Clear Sky Science · en
Transplastomic biofactory for the production of functional human α-lactalbumin for nutritional and therapeutic applications
Turning Plants into Milk Protein Makers
Modern diets depend heavily on milk proteins, but raising cows at global scale comes with environmental costs and ethical questions. This study explores a very different way to make a key human milk protein—by teaching plant leaves to produce it. The work shows that tobacco plants can be rewired to manufacture human α‑lactalbumin, a protein important for infant nutrition and with potential anti‑cancer properties, opening a path toward animal‑free baby formula ingredients and new cancer‑fighting food components.
Why This Milk Protein Matters
Human breast milk is more than just food; it is a finely tuned cocktail of proteins, fats, and sugars that changes with an infant’s needs. One of its most important proteins is α‑lactalbumin, which helps build lactose, the main sugar in milk and a critical energy source for babies. α‑Lactalbumin is rich in essential amino acids, including tryptophan, a building block for the brain’s “feel‑good” chemical serotonin, and has been linked to better stress resilience and potential protection against certain cancers. Because it is easy to digest, has a mild taste, and remains stable over a wide range of acidity, it is widely used in infant formulas and nutritional drinks—and demand is rising.
The Problem with Traditional Dairy
Relying on cows to supply milk proteins has drawbacks. Livestock produce a sizable share of global greenhouse gas emissions, and large dairy operations require vast amounts of land, water, and feed. On top of that, farm management can introduce concerns about hormones, antibiotics, and pathogens entering the milk stream. These pressures have fueled interest in “cellular agriculture” approaches, where milk proteins are made by microbes or cultured cells instead of animals. Yet many of these systems are still expensive, hard to scale, or produce proteins that are not quite identical to the human versions. Plants, especially their leaf chloroplasts—the tiny green factories that drive photosynthesis—offer a promising alternative platform.
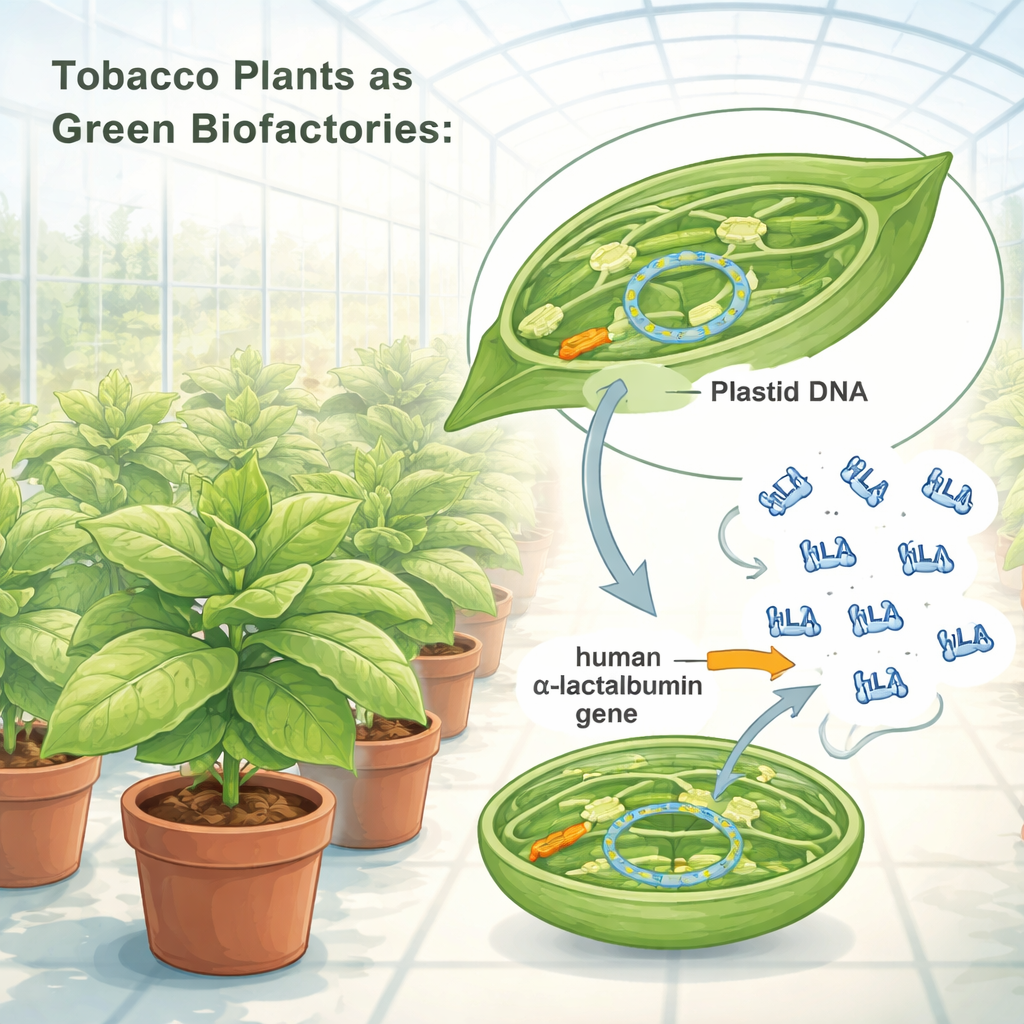
Reprogramming Chloroplasts as Biofactories
The researchers redesigned the genetic instructions for human α‑lactalbumin so that tobacco chloroplasts could read them efficiently. They inserted this optimized gene into the chloroplast DNA, using a “gene cassette” built from strong plant regulatory elements that act like powerful on–off switches. Using a particle‑bombardment method, they delivered this cassette into tobacco leaf cells, then selected plantlets whose chloroplasts had fully adopted the new gene. Genetic tests confirmed that all copies of the chloroplast DNA in these plants now carried the human protein blueprint. The modified plants grew normally in greenhouse conditions, producing seeds and showing no visible penalties in size, shape, or fertility, suggesting that the extra protein load did not harm plant health.
Proving the Plant-Made Protein Works
Next, the team asked whether the plant‑made α‑lactalbumin behaved like the real human protein. They extracted soluble proteins from the leaves, purified the α‑lactalbumin, and examined its structure using a technique that reports on how proteins fold. The spectral “fingerprint” of the plant‑derived protein nearly overlapped with that of commercial human α‑lactalbumin, indicating similar folding. Functionally, the plant‑made protein activated the lactose‑synthesizing enzyme almost as well as the native version, driving lactose formation at about 93% of the standard protein’s rate. Remarkably, α‑lactalbumin accumulated to about 23% of all soluble protein in the leaves—among the highest levels reported for human proteins made in plant chloroplasts—suggesting that this system can deliver industrially relevant yields.
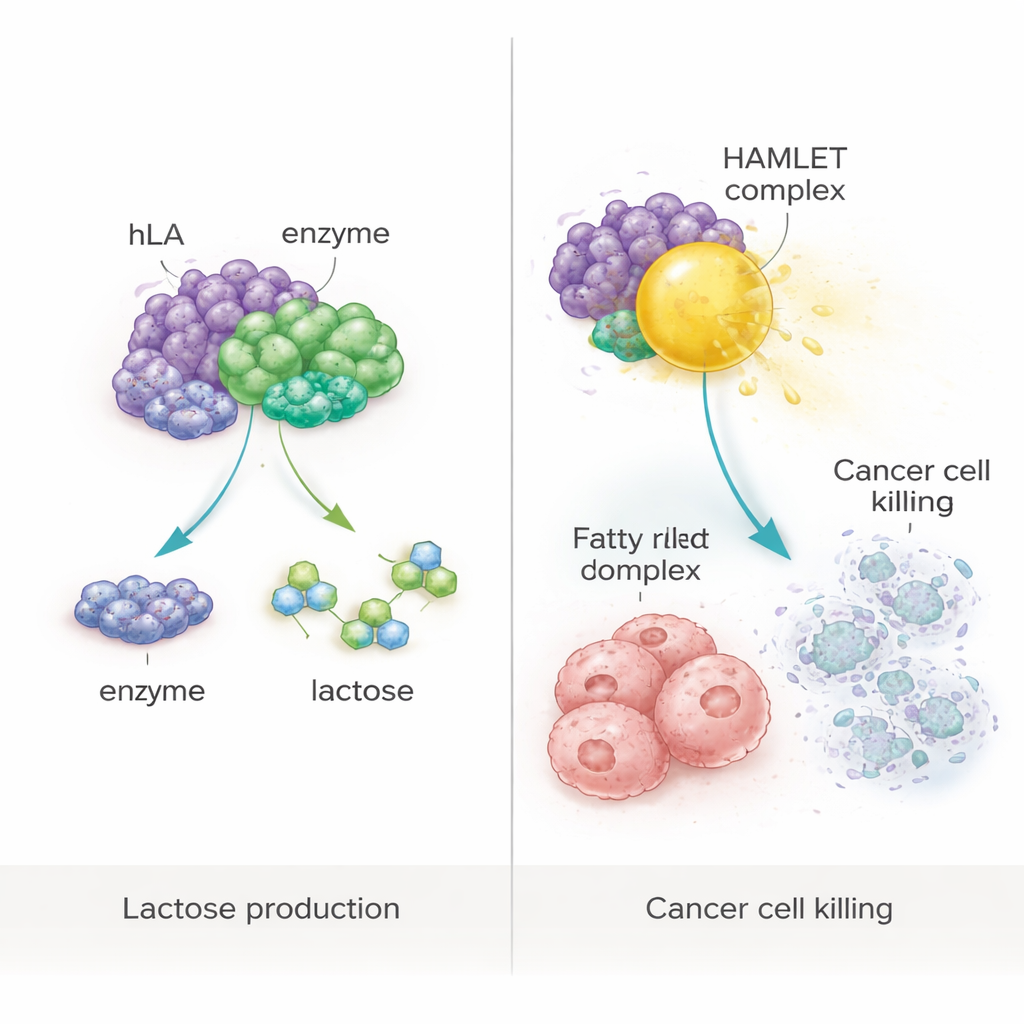
From Nutrition to Cancer-Fighting Complexes
The study also tested a more ambitious idea: using the plant‑made protein as a building block for a known anti‑cancer complex called HAMLET, formed when α‑lactalbumin binds tightly to oleic acid, a common fatty acid. By gently heating the purified plant protein with oleic acid under controlled conditions, the researchers created a HAMLET‑like complex. When applied to human colorectal (WiDr) and breast (MCF‑7) cancer cells in culture, this complex dramatically reduced cell survival to below 10%. Follow‑up analysis showed that most of the affected cells were undergoing apoptosis, a form of programmed cell death rather than uncontrolled rupture. Importantly, neither the protein alone, the fatty acid alone, nor extracts from unmodified plants triggered significant cell death, confirming that the cancer‑killing effect depended on the specific complex.
What This Could Mean for Future Foods and Medicines
Taken together, the work shows that plant chloroplasts can reliably produce large amounts of a human milk protein that is both nutritionally relevant and therapeutically active. For a non‑specialist, this means that future infant formulas, functional foods, or supplements might one day be made using proteins grown in leaves rather than harvested from cows, reducing environmental impact while matching human biology more closely. At the same time, the very same plant‑derived protein can be converted into a targeted anti‑cancer complex in the lab, hinting at plant‑based pipelines for affordable biotherapeutics. While more research is needed to confirm safety and performance in animals and people—and to transfer this technology into edible crops—the study offers a compelling glimpse of plants doubling as mini‑factories for next‑generation nutrition and medicine.
Citation: Ehsasatvatan, M., Kohnehrouz, B.B. Transplastomic biofactory for the production of functional human α-lactalbumin for nutritional and therapeutic applications. Sci Rep 16, 7359 (2026). https://doi.org/10.1038/s41598-026-38965-y
Keywords: plant-made milk proteins, chloroplast biofactory, human alpha-lactalbumin, sustainable dairy alternatives, HAMLET cancer therapy