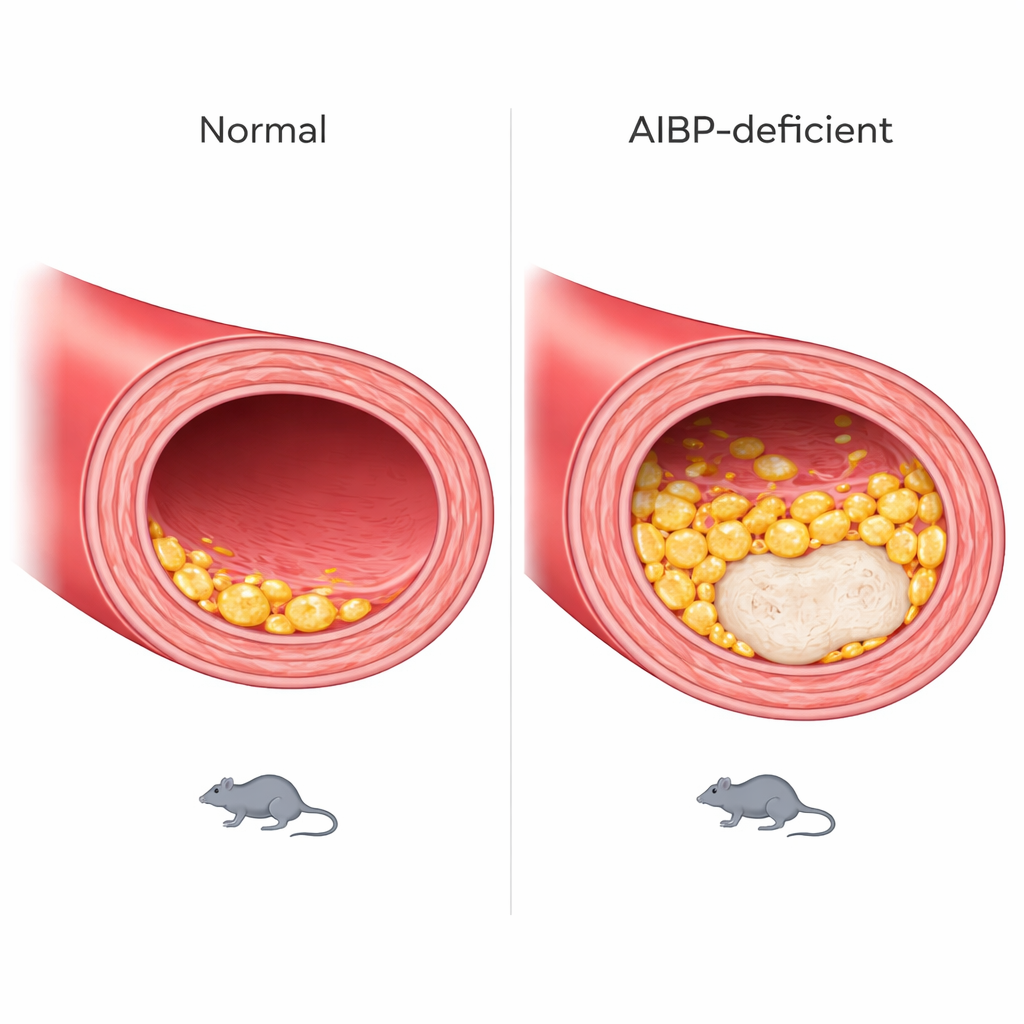
The team compared mice that lacked both AIBP and a key cholesterol‑clearing receptor (making them highly prone to artery disease) with control mice missing only the receptor. After 16 weeks on a high‑fat diet, the AIBP‑deficient mice had plaques containing more foam cells, each stuffed with more neutral fat. Crucially, these foam cells now showed high levels of TLR4 pairs and cholesterol‑rich membrane patches—the hallmarks of inflammarafts—making them the main inflammatory cell type in the plaque rather than the calmer, storage‑oriented cells seen before. In lab dishes, macrophages from AIBP‑deficient mice that were turned into foam cells with oxidized cholesterol switched on genes linked to inflammation, tissue breakdown, and vascular remodeling, and showed more oxidative stress and stressed mitochondria.
Growing, unstable plaques in the vessel wall
These cellular changes were mirrored by more severe disease in the arteries. Cross‑sections of the heart’s main outlet vessel revealed that AIBP‑deficient mice developed larger plaques and, importantly, larger necrotic cores—dead zones within the plaque associated with rupture‑prone, “vulnerable” lesions. Both male and female mice were affected, though the pattern of where along the vessel the worst damage appeared differed between sexes. The animals also gained weight more quickly and had higher blood triglyceride levels, pointing to broader disturbances in fat handling.
What this could mean for future treatments
To a non‑specialist, the key message is that not all cholesterol‑loaded cells in a plaque are the same. When AIBP is present, foam cells tend to act more like storage units, quietly holding excess fat. When AIBP is missing, those same cells remodel their surfaces, build inflammarafts, and start sending loud inflammatory signals that help drive plaques toward dangerous, advanced stages. While the work was done in mice, it highlights AIBP and the control of these membrane hot spots as potential levers for future therapies aimed at keeping artery plaques from becoming unstable and life‑threatening.
Citation: Li, S., Nazarenkov, N., Alekseeva, E. et al. Increased atherosclerosis and expression of inflammarafts in macrophage foam cells in AIBP-deficient mice.. Sci Rep 16, 7645 (2026). https://doi.org/10.1038/s41598-026-39113-2
Keywords: atherosclerosis, macrophage foam cells, inflammation, cholesterol metabolism, AIBP