Clear Sky Science · en
Morphophysiological disorders and metabolic reprogramming in Physalis peruviana infected with the physalis rugose mosaic virus
Why a small fruit and its virus matter
Cape gooseberry, a bright orange berry wrapped in a papery lantern, is gaining popularity with growers and consumers around the world. But the plants behind this tasty fruit are increasingly threatened by a newly described virus called physalis rugose mosaic virus. This study asks a simple but crucial question: what exactly does this virus do inside the plant, from the first contact to the final harvest, and why does that matter for farmers and food production?

From invisible visitor to sick plant
The researchers grew Cape gooseberry plants in a greenhouse and gently rubbed some leaves with a liquid carrying the virus, while others received a harmless buffer as a control. Over six weeks they tracked how the virus spread and how the plants changed. Using a sensitive genetic test, they detected virus multiplication in the inoculated leaves just three days after infection. By two weeks, the virus had reached young leaves elsewhere on the plant, and its numbers kept rising through day 42. As the viral load climbed, symptoms followed: leaves developed light-and-dark patchy mosaics, yellowing, rough texture, and distortions, and the plants became shorter with more side branches, a sign that their normal growth patterns were being disrupted.
What happens inside a sick leaf
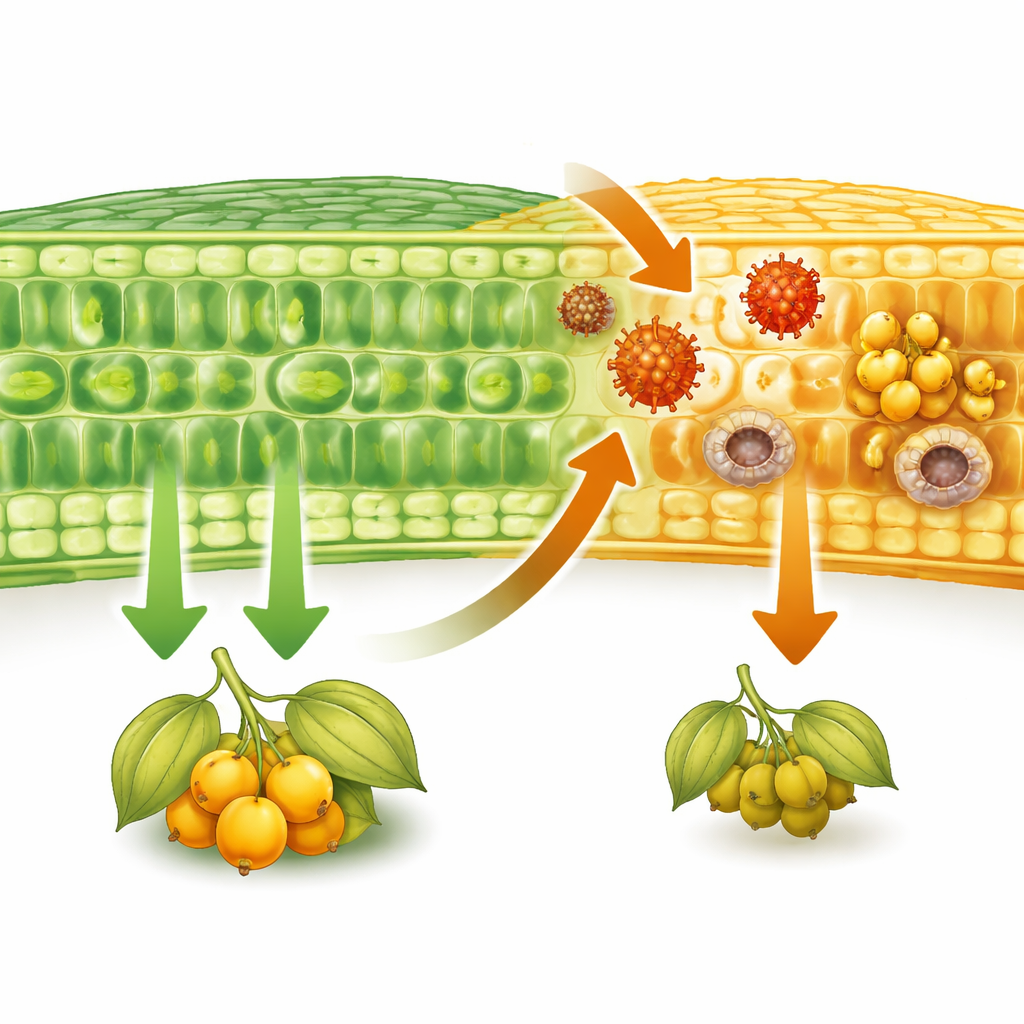
To see the damage up close, the team examined thin slices of leaves under the microscope. In the directly infected leaves, the outer skin cells and inner photosynthetic tissue were shrunken, collapsed, and disorganized, with enlarged nuclei that hint at intense activity hijacked by the virus. The plumbing tissue that normally moves sugars out of the leaf showed structural changes, suggesting that transport was blocked. When the same leaves were stained for starch, they were packed with dark granules, evidence that sugars produced by photosynthesis were getting stuck instead of being shipped to other parts of the plant. Even young, systemically infected leaves that looked less damaged showed abnormal cell proliferation and extra starch build-up, indicating that the virus was quietly reshaping their internal architecture.
The plant’s energy is retuned
The scientists also asked how the virus alters the plant’s chemistry over time. They used gas chromatography–mass spectrometry, a technique that can detect dozens of small molecules at once, to profile sugars, organic acids, and amino acids in local and systemic leaves at several time points. Early in infection, the profiles of healthy and infected leaves overlapped, suggesting the plant could still hold its basic metabolism steady. By day 42, however, systemically infected leaves showed a very different pattern. Levels of sucrose and pyruvate were higher, as were several key components of the tricarboxylic acid (TCA) cycle—the cell’s central energy hub, including citrate, malate, fumarate, and isocitrate. This pattern points to a ramped-up energy supply, likely fueling the heavy costs of viral replication and the plant’s attempt to respond.
Defense comes at a cost
Not all changes favored the virus. Some compounds that rose in infected leaves, such as glutamate, isoleucine, malonate, and shikimate, are tied to the production of signaling molecules and defensive substances. Network analysis of how these metabolites fluctuate together showed that healthy plants maintain a rich, flexible web of connections that supports growth. In contrast, infected plants at the late stage had simpler, tighter networks dominated by amino acids and related molecules linked to stress responses. In other words, the plant appears to divert resources away from growth and toward defense. That trade-off shows up plainly in the field-like measurements: infected plants had less chlorophyll, flowered later, and produced 31% fewer fruits by harvest, and the fruits were fewer and generally of lower commercial quality.
What this means for growers and crops
Seen from the outside, this virus simply makes Cape gooseberry plants look sick and yield less fruit. Inside, the story is more complex: the virus rewires the plant’s inner energy circuits, clogs up sugar transport, and forces the plant to choose defense over growth. By mapping these anatomical and metabolic shifts in detail, the study points to potential chemical markers of infection and to metabolic pathways that might be strengthened through breeding, biotechnology, or protective treatments. Ultimately, such insight could help farmers manage this virus more effectively and protect the promising future of Cape gooseberry and related crops.
Citation: Studnicka, M.H., Bianchini, J.R., Felisberto, N.B. et al. Morphophysiological disorders and metabolic reprogramming in Physalis peruviana infected with the physalis rugose mosaic virus. Sci Rep 16, 9015 (2026). https://doi.org/10.1038/s41598-026-39107-0
Keywords: Cape gooseberry, plant viruses, crop yield loss, plant metabolism, plant defense