Clear Sky Science · en
Traction force transmission via bioactive matrix hydrogel promotes epithelial collective migration mediated by integrin
How Cells Pull Together to Heal
When you cut your skin or damage an organ, sheets of cells must move in a coordinated way to seal the wound. This study asks a deceptively simple question: as cells crawl together, do they just tug on each other, or do they also "talk" through the soft material underneath them? By recreating a realistic, gel-like environment in the lab, the authors show that cells can send mechanical forces through this soft matrix to guide group movement, revealing a hidden layer of communication that may be crucial for wound healing, tissue repair, and even cancer spread.
A Soft Ground That Carries Force
In our bodies, many cells sit on a soft, protein-rich scaffold rather than on something rigid like glass or plastic. To mimic this, the researchers grew kidney epithelial cells as a continuous sheet on a bioactive hydrogel made of Matrigel and collagen, two common components of natural tissue. They then created a small empty zone in the middle of the gel, like a tiny wound, and watched the sheet of cells march inward to fill the gap. To track how active a key signaling pathway was inside the cells, they used a fluorescent biosensor that reports the activity of ERK, a protein that often responds to mechanical cues. This setup allowed them to see, in real time, how mechanical forces in the soft gel related to both cell movement and internal signaling. 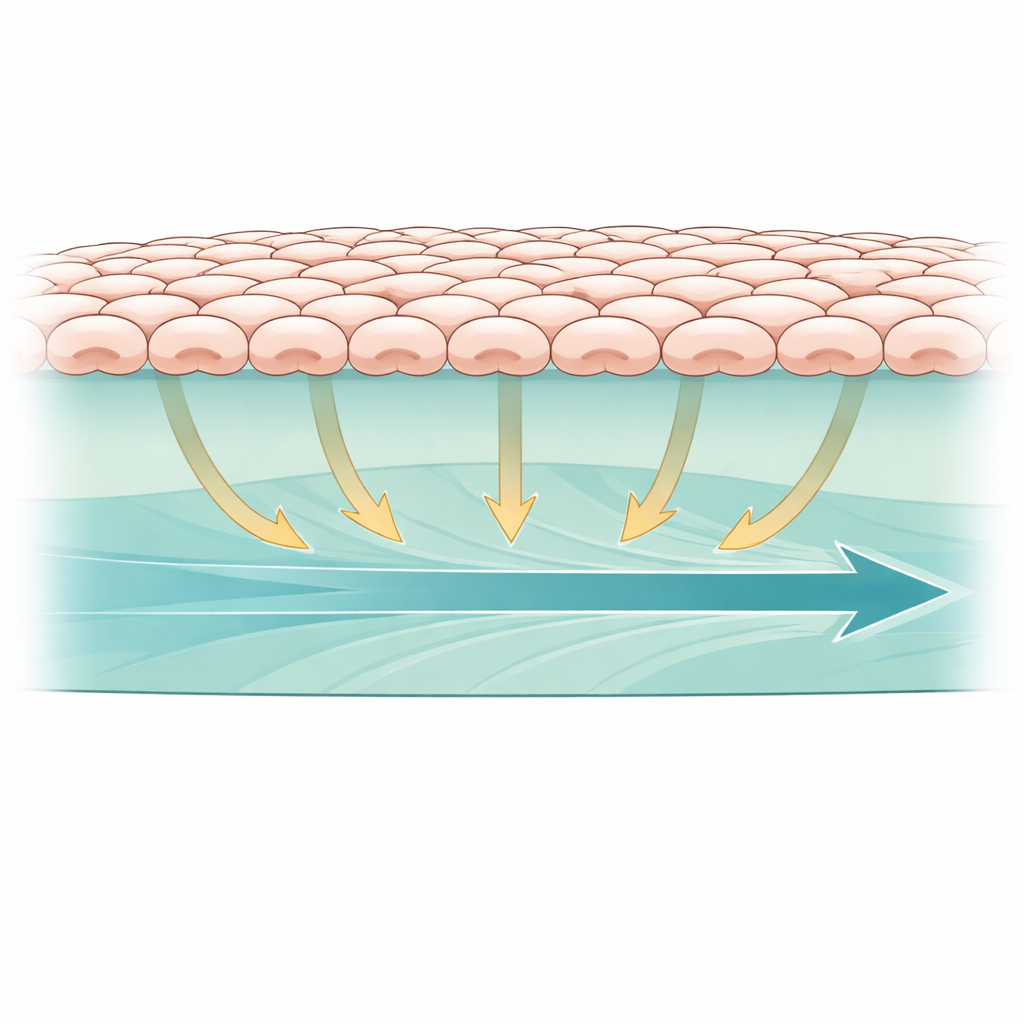
Watching the Ground Move Under the Cells
To tell whether the gel was actually bearing and transmitting force, the team mixed tiny magnetic beads into the material and followed their motion during cell migration. On the normal gel containing both Matrigel and collagen, the beads slowly slid forward in the same direction as the advancing cell sheet, indicating that the cells were gripping and dragging the matrix beneath them. On gels made of Matrigel alone, the beads moved more slowly, suggesting weaker force transmission. When the gel was chemically crosslinked with glutaraldehyde to stiffen and "lock" the network, the beads barely shifted at all. Under these locked conditions, the cells still attached but their collective advance slowed dramatically, and the ERK signal became weaker and less concentrated at the leading edge, tying strong matrix traction to faster, more directed group movement.
Internal Engines and Ion Gates Drive Motion
The authors next probed what inside the cells generates these pulling forces and how that activity is sensed. They used drugs to block myosin-based contraction, the same type of molecular machinery that powers muscle. When contraction was inhibited, ERK activity dropped and the sheet of cells advanced more slowly, consistent with the idea that internal pulling is needed to transmit tension into the matrix. They also interfered with several types of calcium channels in the cell membrane and the internal calcium store. Blocking these ion pathways dampened ERK activity and slowed collective migration, pointing to a chain of events in which mechanical tugging on the matrix is converted into calcium and ERK signals that help keep the group moving together.
Guiding Direction Through Surface Grips
A particularly striking finding came from targeting integrins, the surface molecules that act like tiny anchors connecting cells to their surroundings. When the researchers blocked a major integrin subtype, the sheet no longer moved forward as a coherent front, even though individual cells could still wiggle around locally at nearly normal speed. At the same time, bead motion in the gel almost stopped, showing that integrins are essential for transmitting force from the contracting cells into the matrix. In contrast, blocking Piezo1-related mechanosensitive channels, which help cells feel physical pressure, reduced both the pace of migration and the traction seen in the gel. Together, these results sketch a picture in which contraction generates force, integrins transmit it to the matrix, and mechanosensitive channels and ERK signaling interpret that force to coordinate the direction and efficiency of collective movement. 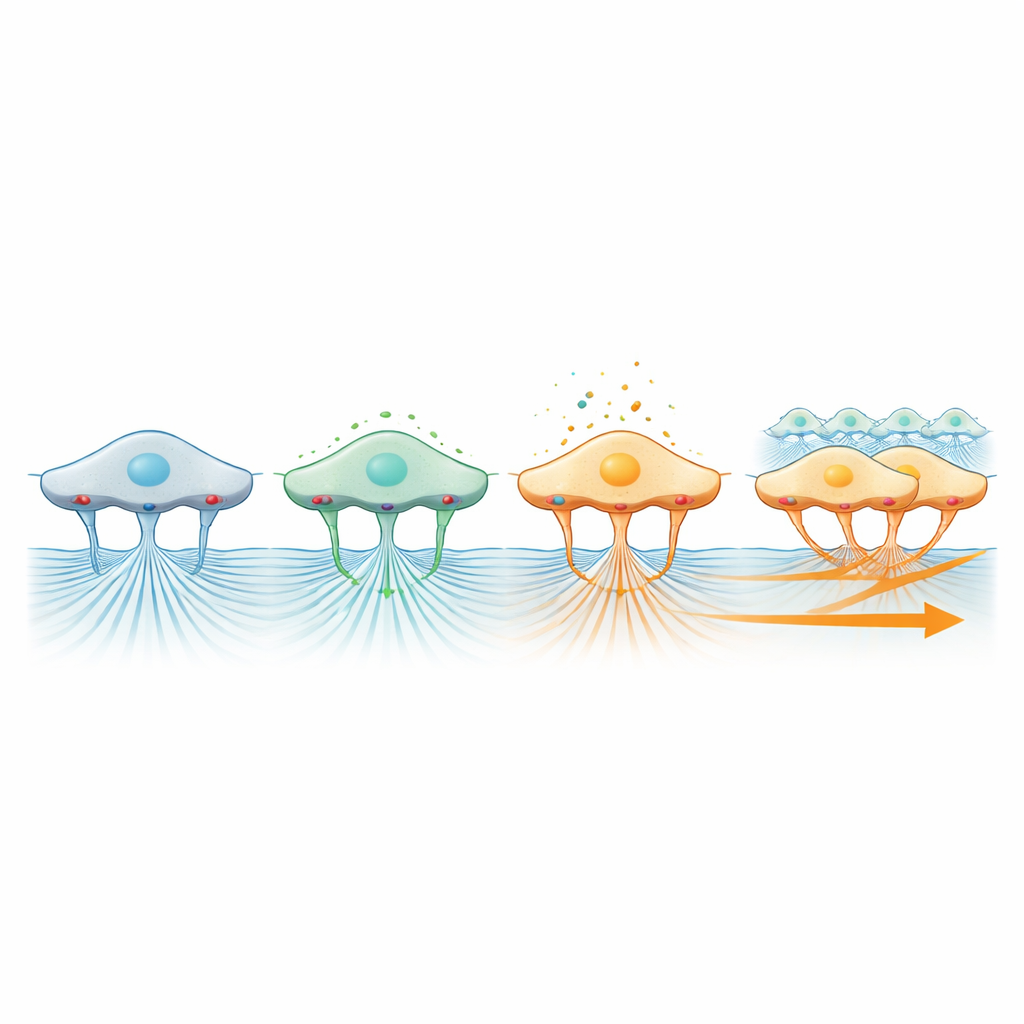
Why This Matters for Healing and Disease
Put simply, this work shows that sheets of cells do not just pull on each other; they also pull on, and through, the soft material beneath them, using it as a kind of mechanical telephone line. The cell layer’s contractions send tension through the matrix, and this tension, sensed via integrins and ion channels and read out by ERK activity, helps the group move quickly and in a unified direction. When the matrix is chemically locked so that forces cannot propagate, or when key surface grips and sensors are blocked, the collective march falters. These insights deepen our understanding of how wounds close, how tissues maintain their structure, and how invasive cell groups, such as those in cancer and fibrosis, might exploit mechanical pathways in their environment to spread.
Citation: Ouyang, M., Cao, Y., Sheng, H. et al. Traction force transmission via bioactive matrix hydrogel promotes epithelial collective migration mediated by integrin. Sci Rep 16, 8923 (2026). https://doi.org/10.1038/s41598-026-39048-8
Keywords: collective cell migration, mechanical signaling, extracellular matrix, integrin, wound healing