Clear Sky Science · en
Gut microbiota and metabolic pathway profiles in infected and non-infected heart transplant patients before and after surgery
Why your gut matters after a new heart
Heart transplantation can give people with severe heart failure a second chance at life, but infections in the weeks after surgery remain a major threat. This study asks a deceptively simple question with big implications: how does the community of microbes living in our intestines—the gut microbiota—change around the time of heart transplant, and can those changes help explain who gets infections and who does not?
The tiny ecosystem inside transplant patients
Our intestines host trillions of bacteria that help digest food, train the immune system and keep invading germs at bay. Surgery, strong immune-suppressing drugs and broad-spectrum antibiotics can all jolt this fragile ecosystem. To track what happens in heart transplant patients, researchers in China followed 20 adults receiving donor hearts. They collected stool samples about a week before surgery and again 30 days afterward, then used DNA sequencing to catalogue which bacteria were present and computer tools to predict what those microbes might be doing metabolically.
Four patient groups, four microbial patterns
The team divided patients into four groups: those with and without infections before surgery, and those with and without infections in the first month after surgery. Before transplantation, the overall diversity and structure of the gut microbiota looked surprisingly similar whether or not patients already had an infection. People without pre-surgery infections were mostly colonized by a familiar genus called Bacteroides, often linked to stable gut health. In contrast, infected patients showed a mix that included potentially harmful Enterococcus casseliflavus alongside bacteria often considered friendly, such as Limosilactobacillus and Weissella cibaria, suggesting a gut community in a delicate, shifting balance.
After surgery: a fork in the microbial road
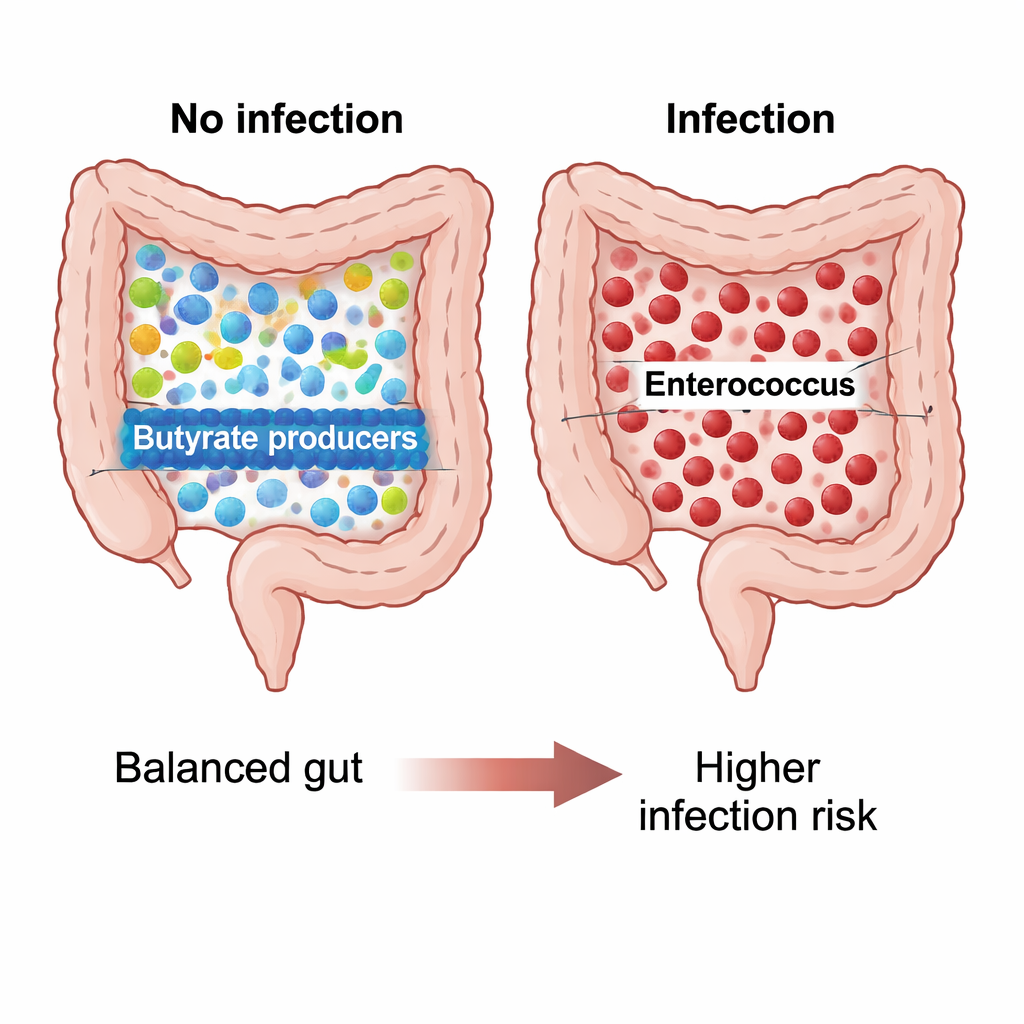
By 30 days after the operation, the microbiota of patients who avoided infection formed one recognizable pattern, while those who developed infections looked very different. Non-infected patients had richer, more even microbial communities, with many members of a genus called Blautia and related bacteria that produce butyrate, a short-chain fatty acid known to nourish gut cells and support immune stability. Their gut ecosystems looked diverse and balanced. In patients who suffered infections, however, diversity dropped and the community was dominated by Enterococcus faecium, a hardy bacterium often found in hospital-acquired infections and known for antibiotic resistance. Statistical analyses showed that this infected group’s gut microbes clustered apart from all the others, pointing to a clear case of post-surgical microbial disruption, or dysbiosis.
Microbial chemistry linked to protection or risk
The researchers also asked what kinds of chemical jobs these microbes might be doing. Using predictive software, they found that in the non-infected, post-surgery group, Blautia was tied to pathways that break down a compound called L-1,2-propanediol, an intermediate in fiber fermentation that ultimately feeds the production of beneficial fatty acids such as propionate. In infected patients, this predicted metabolic potential was markedly lower, consistent with a gut community less able to make protective molecules. Before surgery, Enterococcus casseliflavus in infected patients was linked to unusual fermentation routes that may reflect a stressed, energy-hungry microbial environment, though this needs direct laboratory confirmation.
What this means for patients and care teams
For people facing a heart transplant, these findings suggest that the gut is not just a bystander; it may help shape the risk of dangerous infections. The study points to a dynamic shift from Bacteroides dominance before surgery to either protective Blautia enrichment or harmful Enterococcus overgrowth afterward, depending on how the early recovery period unfolds. Heavy infection-related stress and antibiotic use appear to push the gut community toward the Enterococcus-dominated, low-diversity state seen in infected patients. The authors propose a "dual-target" strategy: support the return of butyrate-producing bacteria—perhaps through diet or carefully chosen probiotics—while closely monitoring Enterococcus levels. Though larger and longer studies are needed, this work opens the door to future transplant care in which tuning the gut microbiota becomes a routine part of protecting patients from life-threatening infections.
Citation: Han, J., Hua, L., Yang, B. et al. Gut microbiota and metabolic pathway profiles in infected and non-infected heart transplant patients before and after surgery. Sci Rep 16, 7697 (2026). https://doi.org/10.1038/s41598-026-38911-y
Keywords: heart transplantation, gut microbiota, postoperative infection, antibiotics, butyrate-producing bacteria