Clear Sky Science · en
Autofluorescence and Fourier transform infrared analyses trace dietary fluorophores and reveal plastic contamination in the gut of mosquito larvae
Why this mosquito study matters
Mosquitoes are notorious for spreading diseases, but before they ever bite, they spend days as tiny larvae feeding in water-filled containers. These early stages are the workhorses of mosquito growth and a prime target for control strategies. This study shows how scientists can “see” what larvae are eating—without adding any dyes—and even detect traces of plastic that quietly leach from common lab containers into their guts. The findings are important both for designing safer mosquito-control methods and for understanding how microplastic pollution may move through small aquatic animals.
Glowing clues inside tiny bodies
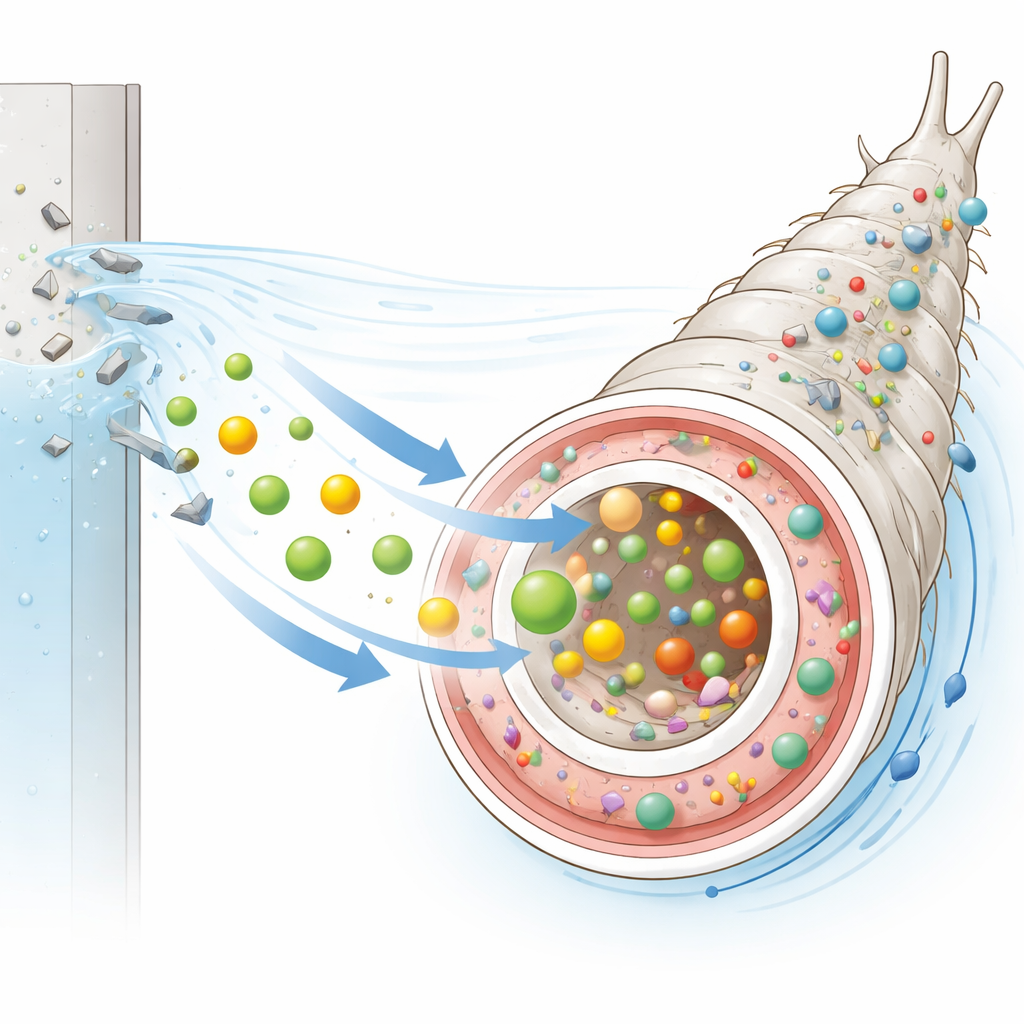
Many natural molecules glow faintly when illuminated with certain colors of light, a property called autofluorescence. The researchers took advantage of this built-in glow to track food in larvae of the Asian tiger mosquito, a major disease vector. Using powerful microscopes and spectral imaging, they looked at commercial larval food, the larval gut, and thin tissue slices. Food particles showed two main glowing signals: a broad bluish band from protein-rich material and a sharp red band from chlorophyll-related pigments derived from plants and algae. When larvae ate this food, their gut contents displayed the same signatures, confirming that these light signals could serve as natural markers of what they had ingested.
Food pigments escape the gut
By zooming in with confocal microscopy, the team found something more surprising: the red chlorophyll-like signal was not confined to the gut lumen. It also appeared in the surrounding body cavity, but not in the outer cuticle. This pattern suggests that some food-derived pigments survive digestion and move into internal body fluids, potentially accumulating in other tissues. The same red emission was also detected in the water where larvae had been reared, even though it was absent from water containing food alone. This indicates that larvae take up chlorophyll-related compounds and later release some back into their environment, offering a way to follow how naturally fluorescent dietary components circulate through the animal and its habitat.
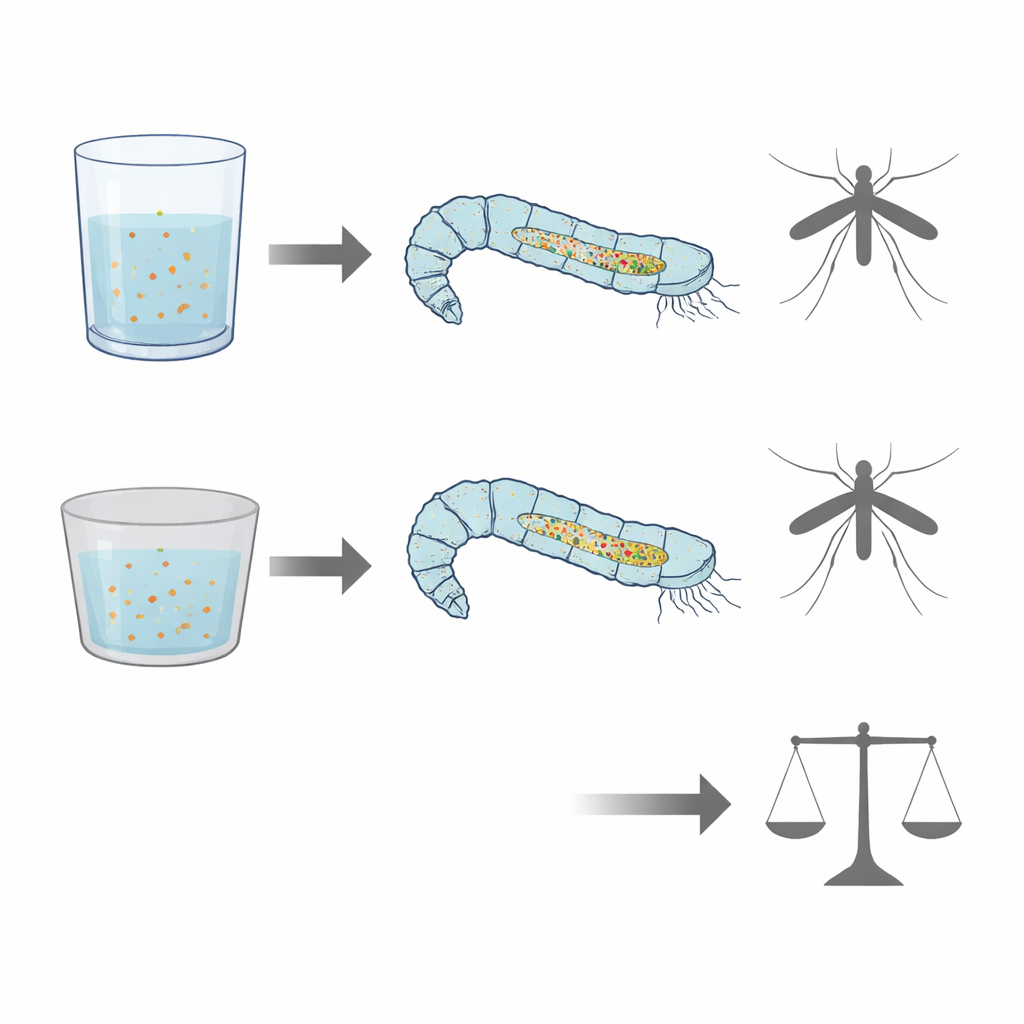
Container material changes what larvae take in
To test how the rearing environment might influence feeding, larvae were grown either in glass dishes or in polystyrene plastic dishes under identical conditions. The overall glow from food in the gut was consistently stronger and more abundant in larvae from plastic containers than in those from glass, hinting at differences in how much or how efficiently they fed or processed their diet. Spectrofluorimetric measurements of the surrounding water showed changes in another group of glowing compounds, flavins, which are linked to energy metabolism and B vitamins. These shifts suggest that larvae raised in different container materials may handle certain nutrients differently, even when their food is the same.
Hidden plastic fingerprints in the gut
Beyond light-based imaging, the scientists used a technique called ATR-FTIR spectroscopy to read the chemical “fingerprints” of larval guts and rearing materials. Guts from larvae reared in plastic dishes showed a distinct signal in the infrared region that closely matched a key feature of polystyrene and of larvae experimentally exposed to polystyrene beads. This peak was absent from guts of larvae reared in glass and from the food itself, strongly pointing to the presence of plastic-derived material in larvae raised in plastic containers. Although electron microscopy did not reveal obvious tooth marks or scraping damage on the inside surface of the dishes, prior work has shown that plastic can shed microscopic and even nanoscopic fragments during normal use, which can be taken up by aquatic organisms.
What all this means for mosquito control and pollution
Despite these subtle chemical and optical differences, standard measures such as larval survival, development time, and adult body size changed little between glass- and plastic-reared groups, apart from a modest shift in larval development duration. To a casual observer, the mosquitoes would appear nearly identical. Yet their guts tell a more complex story: they differ in how much food-linked glow they carry, how they process certain vitamins and plant pigments, and whether fragments of plastic have entered their tissues. For vector-control programs that rely on mass-rearing mosquitoes—especially those using the sterile insect technique—such hidden effects could matter for long-term performance and health. More broadly, the work demonstrates that natural fluorescence and infrared spectroscopy can act as sensitive, non-destructive tools to monitor diet, rearing conditions, and plastic contamination in small aquatic animals. These methods could help refine greener larval control strategies and deepen our understanding of how microplastics quietly move through freshwater ecosystems.
Citation: Soldano, S., Weththimuni, M.L., Oldani, A. et al. Autofluorescence and Fourier transform infrared analyses trace dietary fluorophores and reveal plastic contamination in the gut of mosquito larvae. Sci Rep 16, 7841 (2026). https://doi.org/10.1038/s41598-026-38938-1
Keywords: mosquito larvae, autofluorescence, microplastics, chlorophyll pigments, vector control