Clear Sky Science · en
Interface-directed design of glycidyl methacrylate-functionalized UiO-66 for thin film nanofiltration membranes in heavy metals rejection
Cleaner Water from Smart Filters
Lead and arsenic in drinking water may sound like problems from the past, but they remain serious threats in many parts of the world. This study explores a new type of "smart" water filter that can more effectively strain out these toxic metals before they reach the tap. By carefully redesigning the microscopic interface inside a common filtration membrane, the researchers show how subtle chemistry can translate into safer water without needing huge amounts of energy or chemicals.
Why Heavy Metals Are Hard to Remove
Unlike many organic pollutants that can break down over time, heavy metals such as lead and arsenic persist and build up in ecosystems and human bodies. Conventional treatments—like adding chemicals to force metals to settle out, or using absorbent powders—can work, but they often create sludge that must be handled as hazardous waste and may struggle at the very low concentrations relevant for drinking water. Pressure-driven membranes offer a cleaner route: water is pushed through a thin barrier that holds back larger or more highly charged particles. Nanofiltration, a type of membrane process that sits between ultrafiltration and reverse osmosis, is especially promising because it can remove harmful multivalent ions, like many metal species, while still allowing some useful mineral salts to pass.
From Standard Films to Nanocomposite Filters
Most commercial nanofiltration systems rely on a thin film composite membrane. This is essentially a sandwich: a very thin, dense, selective polyamide skin is formed on top of a more porous support. The top skin does the fine filtering, while the support gives mechanical strength. However, there is a built-in trade-off. Making the top layer tighter improves rejection of contaminants but tends to slow down water flow. Researchers have tried adding tiny particles—such as metal–organic frameworks (MOFs), porous crystals made from metal nodes and organic linkers—into this top layer to create thin film nanocomposite membranes. In principle, MOFs can offer extra internal pores and chemical sites that help water move quickly while grabbing target pollutants. In practice, though, mixing rigid crystals into a soft polymer can cause poor contact, gaps, or clumps that leak or weaken performance.
Grafting a Better Interface
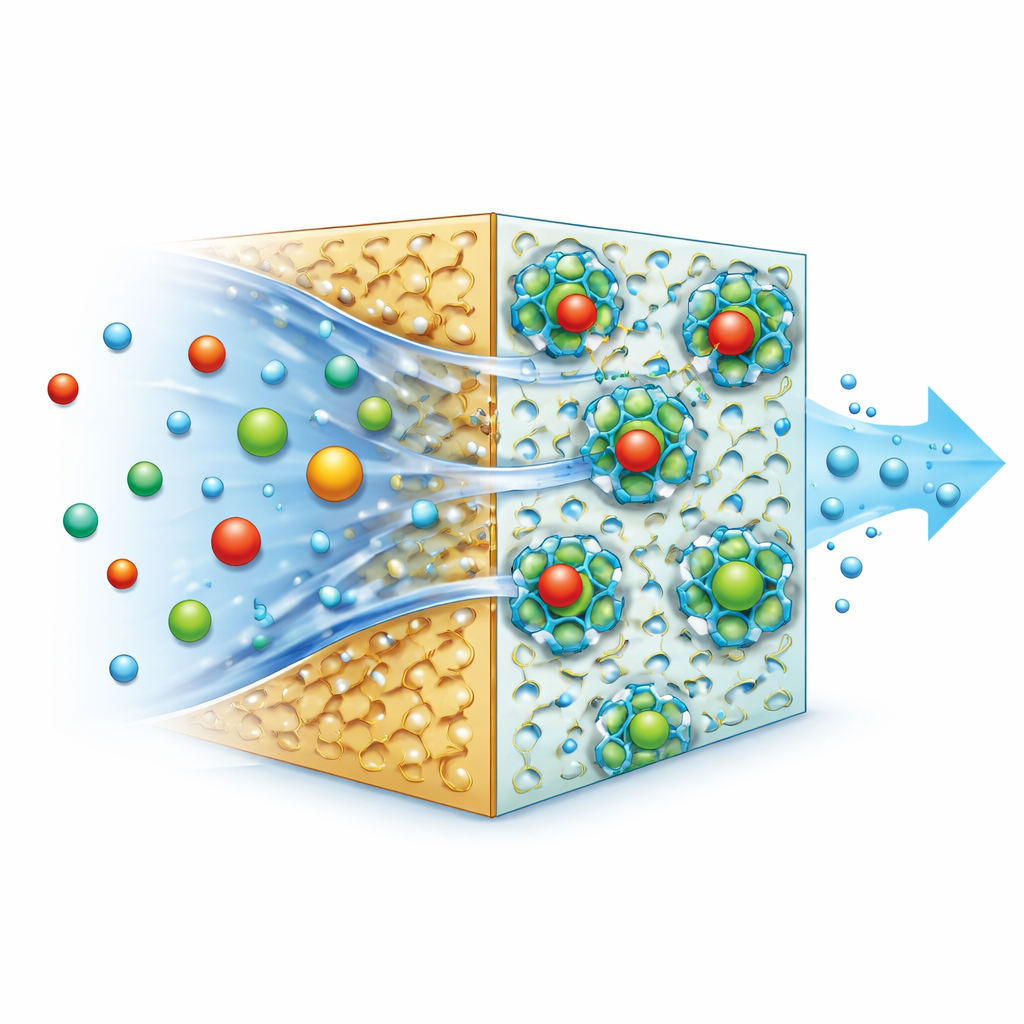
To tackle this compatibility problem, the team focused not on packing in more pores, but on engineering the boundary where the MOF meets the polyamide. They started with UiO-66-NH₂, a zirconium-based MOF known for its stability in water. They then chemically attached a small organic molecule called glycidyl methacrylate to the MOF’s surface, creating GMA–UiO‑66. This adjustment adds reactive and polar groups that can interact strongly with the forming polyamide layer. Tests using X-ray diffraction and infrared spectroscopy showed that the crystal structure of UiO‑66 remained intact after this treatment, though some of its internal surface area and pore volume were gently reduced as the new chains partially occupied existing pores. Electron microscopy revealed that membranes made with the modified MOF had a more continuous, defect-free top layer compared with those made with the unmodified version.
How the New Membranes Perform
The researchers fabricated a series of membranes on a porous polyacrylonitrile support and varied how much MOF they added. They then filtered water containing relatively high levels of lead and arsenate—50 milligrams per liter, far above typical drinking-water limits—under moderate pressure. As MOF content increased, both water flow and metal rejection improved for all membranes. Those made with unmodified UiO‑66‑NH₂ already showed better performance than plain polyamide. But the GMA‑UiO‑66 versions did even better, despite having slightly lower overall porosity. At the optimal loading, the modified membrane rejected about 97% of lead and 93% of arsenate while maintaining stable water flux. Measurements of porosity, water contact angle, and imaging of the cross-sections all pointed to the same conclusion: the tailored interface between MOF and polymer creates more efficient pathways for water while tightening the barrier against metal ions.
What This Means for Real-World Water Treatment
Even with above-90% rejection, a single pass through these membranes would not always push metal levels all the way down to strict drinking-water standards when starting from very contaminated sources. Instead, the authors argue that their design is best viewed as a powerful pretreatment step. In such a role, the membrane would greatly reduce the metal load before further polishing steps, cutting the burden on downstream systems. Just as important, the study offers a clear mechanistic lesson: by thoughtfully modifying the surface of porous particles, engineers can strengthen the "handshake" between fillers and polymers, overcoming the usual trade-off between speed and selectivity. This interface-directed approach could guide the next generation of nanocomposite membranes aimed not only at heavy metals, but also at other emerging contaminants in our increasingly stressed water supplies.
Citation: Yousaf, I., Haq, N.U., Batool, M. et al. Interface-directed design of glycidyl methacrylate-functionalized UiO-66 for thin film nanofiltration membranes in heavy metals rejection. Sci Rep 16, 9443 (2026). https://doi.org/10.1038/s41598-026-39047-9
Keywords: heavy metal removal, nanofiltration membranes, metal–organic frameworks, water purification, polyamide thin films