Clear Sky Science · en
Investigation of water softening using ceramic adsorbents in a continuous adsorption process
Cleaner Water from Simple Minerals
Many people worry about germs or chemicals in their tap water, but another invisible problem quietly clogs pipes and appliances: hardness. Hard water, rich in dissolved minerals, can shorten the life of washing machines, boilers, and even affect our health. This study explores how inexpensive, rock-like materials called ceramics can be used in simple columns to strip hardness from drinking water continuously, offering a practical route to safer, gentler water for homes and communities.
Why Hard Water Is a Hidden Burden
Hardness in water mainly comes from calcium and magnesium. At high levels, these minerals leave crusty deposits in kettles and pipes, drive up energy bills by insulating heating elements, and force utilities to spend more on maintenance. Some studies also link long-term exposure to very hard water with health problems such as kidney stones and high blood pressure. Conventional softening methods, like ion exchange resins or advanced membranes, can work well but are costly, often require sophisticated operation, and may not suit smaller or resource-limited communities. The appeal of ceramic minerals is that they are cheap, abundant, and naturally good at trapping unwanted ions.
Turning Rocks into Water Filters
The researchers focused on two ceramic materials already used in water treatment: a natural mineral called clinoptilolite zeolite and a manufactured product known as activated alumina. Both were packed into narrow vertical tubes, or fixed-bed columns, through which hard drinking water from a city in Iran was pumped for up to ten hours at a time. By adjusting how wide the columns were and how fast the water flowed, the team tested how much hardness the beds could remove before they “filled up” and started letting minerals through again. They also chemically “tuned” the surfaces: the zeolite was treated with a sodium-rich salt solution, and the alumina was exposed to sulfuric acid to add new reactive sites.
What Happens Inside the Mineral Beds
Under the microscope and with surface-analysis tools, the team saw that both ceramics had porous, craggy structures that offered many nooks for ions to lodge in. Treating the zeolite with sodium made its surface more uniformly covered with easily swapped ions, so incoming calcium and magnesium could more readily kick sodium off and take its place. The modified alumina became rougher and more porous after acid treatment, adding new spots where ions could stick. Measurements of surface charge showed that zeolite carried a stronger negative charge than alumina, helping it attract positively charged hardness ions more effectively.
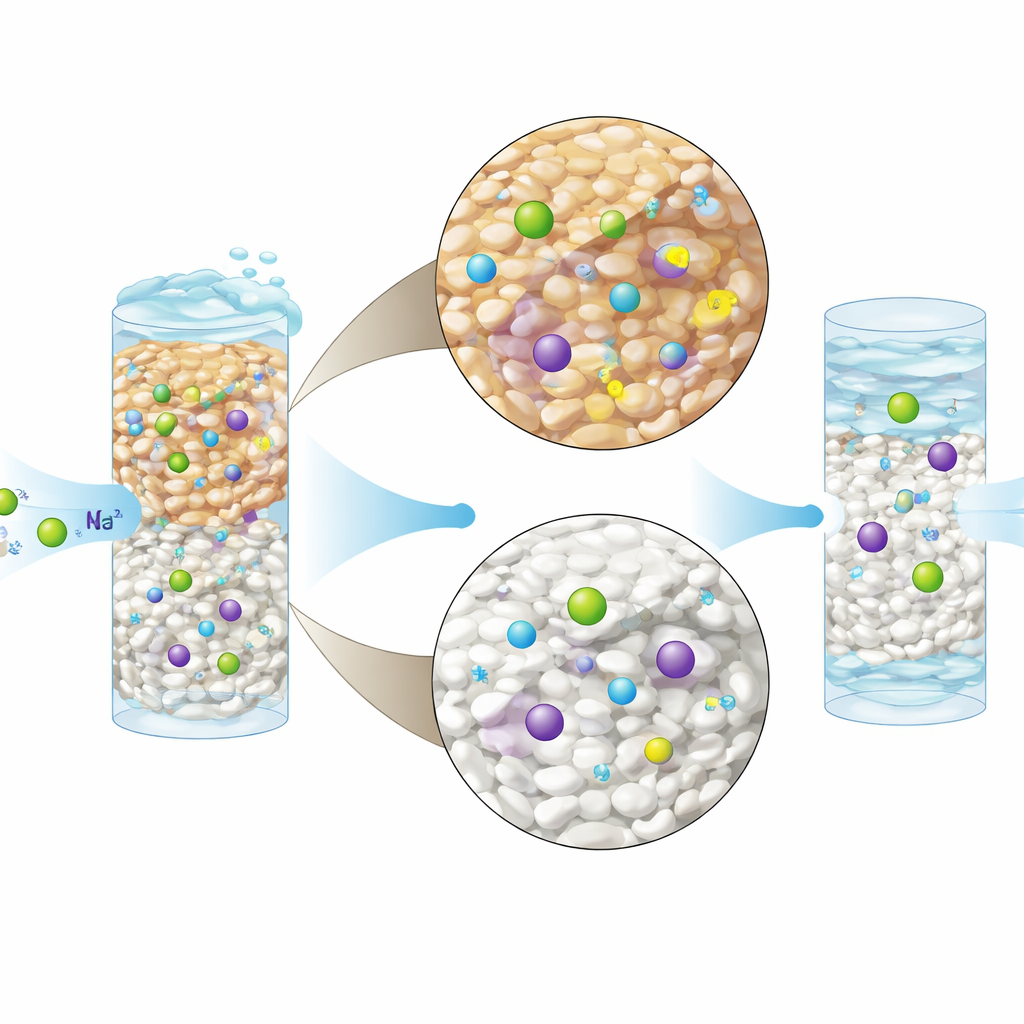
How Well the Columns Performed
In practice, the tuned zeolite stood out. In a wider column run at a slower flow—conditions that give water more time to interact with the mineral—the sodium-treated zeolite removed more than 99 percent of total hardness, as well as nearly all calcium and magnesium, over many hours before the bed became saturated. Even the unmodified zeolite performed strongly, while activated alumina and its modified form achieved slightly lower, but still impressive, removal levels in the mid-90s to high-90s percent range. The researchers also compared how the mineral beds behaved against standard mathematical descriptions of filtration systems. Two of these, known as the Thomas and Yoon–Nelson models, tracked the real-world behavior closely, giving engineers reliable tools to predict when a column will need to be regenerated or replaced.
Why This Matters for Real-World Water
Because zeolite is mined cheaply and activated alumina is relatively inexpensive, these materials cost far less than many commercial softening media. The gentle chemical treatments used to enhance them rely on simple, readily available compounds, keeping both material and operating costs low. The study suggests that properly designed fixed-bed columns filled with modified zeolite, and to a lesser extent modified alumina, could offer a high-performance yet affordable way to soften drinking water continuously—especially attractive for small cities, rural systems, or regions with limited budgets.
Big Picture for Everyday Users
For non-specialists, the message is straightforward: common mineral powders, packed into simple tubes and gently pre-treated, can strip almost all hardness from water for long periods, using modest equipment and little energy. By showing exactly how column size, flow rate, and surface treatment affect performance—and by confirming that the behavior can be predicted with well-tested formulas—this work moves ceramic-based softeners closer to practical use. In the future, such systems could help deliver scale-free pipes, longer-lasting appliances, and more comfortable water at a fraction of today’s softening costs.
Citation: Danesh, E., Abbasi, M., Noroozi, M. et al. Investigation of water softening using ceramic adsorbents in a continuous adsorption process. Sci Rep 16, 9057 (2026). https://doi.org/10.1038/s41598-026-38953-2
Keywords: water softening, hard water, zeolite, activated alumina, adsorption column