Clear Sky Science · en
Zebrafish casr modulates cardiac structure and function
Why Fish Hearts Matter to Human Health
Our hearts rely on calcium, a simple mineral, to beat properly. When calcium signals go wrong, the result can be heart failure or other serious disease. This study uses tiny zebrafish—transparent freshwater fish often used in labs—to explore how a calcium-sensing switch in cells shapes heart strength and survival. By turning off a single gene that helps cells sense calcium, the researchers uncovered a chain of problems in heart pumping, body shape, and even the fish’s air-filled swim bladder. Their work also shows that an animal’s broader genetic background can dramatically soften or worsen the impact of a harmful mutation, a lesson with clear echoes in human medicine.
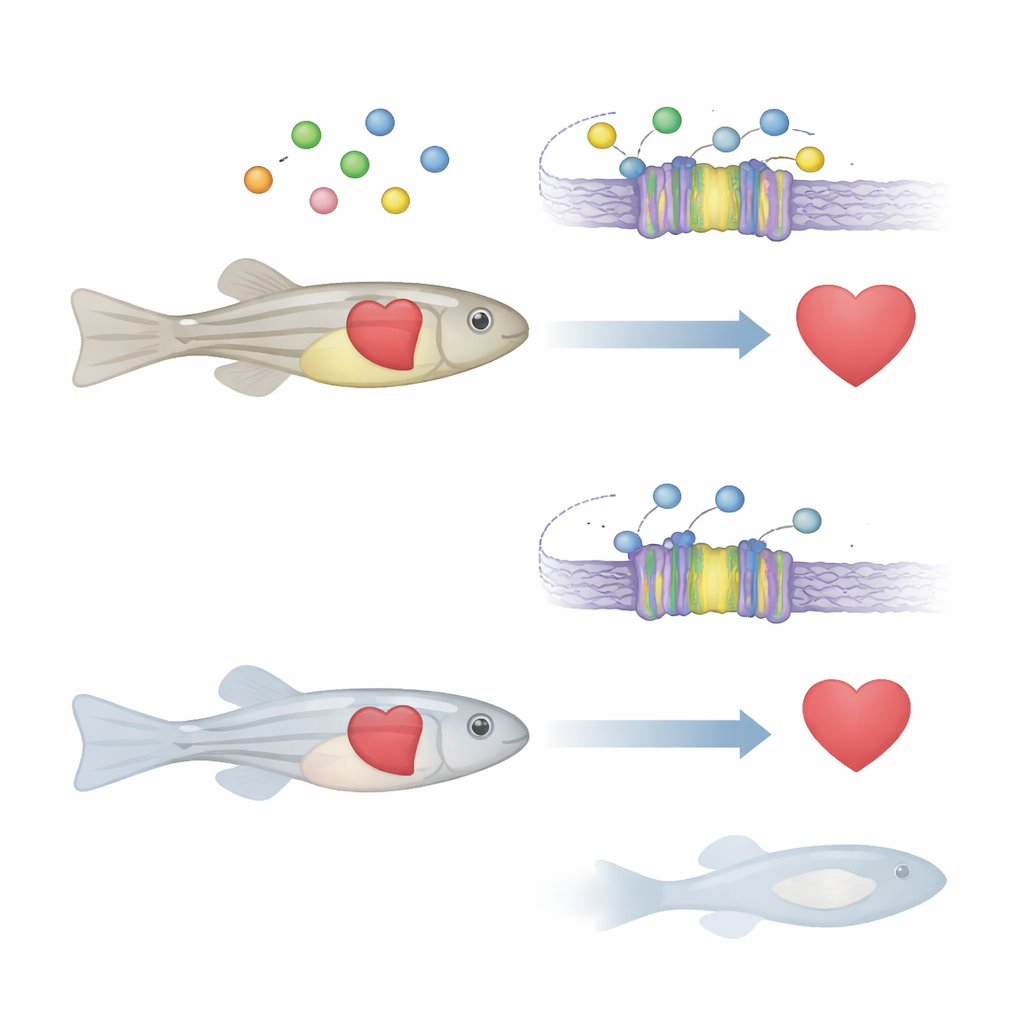
A Cellular "Thermostat" for Calcium
Calcium inside and outside cells acts like a universal signal that controls muscle movement, hormone release, and even which genes turn on. Many organs rely on a protein called the calcium-sensing receptor, which acts like a thermostat for calcium levels. When calcium outside the cell changes, this receptor adjusts hormone release and other processes to restore balance. In people, defects in this receptor are already linked to bone and mineral disorders, and are suspected players in heart disease and some cancers. However, exactly how this calcium sensor influences heart development and function in a whole animal has remained unclear.
What Happens When the Calcium Sensor Is Switched Off
The team focused on the zebrafish version of this gene, called casr, which is strongly conserved with its human counterpart. Using modern gene-editing tools, they created zebrafish in which both copies of casr were disabled. At first, the young fish looked nearly normal. But by five days after fertilization, many mutants failed to inflate their swim bladder, a small gas-filled organ that controls buoyancy. These fish sank to the bottom, swam poorly, soon developed spine curvature, and all died by two weeks of age. These outward signs pointed to serious internal problems affecting more than one organ system.
A Weaker, Smaller, and Slower Heart
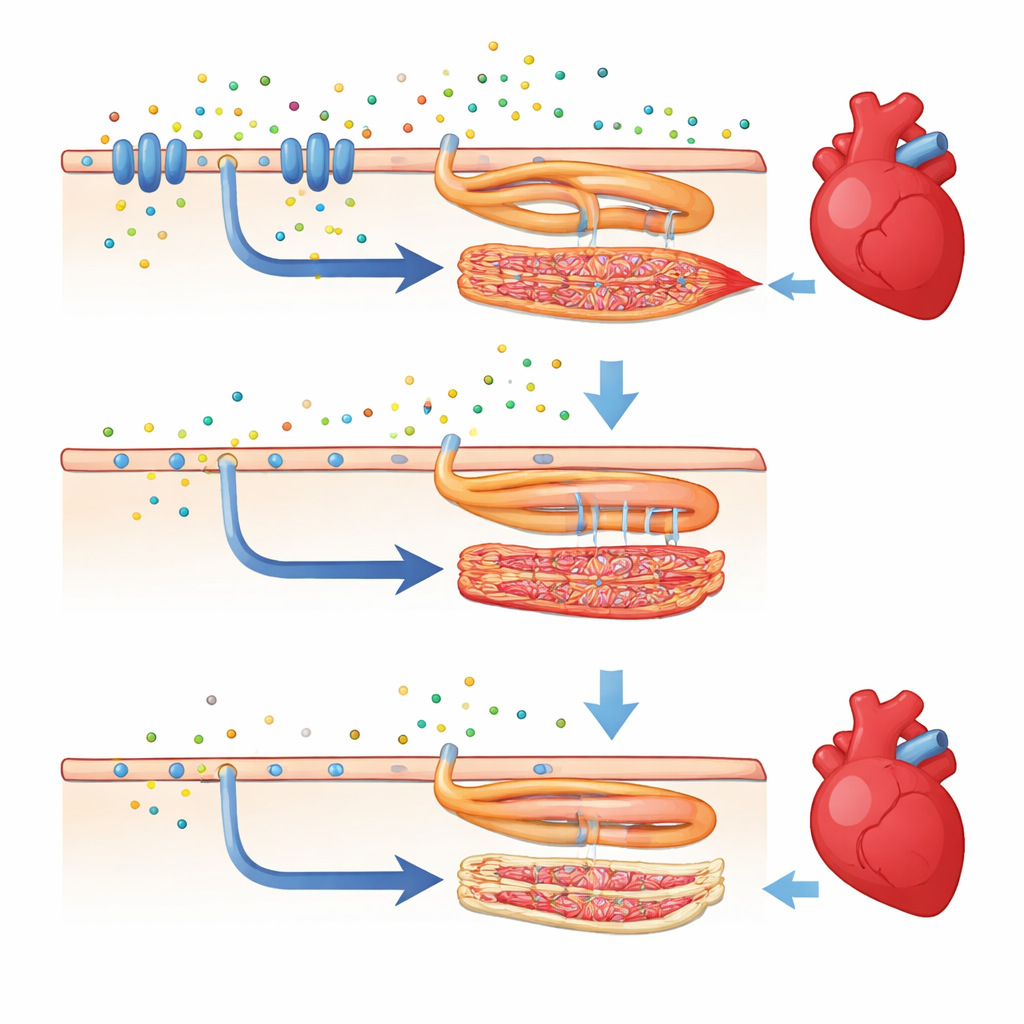
High-speed video and image analysis of beating hearts revealed how profoundly the missing calcium sensor affected cardiac performance. In normal zebrafish larvae, the heart ventricle fills and empties in a strong rhythmic pattern. In the mutants, the ventricle was noticeably smaller during filling, the heart beat more slowly, and each beat pushed out less blood. Measures of how much the heart muscle shortens and how much its pumping chamber area changes with each beat were both reduced, showing weakened contraction. Overall cardiac output—the volume of blood moved per minute—dropped sharply. At the gene level, hundreds of transcripts changed, and key networks controlling calcium handling and the machinery of muscle contraction were dialed down, linking the gene edit to both molecular and mechanical heart failure.
How Background Genes Can Rescue a Sick Heart
Surprisingly, the same casr mutation behaved very differently in another common zebrafish strain, known as TL (Tupfel long-fin). When the gene knockout was moved into this new genetic background, the fish were able to inflate their swim bladders, survive to adulthood, and showed much better heart pumping. Their hearts were still somewhat compromised—heart rate remained lower than normal—but measures of how forcefully the ventricle contracted and the amount of blood pumped per minute nearly matched healthy fish. In these rescued animals, many of the genes tied to calcium signaling and heart muscle contraction rebounded to intermediate levels, higher than in the original sick line. This reveals that other genes scattered across the genome can buffer or intensify the impact of a harmful change.
Signals That Shape Hearts and Buoyancy
Beyond the heart, the loss of the calcium sensor also disturbed major growth-control systems in the embryo. Genes in the Wnt and Hedgehog signaling pathways—well-known architects of organ formation—were turned down in the mutants. These changes likely contribute to the failed inflation of the swim bladder, which in turn can worsen survival by making it harder for the fish to feed and move efficiently. Markers of heart muscle cell differentiation were also reduced, and the ventricles were smaller, hinting that the calcium sensor helps guide how heart muscle cells grow and mature.
What This Means for Understanding Disease Risk
Taken together, this work shows that the calcium-sensing receptor is a crucial guardian of both heart function and proper organ development in zebrafish. When this sensor is lost, calcium signals falter, heart muscle contracts weakly, the swim bladder fails, and the animals die young. Yet the dramatic rescue in a different genetic strain highlights a key message: the rest of the genome can strongly modify how dangerous a given mutation really is. For human health, this underscores why people carrying similar risky gene variants may experience very different disease outcomes, and why understanding both core disease genes and their genetic background is essential for predicting and treating heart conditions.
Citation: Liu, L., Hu, Y., Xie, B. et al. Zebrafish casr modulates cardiac structure and function. Sci Rep 16, 8543 (2026). https://doi.org/10.1038/s41598-026-39063-9
Keywords: calcium signaling, heart development, zebrafish model, genetic background, cardiac function