Clear Sky Science · en
miR-340 improves the efficiency of p53 gene therapy in metastatic prostate cancer cells through downregulation of MDM2
Why this research matters for patients
For men with advanced prostate cancer that has spread and no longer responds to hormone therapy, today’s treatments can slow the disease but rarely cure it, and only about half of patients live longer than five years. This study explores a new way to make existing treatments work better by reinforcing one of the body’s own natural defenses against cancer—the p53 “guardian” protein—using a tiny genetic helper called miR-340.
Reawakening the body’s tumor guardian
Many cancers, including aggressive prostate tumors, disable p53, a protein that normally halts damaged cells, repairs them, or triggers their self-destruction. One approach, already tested in patients with other cancers, is p53 gene therapy: delivering a healthy copy of the p53 gene into tumor cells so they once again respond to damage. But tumors often fight back by making more of MDM2, a protein that tags p53 for destruction. The authors reasoned that if they could lower MDM2 at the same time as adding p53, they might restore the full power of this tumor-guarding pathway in difficult-to-treat prostate cancer.
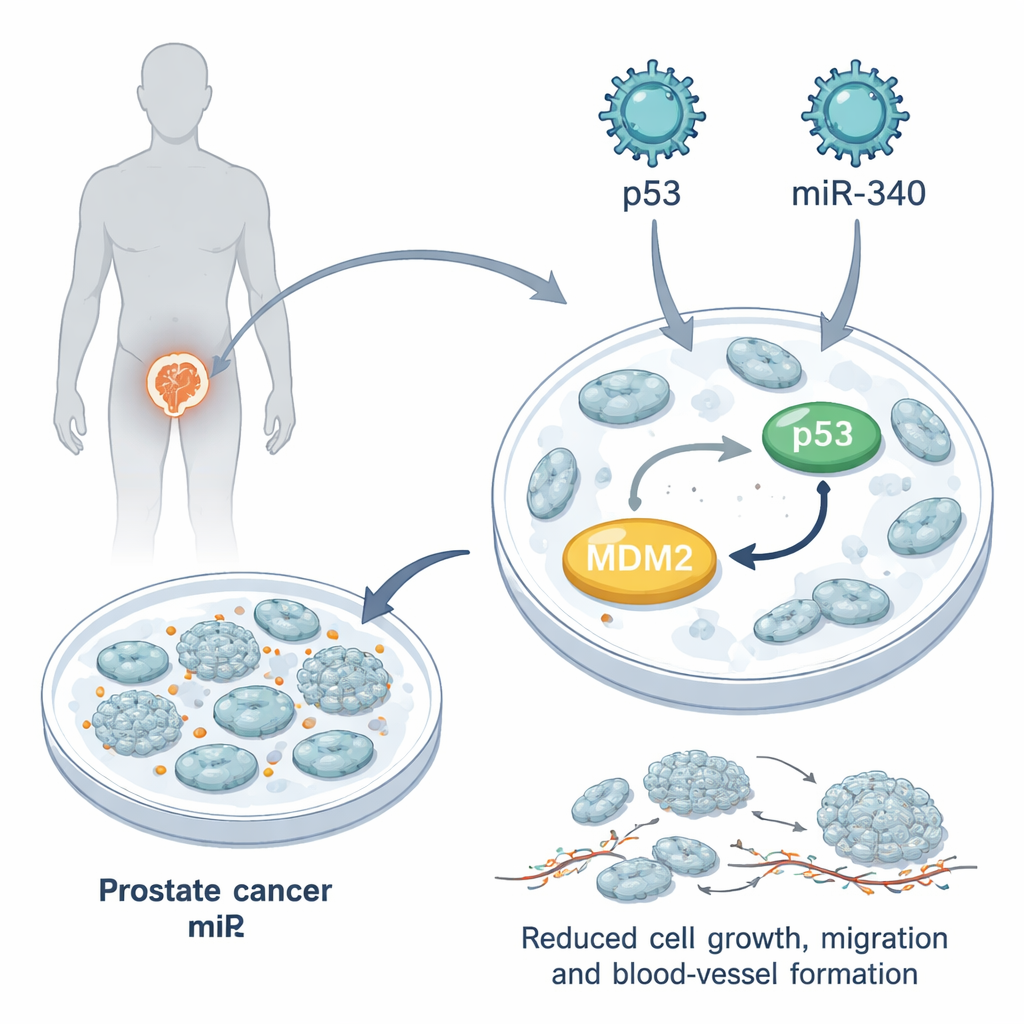
A tiny RNA with a big impact
The team focused on miR-340, a small RNA molecule naturally present in cells that helps fine-tune gene activity. Database analyses showed that miR-340 levels are lower in prostate tumors than in healthy prostate tissue, and even lower in castration-resistant disease. Earlier work suggested that miR-340 can bind to MDM2’s genetic message and reduce its production. In this study, the researchers used engineered lentiviral particles to introduce extra miR-340, extra p53, or both together into PC3 prostate cancer cells, a widely used lab model that completely lacks functional p53. They confirmed that miR-340 strongly reduced MDM2 and that the combination of miR-340 plus p53 led to much higher levels of p53’s key partner p21, which is crucial for stopping cell division.
Slowing cancer cell growth and spread
Once the prostate cancer cells had been modified, the researchers tested how quickly they grew, how they moved, and how well they could support blood-vessel formation, all hallmarks of aggressive tumors. p53 alone slowed cell growth and caused more cells to pause in the resting G0/G1 phase of the cell cycle. miR-340 alone did not have a major effect on growth, but when combined with p53 it greatly amplified this slowdown: the cells took longer to double in number and formed smaller, weaker 3D tumor-like spheroids. In scratch “wound” tests that mimic tumor cell migration, miR-340 by itself barely changed how cells moved, but it boosted p53’s ability to keep cells from crawling into the gap. The combination also reduced signals that encourage blood vessel growth, and fluid taken from treated prostate cancer cells made blood-vessel cells grow and move less.
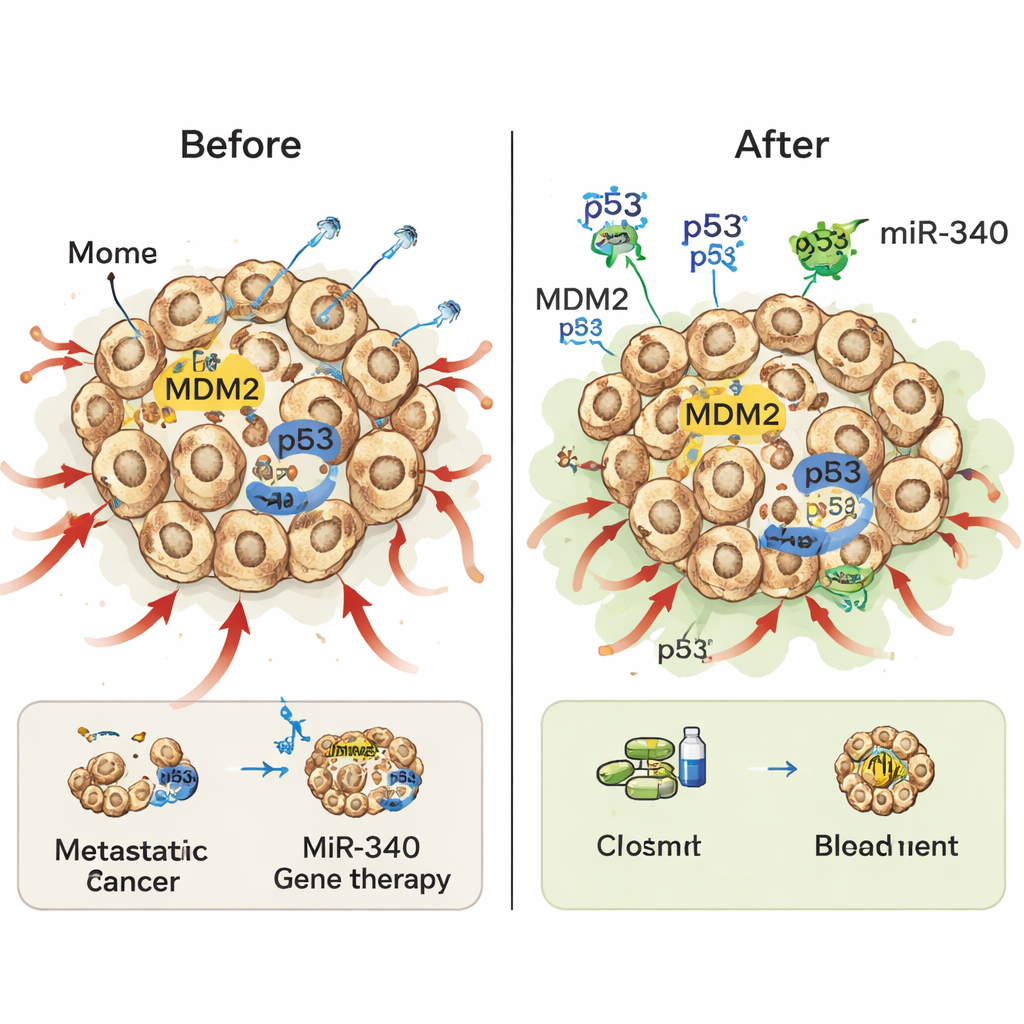
Making chemo and radiation hit harder
A striking finding was that the combined miR-340/p53 gene therapy made standard treatments more potent without being strongly toxic on its own. When the modified prostate cancer cells were exposed to low doses of the chemotherapy drug docetaxel, cells with both miR-340 and p53 showed a sharp rise in programmed cell death compared with cells given p53 alone or miR-340 alone. A similar pattern appeared with X‑ray radiation: miR-340 by itself did little, but together with p53 it made cancer cells far more sensitive, sharply reducing their ability to form colonies after irradiation. In both cases, miR-340 appears to help by cutting down MDM2, allowing the added p53 to accumulate and fully activate its death-inducing programs once chemotherapy or radiation has damaged the cancer cells.
What this could mean for future care
The authors conclude that adding miR-340 to p53 gene therapy can meaningfully strengthen this approach against metastatic, treatment-resistant prostate cancer—particularly when used alongside existing chemo and radiotherapy. While this work was done in cells in the lab and still faces practical hurdles, such as improving gene delivery methods and confirming safety in animals and people, it outlines a clear and testable strategy: weaken the tumor’s brakes on p53 by lowering MDM2, then let standard treatments trigger a more powerful, targeted response. If future studies bear this out, patients could one day receive lower doses of toxic drugs while still achieving stronger tumor control.
Citation: Deyhimfar, R., Kehtari, M., Saadatpour, F. et al. miR-340 improves the efficiency of p53 gene therapy in metastatic prostate cancer cells through downregulation of MDM2. Sci Rep 16, 7527 (2026). https://doi.org/10.1038/s41598-026-38963-0
Keywords: prostate cancer, gene therapy, p53, MDM2, microRNA-340