Clear Sky Science · en
Bee pollen-derived peptide with dual DPP-IV Inhibition and glucose transport modulation
Why bee pollen could matter for blood sugar
Type 2 diabetes is often framed as a problem of too much sugar and too little insulin, but behind the scenes a web of enzymes and transporters quietly steer how our bodies handle glucose. This study explores an intriguing idea: that bee pollen, a natural "superfood," might be a source of tiny protein fragments (peptides) that both protect hormone signals controlling blood sugar and gently tune how sugar is absorbed from the gut. The work focuses on one such peptide, called AA-7, and asks whether it could someday inspire safer, food-based strategies to help manage diabetes.
From hive to lab bench
Bee pollen is a mix of plant pollen and bee-derived materials, rich in proteins, vitamins, and other nutrients. To mimic human digestion, the researchers treated bee pollen proteins with two digestive enzymes, pepsin and pancreatin, similar to those in our stomach and small intestine. This process broke large proteins into many smaller pieces, some only a few amino acids long. They then separated these fragments by size and chemical properties, testing each fraction for its ability to block an enzyme called DPP-IV, which normally chops up and inactivates hormones that stimulate insulin release. One fraction of very small, relatively oily (hydrophobic) peptides stood out for its strong DPP-IV-blocking activity. 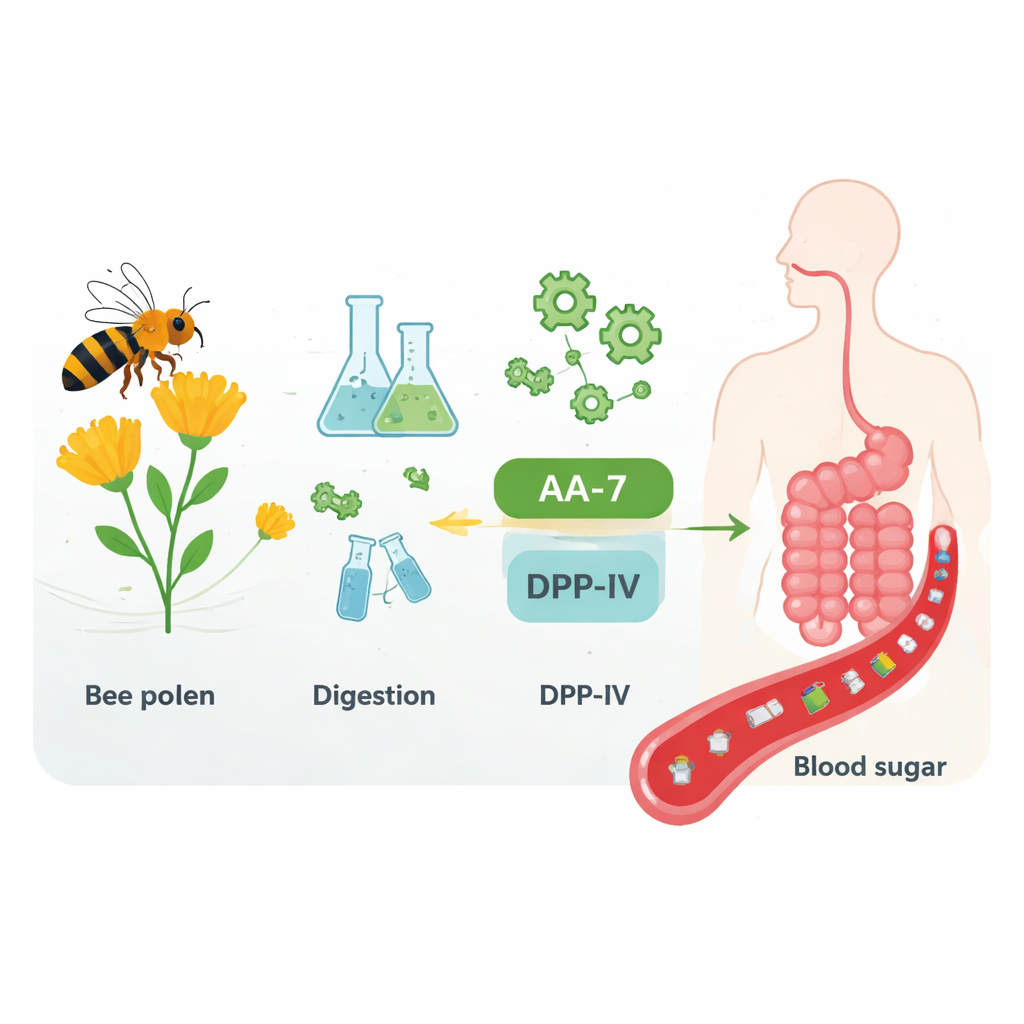
Finding a standout peptide
Using high-resolution mass spectrometry, the team identified a seven–amino acid peptide with the sequence Ala-Thr-His-Ala-Leu-Leu-Ala, which they named AA-7. They synthesized this peptide to test it in isolation. In enzyme assays, AA-7 inhibited DPP-IV at micromolar concentrations, somewhat weaker than a standard lab drug but comparable to or better than many food-derived peptides reported in the scientific literature. By examining how the reaction rate changed with different amounts of peptide and substrate, they showed that AA-7 acts as a competitive inhibitor: it sits in the same active pocket of DPP-IV that the natural substrates use, getting in the way without destroying the enzyme.
Peptide–enzyme interactions in atomic detail
To understand why AA-7 works, the researchers used computer simulations to dock the peptide into a three-dimensional model of DPP-IV and then ran molecular dynamics simulations over time. The models suggested that AA-7 nests in the enzyme’s catalytic pocket, contacting key amino acids that are already known to be crucial for DPP-IV’s activity. The binding was predicted to be at least as stable as that of a reference inhibitor. Further computer analyses of absorption, distribution, metabolism, and toxicity (ADMET) offered a realistic caution: AA-7 is likely safe but not efficiently absorbed through the gut wall as-is, a common limitation for small peptides. This points to a need for delivery strategies or structural tweaks if AA-7, or molecules modeled on it, are ever used in people.
Tuning how the gut handles sugar
The study did not stop at enzymes in test tubes. The team exposed cultured human intestinal cells (Caco-2 cells) to AA-7 and measured both glucose uptake and the activity of genes that encode two main intestinal glucose transporters, SGLT1 and GLUT2. At non-toxic doses, AA-7 altered how quickly a fluorescent form of glucose entered the cells, with effects depending on dose and time. It also changed the levels of SGLT1 and GLUT2 gene expression in different ways over short (30-minute) and longer (24-hour) periods. Computer docking suggested that AA-7 can interact with structural regions of these transporters, although these models are meant as hypotheses rather than final proof. Together, the cell and modeling data suggest that AA-7 does more than block DPP-IV—it also nudges the machinery that moves sugar from the gut into the bloodstream. 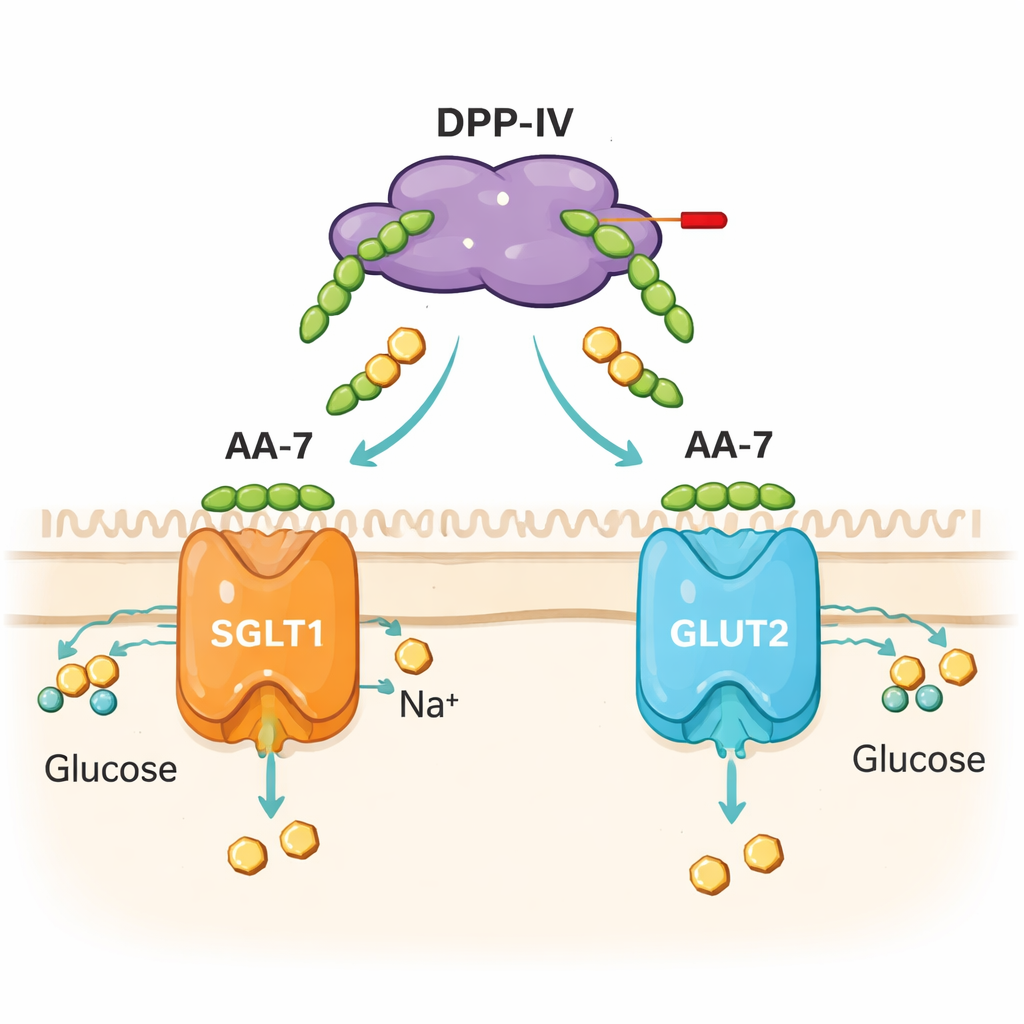
What this could mean for future foods
For a non-specialist, the take-home message is that the bee-pollen peptide AA-7 appears to act on two fronts relevant to type 2 diabetes: it slows down a key enzyme that degrades insulin-stimulating hormones, and it influences how intestinal cells take up glucose. On its own, AA-7 is not ready to be a drug or supplement, partly because it may not be absorbed efficiently. However, it points to bee pollen—and perhaps other protein-rich foods—as reservoirs of small, multitasking peptides that could inspire new functional foods or peptide-based treatments. With further work in animals and humans, and with better ways to deliver such peptides, this kind of dual-action molecule could become part of a more nuanced, food-informed approach to keeping blood sugar in check.
Citation: Mongkolnkrajang, U., Kuptawach, K., Sangtanoo, P. et al. Bee pollen-derived peptide with dual DPP-IV Inhibition and glucose transport modulation. Sci Rep 16, 7616 (2026). https://doi.org/10.1038/s41598-026-39009-1
Keywords: bee pollen peptides, DPP-IV inhibition, glucose transport, type 2 diabetes, functional foods