Clear Sky Science · en
MUC14 suppresses lung adenocarcinoma via integrin α8β6/PI3K/AKT/MAPK modulating cisplatin response and immunity
Why this lung cancer study matters
Lung adenocarcinoma is the most common form of lung cancer and is often treated with the chemotherapy drug cisplatin. Unfortunately, many tumors learn to resist this drug, and patients still face low long-term survival. This study uncovers a little-known cell-surface protein, MUC14, that appears to act as a natural brake on lung tumors. Understanding how MUC14 works may open new ways to make chemotherapy more effective and to help the immune system better recognize and attack cancer cells.
A protective protein hiding in plain sight
MUC14, also called endomucin, sits on the surface of cells and belongs to a family of sugar-coated proteins known as mucins. By mining large public cancer databases and examining patient samples, the researchers found that lung adenocarcinoma tumors usually produce much less MUC14 than nearby normal tissue. Patients whose tumors kept higher levels of MUC14 lived longer and had slower disease progression than those with low levels. Across many cancer types, MUC14 tended to be suppressed, suggesting it often acts as a tumor blocker rather than a tumor promoter. 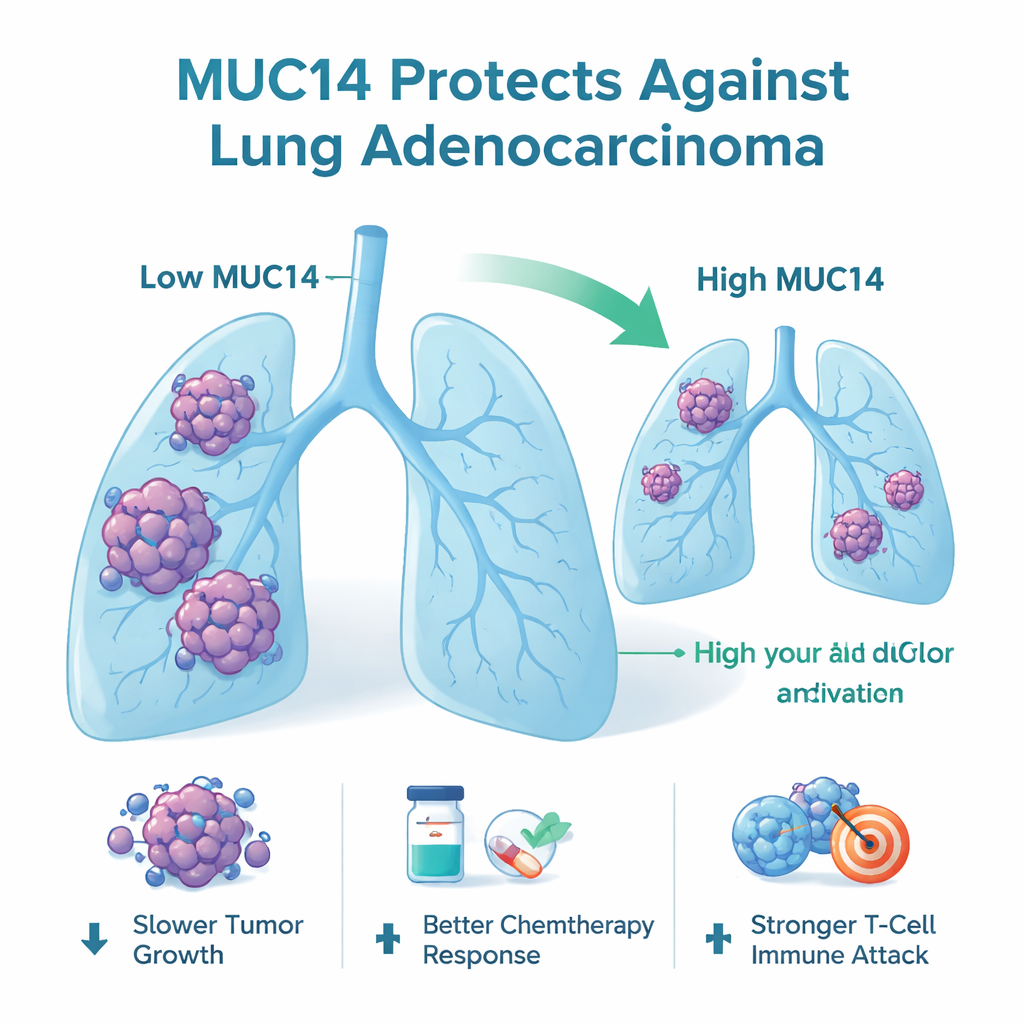
Slowing cancer cells and shrinking tumors
To see what MUC14 actually does, the team manipulated its levels in lung cancer cell lines. When they forced cells to make extra MUC14, the cells divided more slowly, migrated less, and were worse at invading through a gel that mimics body tissue. They also had a harder time sticking to key scaffold molecules, and they formed fewer and smaller colonies. In mice, cancer cells engineered to overproduce MUC14 formed smaller tumors under the skin and caused fewer lung metastases after being injected into the bloodstream. When MUC14 was instead switched off with small interfering RNAs, the opposite happened: cells grew faster, moved more, stuck better, and formed more colonies, all signs of a more aggressive cancer behavior.
Cutting off growth signals at the cell surface
The authors then asked how MUC14 exerts these powerful effects. They focused on integrins, a family of receptor proteins that anchor cells to their surroundings and relay growth and survival signals inside the cell. Data from hundreds of lung tumors pointed to a strong link between MUC14 and two integrin components, called α8 and β6, which pair up on the cell surface. Using a light-based technique that detects proteins within billionths of a meter of each other, the researchers confirmed that MUC14 and the α8β6 integrin complex sit side by side in the cell membrane. Extra MUC14 disrupted clustering of these integrins and dampened two major growth pathways they normally feed: PI3K/AKT and MAPK/ERK. When MUC14 was reduced, these pathways became more active, helping explain why cancer cells grew and spread more readily. 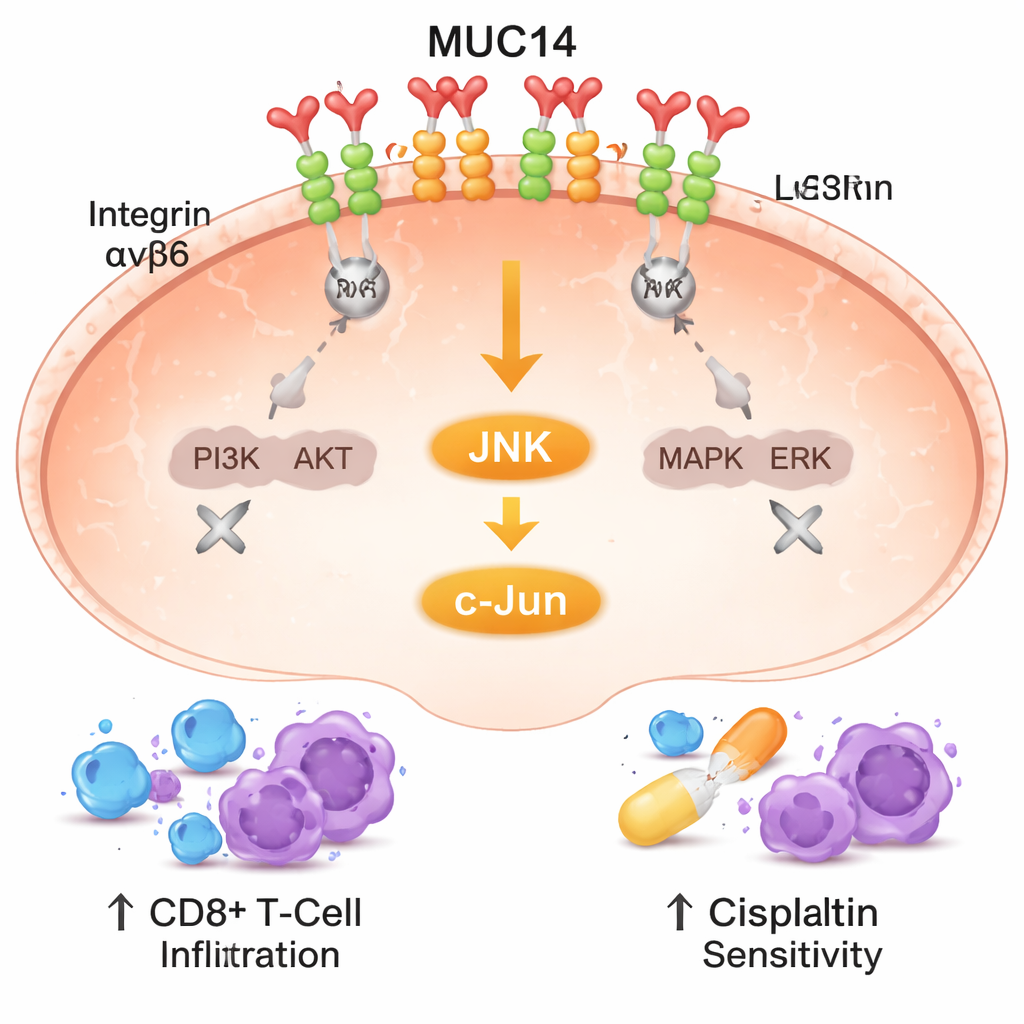
Helping the immune system and chemotherapy work together
Cancer outcome is shaped not only by tumor cells themselves but also by nearby immune cells. By analyzing immune profiling databases and staining human tumor sections, the team found that tumors with higher MUC14 levels had more CD3+ and CD8+ T cells—immune cells that can directly kill cancer cells—nestled close to MUC14-positive tumor regions. This suggests MUC14 helps create a friendlier environment for immune attack. The researchers also explored cisplatin, a mainstay drug for lung adenocarcinoma. In cell culture, tumors with extra MUC14 needed much lower doses of cisplatin to be inhibited, while cells lacking MUC14 became more drug resistant. Importantly, MUC14 did not simply make cells more prone to die at baseline; instead, it amplified cisplatin’s killing effect, in part by activating a stress pathway known as JNK/c-Jun that is triggered by DNA damage.
Toward smarter combinations for lung cancer care
Put simply, this work paints MUC14 as a built-in defense system against lung adenocarcinoma. When present, it blocks key growth switches on the cell surface, slows tumor spread, draws in cancer-fighting T cells, and makes cisplatin treatment hit harder without extra organ damage in animal tests. When it is lost, tumors become tougher, more mobile, and less responsive to standard therapy. The findings suggest that measuring MUC14 could help predict prognosis and treatment response, and that future therapies designed to boost MUC14 activity—or mimic its effects on integrin signaling—might allow doctors to combine chemotherapy and immunotherapy more effectively for patients with advanced lung cancer.
Citation: Li, X., Li, M., Huang, S. et al. MUC14 suppresses lung adenocarcinoma via integrin α8β6/PI3K/AKT/MAPK modulating cisplatin response and immunity. Sci Rep 16, 7784 (2026). https://doi.org/10.1038/s41598-026-39019-z
Keywords: lung adenocarcinoma, MUC14, cisplatin resistance, integrin signaling, tumor immunity