Clear Sky Science · en
Isotherm, kinetic, and thermodynamic insights into textile effluent remediation using acid-treated sugarcane peel
Turning Farm Waste into Clean Water
Colorful clothes and fabrics often hide a dirty secret: the wastewater from textile factories can be loaded with stubborn dyes, salts, and metals that are hard to remove and harmful to rivers and lakes. This study asks a simple question with big implications: can leftover sugarcane peels, a common agricultural waste, be gently treated and turned into an affordable, reusable filter that strips pollution from real textile wastewater? 
Why Textile Wastewater Is So Troublesome
Textile factories use large amounts of water to wash, dye, and finish fabrics. The resulting wastewater is a complex cocktail of bright dyes, fine particles, salts, and heavy metals. These substances can resist natural breakdown, block sunlight in rivers, lower oxygen levels for fish, and introduce toxic elements like chromium and lead into the food chain. Traditional treatment technologies—such as chemical coagulation or advanced oxidation—can work, but they are often expensive, energy-hungry, and generate new wastes like sludge that must also be handled safely. Many regions where textiles are produced simply cannot afford these systems at the scale required.
From Sugarcane Trash to Water-Cleaning Treasure
Sugarcane is grown worldwide, and its peels and bagasse are often discarded or burned. Yet these materials are rich in natural building blocks like cellulose and lignin, which carry many chemical “handles” that can latch onto pollutants. The researchers collected sugarcane peels from juice vendors, washed and dried them, and then compared two versions: the raw peel (UTSP) and peel soaked in a mild solution of phosphoric acid (ATSP). This acid treatment cleans away impurities, opens up more pores, and adds extra reactive sites on the surface without resorting to high-temperature, energy-intensive steps used to make commercial activated carbon. Microscopy images showed that the treated peels had a rougher, more porous texture, and chemical analysis confirmed an increase in active groups that can bind contaminants.
How Well the Natural Filter Performs
To test performance, the team used real textile wastewater collected from a factory, rather than a simple lab dye solution. Under optimized conditions—about two hours of contact time, a slightly acidic pH around 5–6, and a small dose of adsorbent—the acid-treated peels removed about 85% of the wastewater color, compared with 68% for the untreated peels. Measurements of chemical oxygen demand and biological oxygen demand, which reflect the total load of organic pollution, also dropped sharply, as did levels of salts like chlorides and sulfates and metals such as cobalt, nickel, cadmium, and chromium; lead fell below detectable limits. The maximum amount of pollutant that the treated peel could hold reached about 50 milligrams per gram, competitive with many engineered materials despite using a low-cost waste feedstock.
Peering Under the Hood of the Process
The researchers dug deeper to understand how this natural filter works. Tests that track how quickly pollution disappears from the water over time showed that the untreated peel behavior was closer to weak, physical sticking, while the treated peel followed a pattern associated with stronger, chemical-type bonding. Other experiments examined how much pollutant the peels could hold at different starting concentrations and temperatures. These analyses indicated that the treated material offers a varied landscape of high-energy sites where multiple layers of pollutants can accumulate, and that the uptake process is both spontaneous and helped by warmth—adsorption became stronger at higher temperatures. 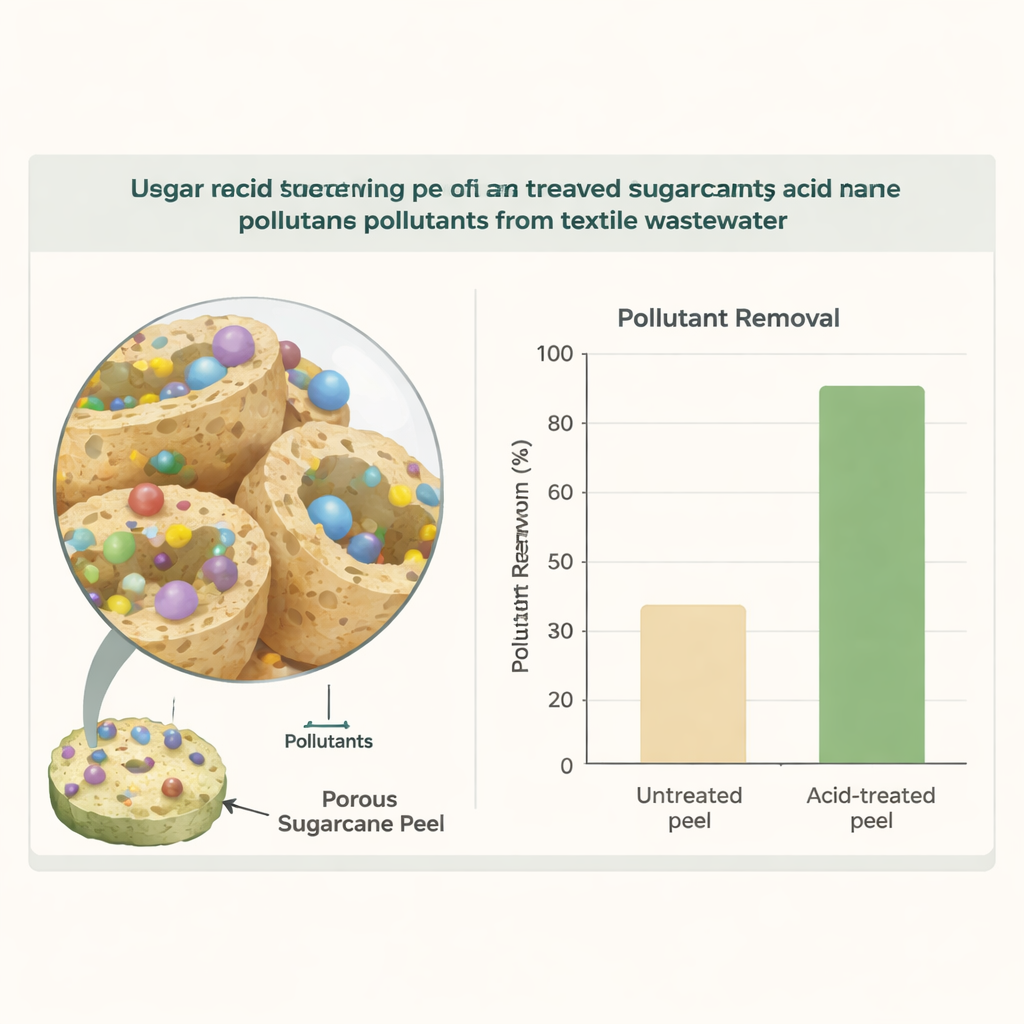
Reusability and Real-World Costs
For any practical technology, cost and durability matter as much as performance. The team repeatedly loaded the peels with pollutants and then washed them with a mild acid to release the captured compounds. After five cycles, the acid-treated peels still kept about 60% of their original efficiency. A simple cost analysis of a pilot-scale plant suggested that cleaning one cubic meter of textile wastewater with untreated peels would cost about 0.48 US dollars, and with acid-treated peels about 0.49 US dollars. In other words, the acid step adds only a tiny cost while delivering a large gain in cleaning power.
What This Means for Cleaner Rivers
In plain terms, this work shows that something as ordinary as discarded sugarcane peels can be upgraded, with a modest chemical treatment, into a robust filter for messy, real-world textile wastewater. The treated peel captures dyes, organic matter, and several heavy metals, can be reused multiple times, and remains inexpensive enough to be attractive in regions where both agricultural waste and textile production are plentiful but funds for high-tech treatment are scarce. While more work is needed to move from batch tests to continuous treatment systems, the study points toward a future where everyday plant waste helps protect rivers and communities from industrial pollution.
Citation: Abouzied, A.S., Kola, O.E., Al-Ahmary, K.M. et al. Isotherm, kinetic, and thermodynamic insights into textile effluent remediation using acid-treated sugarcane peel. Sci Rep 16, 7797 (2026). https://doi.org/10.1038/s41598-026-39059-5
Keywords: textile wastewater, sugarcane peel adsorbent, low-cost water treatment, dye and metal removal, sustainable remediation