Clear Sky Science · en
Dlgap2 deficiency disrupts synaptic homeostasis by promoting ubiquitin-mediated Itsn1 degradation in a valproic acid-induced autism-like model
Why a Common Medicine and Brain Wiring Matter
Valproic acid is a long‑used drug for epilepsy and mood disorders, but when taken during pregnancy it has been linked to a higher risk of autism in children. This study asks a pressing question for families and doctors alike: what exactly does this medication do to the developing brain’s wiring? By tracing changes down to specific molecules at synapses—the communication points between nerve cells—the researchers uncover a new chain of events that may help explain autism‑like behaviors and point toward future treatment strategies. 
Looking Across Species for Shared Trouble Spots
To find reliable clues, the team did not rely on just one animal model. They compared brains or brain‑like tissues from human cortical organoids, monkeys, rats, and mice that had been exposed to valproic acid before birth. Using large‑scale gene and protein surveys, they identified a small set of genes that changed in all these systems and that were tied to synapses and insulation of nerve fibers. Among them, one stood out: Dlgap2, a scaffolding protein that helps organize the postsynaptic side of excitatory synapses. In mice given prenatal valproic acid, Dlgap2 levels fell in the cortex and, more specifically, inside tiny synaptic compartments, suggesting that this structure‑keeping molecule is a key casualty of drug exposure.
From Molecular Shifts to Behavior Changes
The authors then asked whether loss of Dlgap2 alone is enough to disturb brain function. They used viruses carrying short hairpin RNA to reduce Dlgap2 in cultured mouse neurons and in newborn mice. In the dish, neurons with lower Dlgap2 grew shorter branches and had fewer postsynaptic puncta, signs of weakened connections. In living animals, targeted knockdown of Dlgap2 in the brain produced autism‑like behaviors: young male mice had trouble learning the location of a hidden platform in a water maze and showed reduced interest in social partners and social novelty in a three‑chamber test. These changes mimic the learning and social deficits seen in valproic‑acid‑exposed animals, strengthening the case that Dlgap2 loss is not just a side effect but a driver of altered behavior.
Finding a Vulnerable Partner at the Synapse
To understand how Dlgap2 loss ripples through the synapse, the researchers took a closer look at the proteins anchored in the postsynaptic density, the dense molecular pad that receives chemical messages. Using proteomics, they found that reducing Dlgap2 altered the abundance of hundreds of synaptic proteins, particularly those involved in organizing synapses and recycling vesicles by endocytosis. One protein, Intersectin‑1 (Itsn1), dropped especially sharply. Further biochemical work showed that Dlgap2 and Itsn1 physically reside in the same protein complexes in synapse‑enriched fractions and that both appear among autism‑linked genes. This pointed to a focused regulatory partnership between the two molecules within a crowded synaptic landscape. 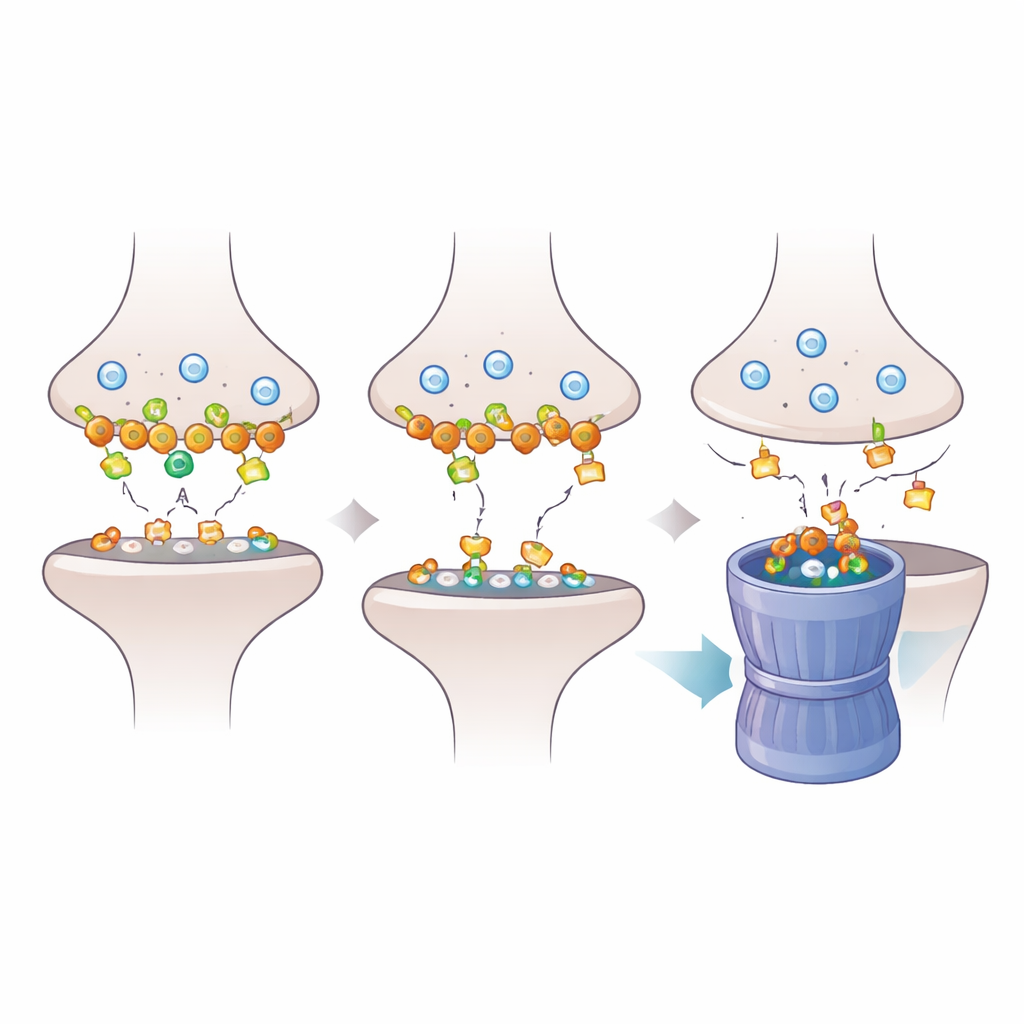
A Hidden Disposal Pathway Comes to Light
The team next asked why Itsn1 levels fall when Dlgap2 is missing. They observed that in synaptic compartments, Dlgap2 knockdown triggered a form of protein tagging known as K48‑linked ubiquitination, which marks proteins for breakdown by the cell’s proteasome “shredder.” Blocking this disposal system with the drug MG132 prevented the loss of Itsn1, indicating that Dlgap2 normally protects Itsn1 from being tagged and destroyed. Without Dlgap2, Itsn1 is increasingly tagged, fed into the proteasome, and depleted from synapses, undermining endocytic recycling and the balance of synaptic activity. The work suggests that subtle shifts in how quickly key proteins are turned over can destabilize synapses during brain development.
What This Means for Autism and Future Treatments
Taken together, the study proposes a simple but powerful idea for non‑specialists: prenatal exposure to valproic acid can weaken a structural “brace” protein (Dlgap2) at synapses. When that brace is lost, its partner protein (Itsn1) becomes over‑targeted for cellular garbage disposal, leading to faulty recycling of synaptic components and, ultimately, to miswired circuits and autism‑like behaviors in mice. While many genes and environmental factors contribute to autism, this Dlgap2–Itsn1 axis offers a concrete molecular chain from prenatal drug exposure to altered brain function. In the long run, strategies that preserve this partnership or fine‑tune the protein disposal machinery at synapses could help protect developing brains from similar forms of disruption.
Citation: Guo, X., Zhang, L. & Zhuang, K. Dlgap2 deficiency disrupts synaptic homeostasis by promoting ubiquitin-mediated Itsn1 degradation in a valproic acid-induced autism-like model. Sci Rep 16, 8305 (2026). https://doi.org/10.1038/s41598-026-39099-x
Keywords: valproic acid and pregnancy, synaptic scaffolding proteins, autism spectrum mechanisms, ubiquitin proteasome pathway, mouse autism models