Clear Sky Science · en
Distribution of artificial radionuclides in particle-size soil fractions
Why the dirt beneath old test sites still matters
Decades after nuclear test explosions ceased, the ground around former test sites can remain quietly contaminated. This study asks a practical question with real-world stakes: can we clean up such soil more efficiently by taking advantage of how radioactivity clings to different sizes of dirt grains? By carefully separating soil from the Semipalatinsk Nuclear Test Site in Kazakhstan into coarse and fine particles, the researchers explored where two key man‑made radionuclides — forms of cesium and americium — actually end up in the soil, and whether simple dry sieving could help reduce the volume of waste that must be handled as dangerous material. 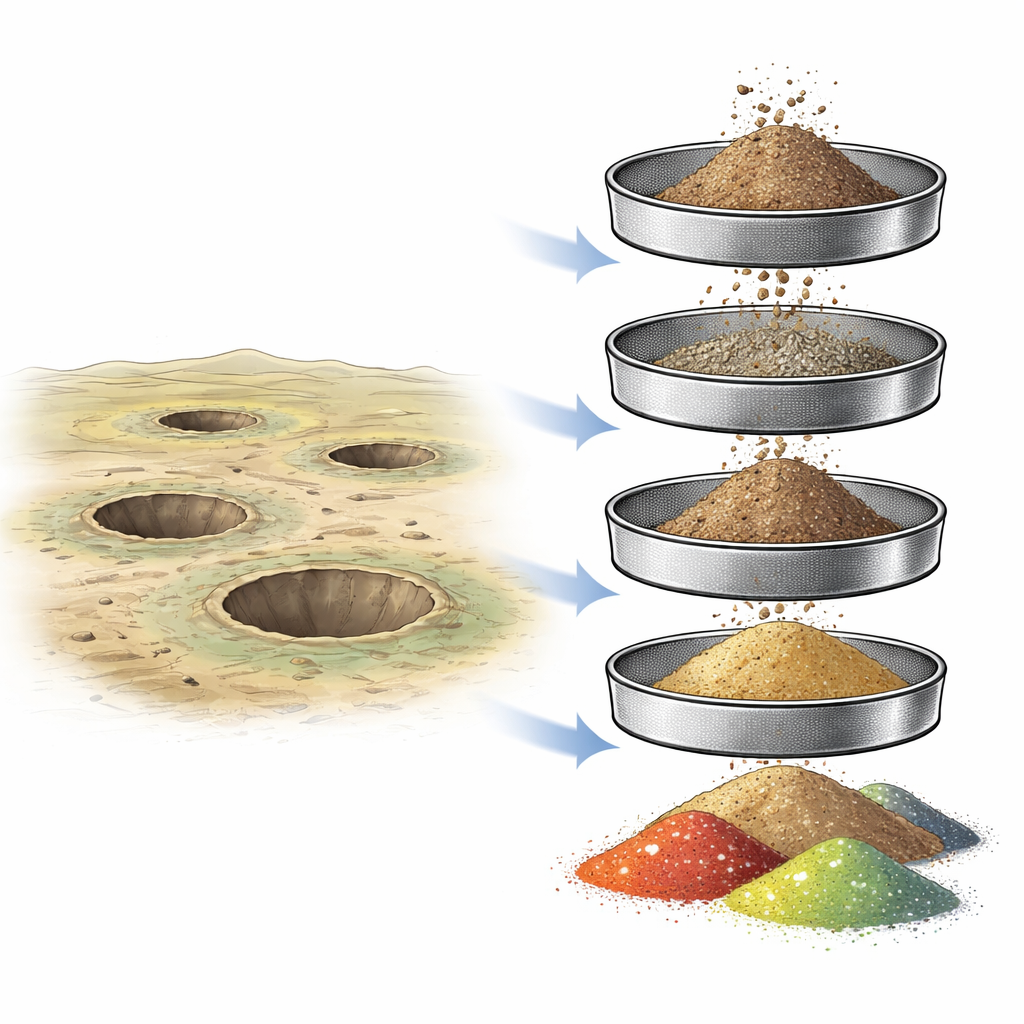
Old craters as natural laboratories
The team worked at four water‑filled craters created by different types of nuclear blasts: a ground explosion, a thermonuclear excavation blast, and two underground excavation tests. These craters are ringed by huge dumps of rock and soil thrown out by the explosions, and earlier surveys had shown that their soils contain high levels of radioactive cesium‑137 and americium‑241. Instead of treating all this soil as equally hazardous, the researchers wondered whether the contamination is unevenly distributed among particles of different sizes — from gravel‑like chunks to dust‑fine grains. If most of the radioactivity is locked into only part of the material, then targeted cleanup might be possible.
Sorting soil by grain size
Back in the laboratory, the scientists air‑dried the soil and passed it through a stack of sieves with decreasing mesh sizes. This produced several distinct fractions: particles larger than 10 millimeters, then 10–5 mm, 5–2 mm, 2–1 mm, 1–0.5 mm, and finally the finest fraction smaller than 0.5 mm (or smaller than 1 mm at one site). Each fraction was weighed to find out how much of the original soil it represented and then analyzed with sensitive gamma‑ray detectors to measure how much cesium‑137 and americium‑241 it contained. The key idea was simple: if the finest grains held much more radioactivity per kilogram than the bulk soil, removing them could sharply cut the hazard of what remained. 
Where the radioactivity hides
The results showed a clear pattern for cesium‑137. Across all four craters, its activity per kilogram consistently increased as particle size decreased, with the highest levels in the smallest grains. In many samples, the finest fraction carried far higher cesium activity than the whole soil, while coarser chunks held less. Americium‑241 behaved more variably. At the two excavation‑blast sites, it too tended to concentrate in the finer fractions, echoing cesium’s behavior. At the ground and thermonuclear excavation sites, however, americium’s distribution depended strongly on direction around the crater, sometimes favoring large particles and sometimes small ones. Overall, when the researchers calculated the absolute activity — taking into account both radioactivity per kilogram and the mass of each fraction — they found that particles smaller than 1 millimeter often dominated the total load of both radionuclides.
Measuring enrichment, not just concentration
To compare how strongly each fraction gained or lost radioactivity relative to the undivided topsoil, the authors used an enrichment factor: the ratio of a radionuclide’s activity in a given fraction to its activity in the bulk soil. An enrichment factor greater than one means the fraction is more contaminated than average; less than one means it is cleaner. Cesium‑137 showed steadily rising enrichment factors toward smaller particle sizes at all sites, confirming that the finest grains are preferentially enriched. Americium‑241’s enrichment was more site‑specific, yet at the excavation‑blast craters the smallest fractions again showed the most buildup. This metric turned out to be the most informative way to describe how artificial radionuclides are distributed across grain sizes.
What this means for cleaning up contaminated land
For a layperson, the takeaway is that radioactive cesium from nuclear blasts tends to cling to the tiniest bits of soil, while americium often does the same at certain types of test sites. Because those fine particles make up only a portion of the total mass, mechanically separating them through dry sieving could, in principle, strip away much of the radioactivity while leaving a larger volume of soil with lower contamination that might be easier and cheaper to manage. The study does not solve every cleanup challenge, especially where americium behaves unpredictably, but it shows that a relatively simple physical process can concentrate the most hazardous material into a smaller fraction, offering a promising tool for more efficient remediation of cesium‑contaminated soils at former nuclear test grounds.
Citation: Kunduzbayeva, A., Kabdyrakova, A., Mendubayev, A. et al. Distribution of artificial radionuclides in particle-size soil fractions. Sci Rep 16, 8068 (2026). https://doi.org/10.1038/s41598-026-39072-8
Keywords: radioactive soil, nuclear test site, cesium-137, americium-241, soil remediation