Clear Sky Science · en
Crumpled Ti₃C₂Tₓ MXene electrodes with tunable surface chemistry for high-performance and selective electrochemical biosensing
Smarter Sensors for Everyday Chemistry
From brain health to pain relief, many key molecules in our bodies—like vitamin C, dopamine, uric acid, and acetaminophen—circulate at tiny concentrations in blood, sweat, and other fluids. Doctors and food scientists need fast, reliable ways to measure these chemicals, even when they are mixed together with countless others. This study shows how a new kind of wrinkled, sheet-like material called a MXene can be shaped and chemically tuned to build small, highly sensitive electrodes that pick out these molecules quickly and accurately, paving the way for better diagnostic gadgets and quality checks for food and the environment.
Why These Tiny Molecules Matter
The researchers focused on four familiar compounds: ascorbic acid (vitamin C), dopamine, uric acid, and acetaminophen. Abnormal levels of these substances are linked to conditions such as neurological disorders, oxidative stress, gout, and liver strain from over-the-counter painkillers. Standard lab tests to measure them can be slow, expensive, or require complex sample preparation. Electrochemical sensors—small electrodes that turn chemical reactions into electrical signals—offer a faster, cheaper alternative. The challenge is to design an electrode surface that is sensitive enough to detect low levels, selective enough to tell similar molecules apart, and robust enough to work inside real biological and food samples full of interfering substances.
Wrinkled Metal Sheets as a New Sensing Surface
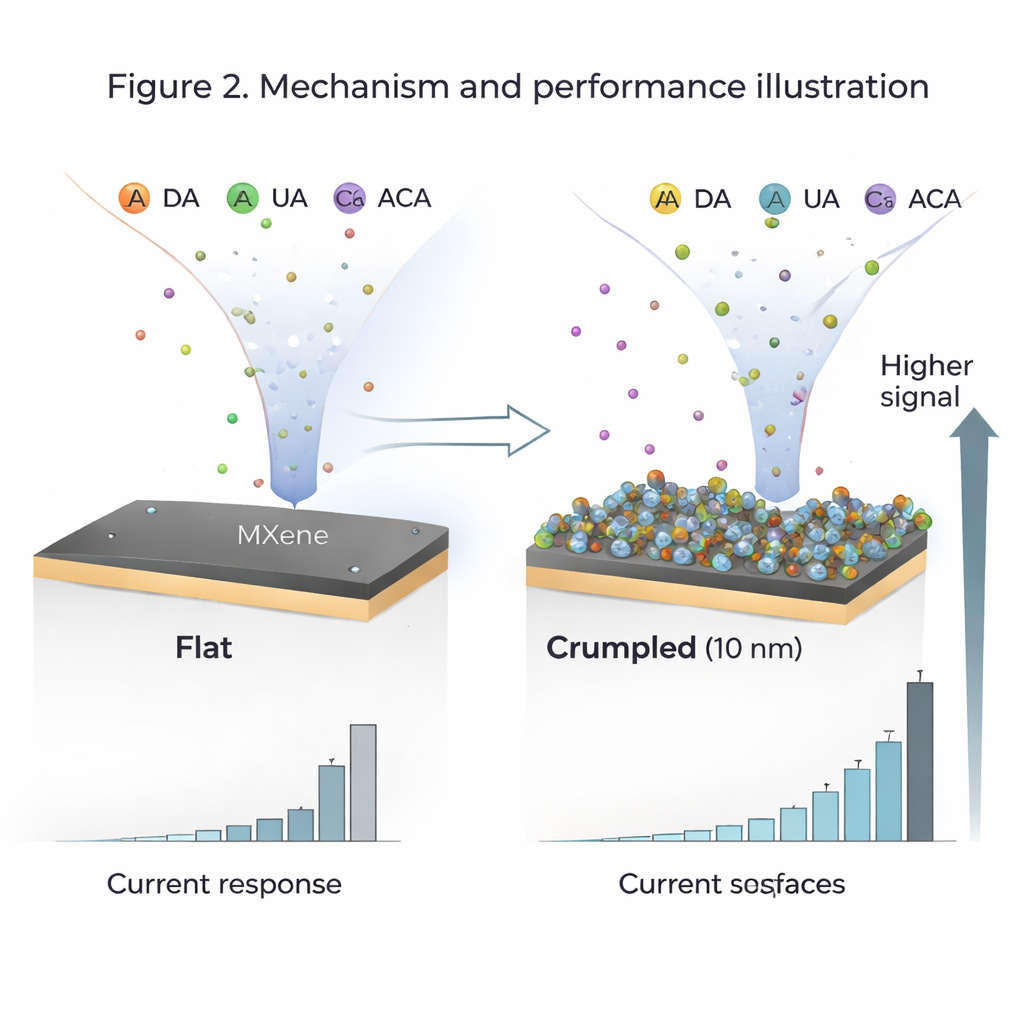
To tackle this, the team used a two-dimensional material called Ti3C2Tx MXene, a stack of atomically thin, electrically conductive layers decorated with chemical groups such as oxygen and hydroxyl (–O and –OH). They coated a standard glassy carbon electrode with MXene and then deliberately crumpled the MXene layer into tiny folds only a few billionths of a meter high. By controlling how much the sheets crumpled (the "amplitude" of the wrinkles) and how thick the MXene layer was, they could tune how much surface area was exposed and how easily molecules could reach and stick to it. They found that a gently crumpled surface with folds about 10 nanometers high and a thin 10-nanometer layer produced the best performance.
How the Sensor Performs in Action
In tests, the crumpled MXene electrode generated strong, clear electrical signals when each target molecule was present at realistic concentrations between 10 and 200 micromoles. Sensitivity—how much current change corresponds to a change in concentration—fell between about 0.77 and 0.82 microamps per micromole, with detection limits below 1 micromole for all four analytes. The wrinkled surface exposes a large area (around 150 square meters per gram of MXene) and many –O/–OH groups that attract the molecules via hydrogen bonding and stacking interactions between carbon rings. Even when all four molecules were present together, the signals only dropped by about 5–8 percent because they had to compete for the same surface sites, meaning the sensor can still distinguish each one in a mixture.
Confirming the Mechanism with Computer Models
To understand what was happening beyond the lab bench, the authors built detailed computer models using COMSOL Multiphysics. They simulated how the molecules diffuse through solution, adsorb on the crumpled surface, and exchange electrons with the electrode. The model predicted diffusion coefficients, response times of roughly 1.5–2.5 seconds, and current levels that matched the experiments closely. By comparing different wrinkle sizes and layer thicknesses in the simulations, they showed why moderate crumpling and thin layers give the best balance: more active sites for binding, shorter paths for molecules to travel, and less crowding in deep folds. The modeling also confirmed that the MXene surface binds the target molecules much more strongly than common interferents such as glucose and citric acid, explaining why the presence of these background species changed the current by less than about 2.5 percent.
What This Means for Real-World Testing
In practical terms, this work demonstrates that carefully wrinkling and chemically tuning MXene films turns simple carbon electrodes into powerful, selective detectors for biologically important molecules. Because these sensors are fast, sensitive at low concentrations, and resilient to common interferents, they could be integrated into portable devices for monitoring health markers, checking the nutritional or spoilage status of foods, or tracking pollutants. The key message is that how a material is shaped at the nanoscale, together with the chemical groups on its surface, can be engineered in tandem to boost performance—offering a roadmap for building the next generation of small, smart electrochemical biosensors.
Citation: Aburub, F., Abdullah, Q., Mohammad, S.I. et al. Crumpled Ti₃C₂Tₓ MXene electrodes with tunable surface chemistry for high-performance and selective electrochemical biosensing. Sci Rep 16, 7663 (2026). https://doi.org/10.1038/s41598-026-38937-2
Keywords: electrochemical biosensor, MXene, dopamine detection, nanostructured electrodes, surface chemistry