Clear Sky Science · en
Molecular characterization of virulence and antibiotic resistance genes in Klebsiella pneumoniae isolated from sputum samples at a tertiary hospital in Ethiopia
Why these lung infections matter
Pneumonia is a leading cause of serious illness and death worldwide, and hospitals are often on the front line of this battle. One of the most worrisome culprits is a bacterium called Klebsiella pneumoniae, which is learning to evade many of our best antibiotics while also becoming better at causing severe disease. This study from Ethiopia takes a close look at how these bacteria in one major hospital are changing at the genetic level, revealing why they are so hard to treat and why this matters far beyond a single country.
Looking for trouble in patients’ sputum
Researchers collected sputum samples—the thick mucus people cough up from deep in the lungs—from 182 patients suspected of having pneumonia at Tikur Anbessa Specialized Hospital, Ethiopia’s largest referral hospital. Using modern identification tools, they confirmed that 32 samples contained Klebsiella pneumoniae. They paid special attention to samples with a “currant jelly” appearance, a classic sign of severe Klebsiella lung infection, and indeed found the highest share of the bacteria in this group. The team then tested how these isolates responded to a broad panel of commonly used antibiotics.
Antibiotics losing their power
The results were alarming. Nearly all of the Klebsiella strains—94%—were resistant to at least three different families of antibiotics, qualifying them as multidrug resistant. Some were resistant to almost everything tested. Drugs that used to be reliable workhorses, such as tetracycline and several cephalosporins, failed against most isolates. Even carbapenems, often regarded as “last resort” antibiotics, showed high failure rates. While a few drugs like amikacin still worked for some patients, the overall picture was one of shrinking treatment options and growing risk of treatment failure.
Hidden tricks inside the bacteria
To understand how these microbes had become so formidable, the researchers examined their DNA for specific resistance and “virulence” genes—genetic instructions that help bacteria fend off drugs and cause more severe disease. Many isolates carried multiple genes that produce enzymes capable of breaking down powerful antibiotics, including carbapenems. One such gene, called blaNDM, was especially common. Other genes altered the bacteria’s outer shell to make it harder for drugs to get in, or powered tiny pumps that actively expelled antibiotics. At the same time, more than half of the isolates carried genes for sticky surface structures that help them cling to lung tissue and form protective biofilms, making infections harder to clear.
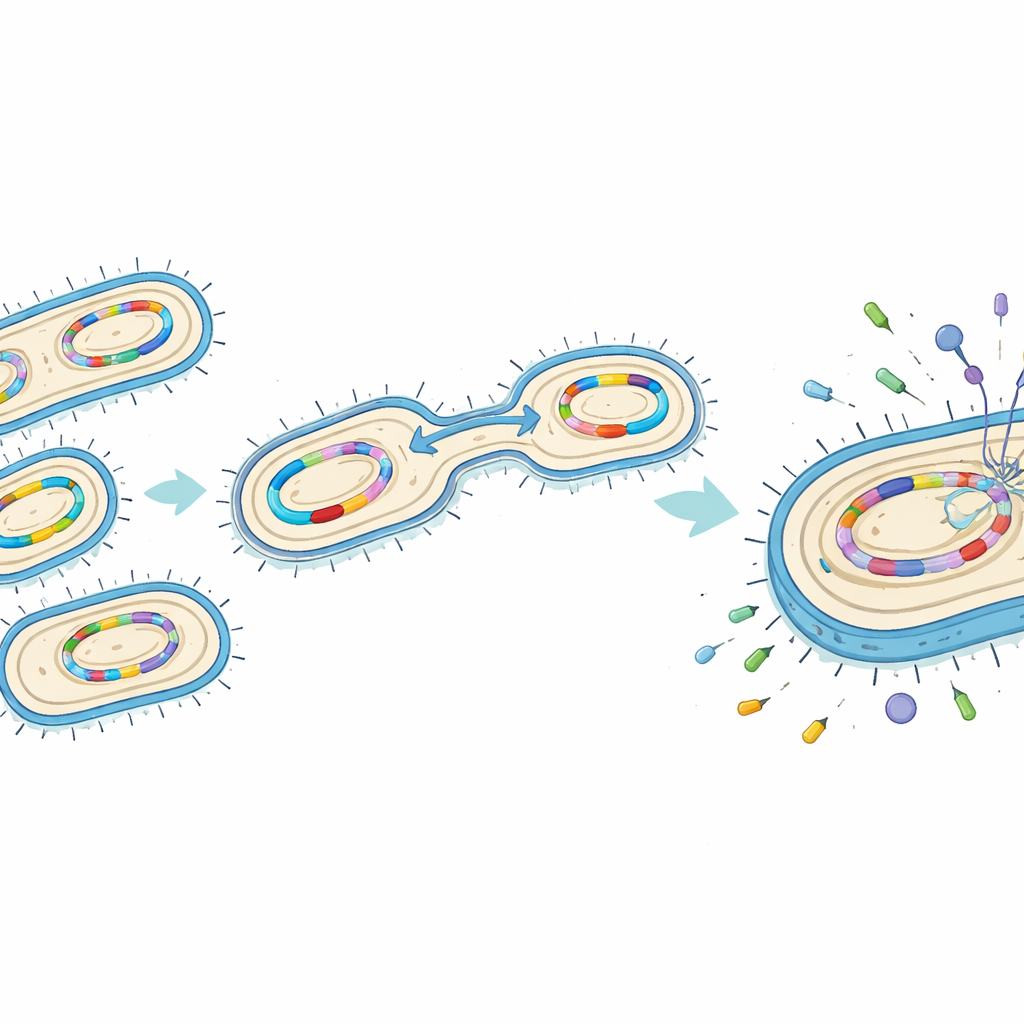
When stickiness turns into danger
A small but important subset of the isolates showed a “hypermucoviscous” behavior: their colonies formed thick, stretchy strings when touched, a sign of an especially heavy, protective capsule. These strains often carried a gene known as rmpA, which boosts capsule production and is linked to so‑called hypervirulent Klebsiella that can spread rapidly in the body and cause severe complications such as liver abscesses or bloodstream infections. In this hospital, such high‑risk strains were already present among pneumonia patients, and some combined these virulence traits with extensive drug resistance, creating a particularly dangerous mix.
What this means for patients and hospitals
For non‑specialists, the key message is that a single species of bacterium is evolving in ways that simultaneously blunt our strongest antibiotics and increase its ability to cause severe disease. In this Ethiopian hospital, Klebsiella pneumoniae is not only common in pneumonia patients but often armed with multiple resistance and virulence tools packed into its DNA. The authors conclude that without tighter infection control, careful antibiotic use, and ongoing genetic surveillance of hospital bacteria, these high‑risk strains will continue to spread and undermine routine treatments. Their detailed molecular snapshot provides crucial data for Ethiopia and adds to a growing global warning: if we do not act, once‑treatable lung infections could again become life‑threatening for many patients.
Citation: Abebe, A.A., Birhanu, A.G. & Tessema, T.S. Molecular characterization of virulence and antibiotic resistance genes in Klebsiella pneumoniae isolated from sputum samples at a tertiary hospital in Ethiopia. Sci Rep 16, 8541 (2026). https://doi.org/10.1038/s41598-026-39069-3
Keywords: Klebsiella pneumoniae, antibiotic resistance, pneumonia, hospital infections, Ethiopia