Clear Sky Science · en
Integrin αv contributes to the regulation of vascular smooth muscle cell stiffness
Why artery "springiness" matters
As we age or develop high blood pressure, our large arteries gradually lose their natural springiness and become stiffer. This change makes the heart work harder and increases the risk of heart attacks, strokes, and other cardiovascular problems. Scientists have long known that the material making up artery walls—proteins like elastin and collagen—affects stiffness. This study asks a more subtle question: can the muscle cells inside artery walls themselves become stiffer, and does a tiny family of surface receptors called integrin αv help keep those cells, and thus our arteries, more flexible?
The muscle cells that shape blood flow
Large arteries are not just passive pipes. Their middle layer is packed with vascular smooth muscle cells, which can contract and relax to fine‑tune blood flow and pressure. These cells are anchored to a surrounding scaffold known as the extracellular matrix. The researchers focused on one group of anchoring molecules, integrin αv, which spans the cell surface and physically links the inner skeleton of the cell to this outer scaffold. Earlier work suggested that integrin αv was involved in scarring and remodeling of blood vessels, but it was unclear whether it also controlled how stiff the muscle cells themselves become, especially under the stress of hormones such as angiotensin II that raise blood pressure and promote fibrosis.
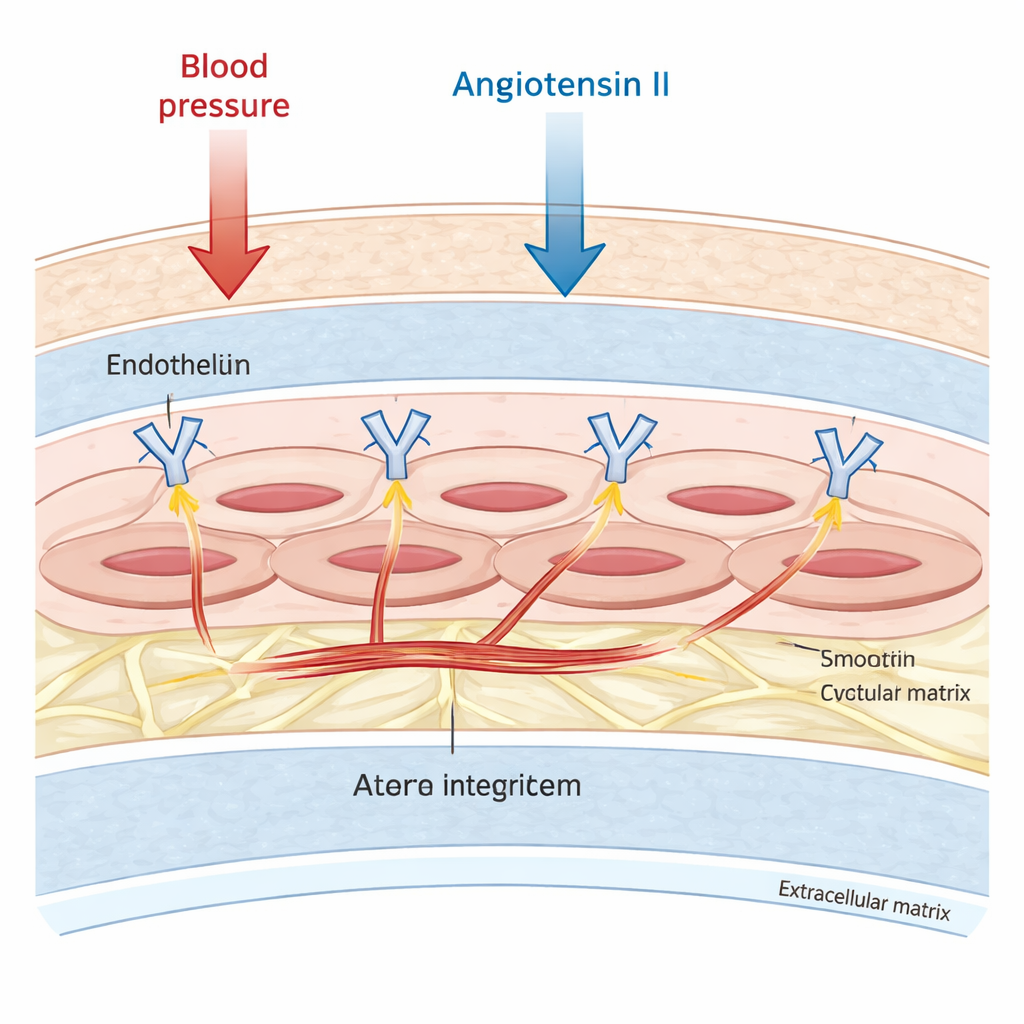
Measuring stiffness one cell at a time
To tackle this, the team used mouse smooth muscle cells grown in the lab and mice engineered to lack integrin αv specifically in their vascular muscle cells. They probed cell stiffness using atomic force microscopy, a technique in which an ultra‑fine tip gently indents the cell surface and records how much force is needed. Cells lacking integrin αv were more than twice as stiff as control cells under calm conditions. When exposed to angiotensin II for two days, these deficient cells became roughly three times stiffer again, while normal cells barely changed. Because their measurements used very shallow indentations, the authors built a detailed computer model of a smooth muscle cell and simulated deeper pokes. The simulations showed that shallow tests emphasize the contribution of the cell’s outer shell and cortical region, and still fell within the range seen in the stiff mutant cells, supporting the biological findings.
A reshaped inner skeleton
The next step was to look inside the cells. Using fluorescent dyes and microscopy, the researchers examined actin, a key filament that forms the cell’s internal scaffolding. Control muscle cells showed a relatively diffuse actin network, even after angiotensin II treatment. By contrast, cells missing integrin αv formed thick stress fibers crisscrossing the cell and, after hormone exposure, an intense band of actin just beneath the cell membrane, known as cortical actin. A quantitative measure of how much actin piled up near the edge of the cell confirmed that this cortical layer was strongly enriched only in the integrin‑deficient cells. These cells also developed unusually long adhesion structures where they grip the surrounding matrix, consistent with a transition toward more tension‑bearing, fibrillar adhesions that can lock the cell into a stiff state.
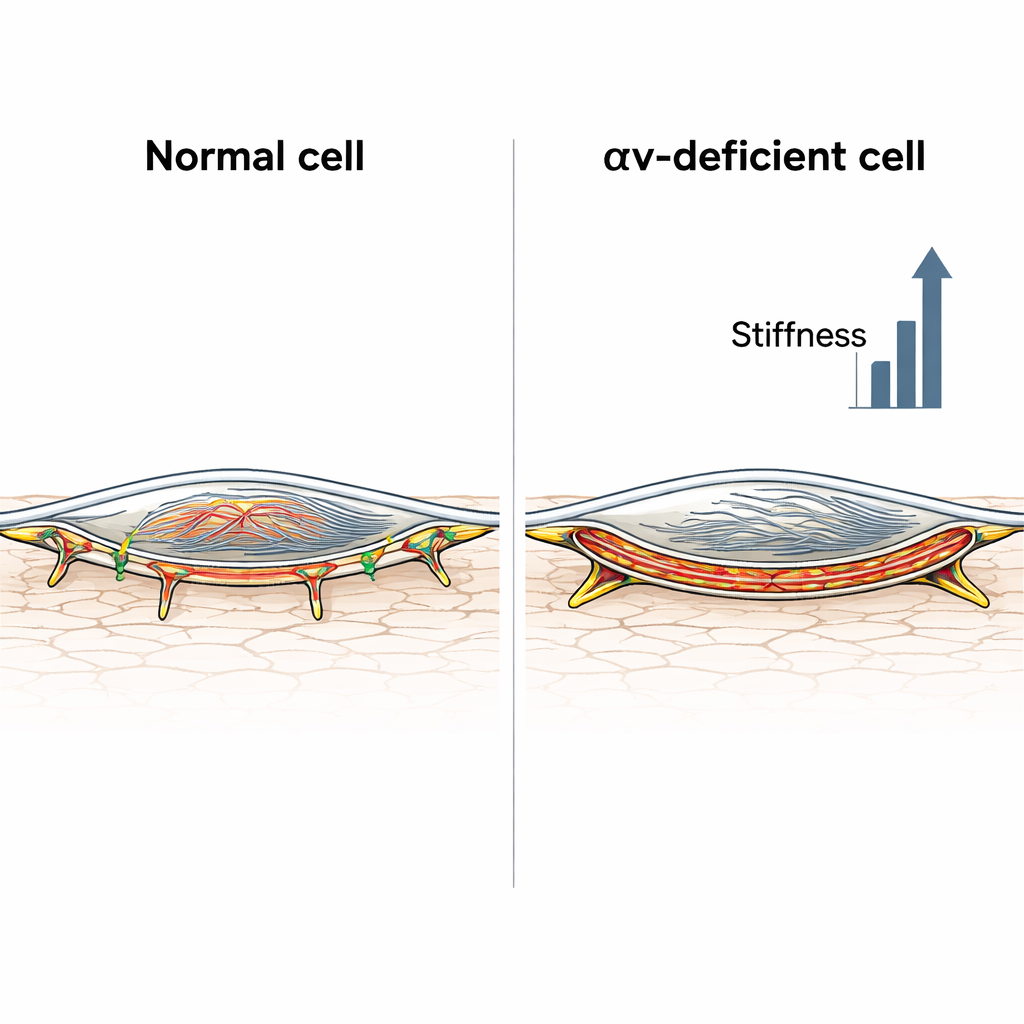
When the wall looks the same but behaves differently
Because arteries are more than just their muscle cells, the scientists also measured mechanical properties of carotid arteries in living mice, with and without integrin αv in their vascular muscle cells, and with or without chronic angiotensin II infusion. Surprisingly, overall arterial pressure and wall stiffness—gauged from ultrasound‑based pressure–diameter curves—were similar in both mouse strains, both at baseline and after hormone treatment. Yet the microscopic makeup of the wall differed. Under angiotensin II, control mice showed lower elastin and higher collagen, classic signs of a stiffer matrix, whereas integrin‑deficient mice had relatively less collagen change but much stiffer muscle cells. In other words, in normal mice the matrix did most of the stiffening; in integrin‑deficient mice, the muscle cells themselves became so rigid that they effectively canceled out their more modest matrix alterations.
What this means for aging arteries
For a layperson, the key message is that artery stiffness is not just about worn‑out elastic fibers; it also depends on how the muscle cells in the wall organize their tiny internal cables. Integrin αv normally helps keep these cells from becoming too rigid when challenged by hormones like angiotensin II. When this control is lost, the cells rearrange their actin skeleton—especially at the cortex just under the membrane—and lock into a stiffer state that can drive artery stiffening even without dramatic changes in the surrounding material. This insight points to new treatment ideas: drugs that gently loosen or reorganize the cortical actin network, or that modulate integrin‑linked signaling, might one day complement blood‑pressure‑lowering therapies to more directly restore the “spring” of aging arteries.
Citation: Bascetin, R., Belozertseva, E., Regnault, V. et al. Integrin αv contributes to the regulation of vascular smooth muscle cell stiffness. Sci Rep 16, 7682 (2026). https://doi.org/10.1038/s41598-026-38948-z
Keywords: arterial stiffness, vascular smooth muscle cells, integrin alpha v, actin cytoskeleton, angiotensin II