Clear Sky Science · en
Enhanced kinetic performance and stability of catalase immobilized on epoxy-functionalized kaolinite
Turning a Natural Clay into a Smart Cleaner
Hydrogen peroxide is widely used to disinfect, bleach, and treat water, but leftover peroxide in industrial waste streams can damage living cells and the environment. Nature already provides a powerful clean-up tool: the enzyme catalase, which breaks hydrogen peroxide into harmless water and oxygen. However, free enzymes are fragile and hard to reuse. This study shows how a common clay mineral, kaolinite, can be gently modified so that catalase sticks strongly to it, becoming a tougher, reusable "cleanup surface" that could help make industrial processes and wastewater treatment safer and more sustainable. 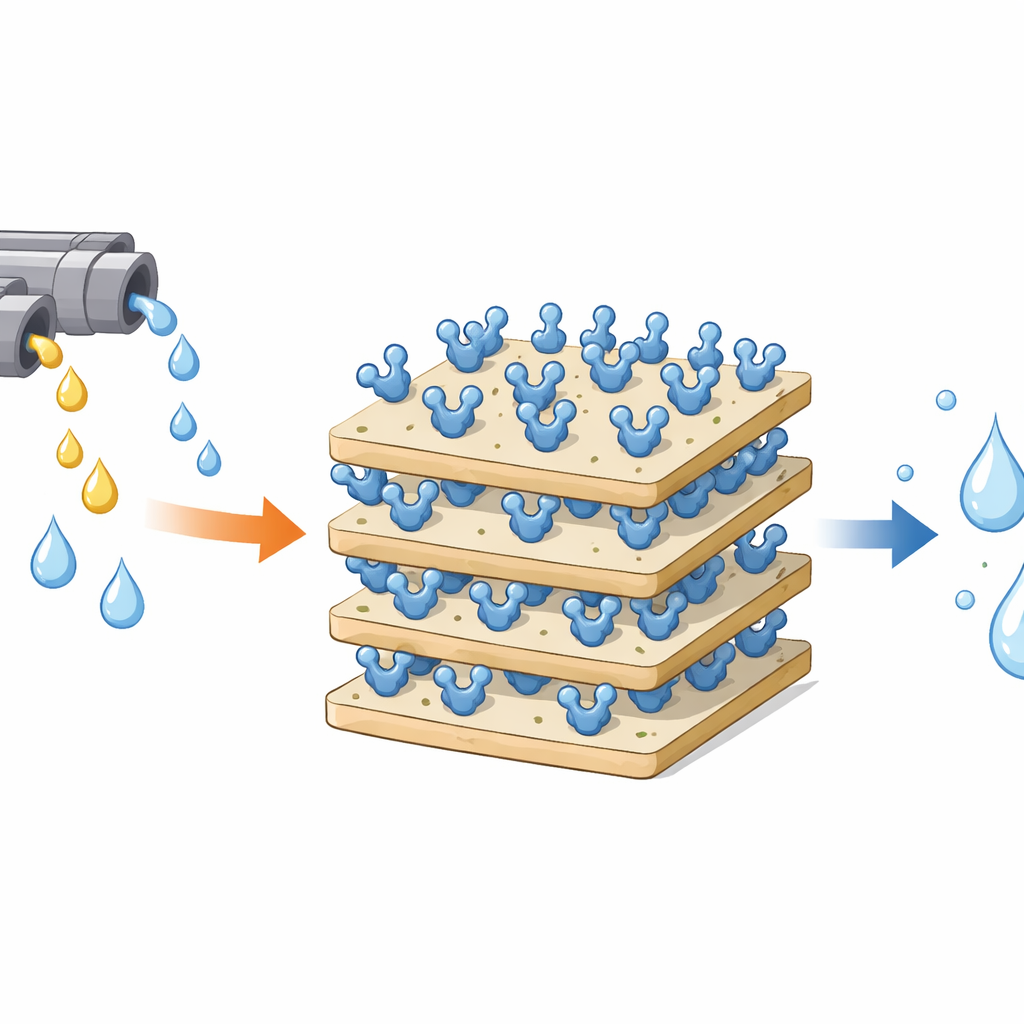
A Common Mineral with Hidden Potential
Kaolinite is a cheap, widely available white clay used in paper, ceramics, and even medicine. It has a layered structure that is mechanically strong and chemically stable, but its surface is relatively inert, so enzymes do not grip it well and tend to wash away. Earlier attempts to hold catalase on plain kaolinite mostly relied on weak attraction forces. These systems could pick up some enzyme but suffered from low loading, easy leaching, and loss of activity over time. The authors reasoned that if they could introduce more reactive sites on kaolinite without destroying its structure, the clay could act as a robust, long-lasting platform for catalase.
Adding a Sticky yet Gentle Coating
To upgrade kaolinite, the researchers coated its surface with a small molecule called GPTMS, a type of silane that carries an epoxy group. In water-alcohol mixtures under mildly acidic conditions, GPTMS first converts into silanol groups that can bind to the clay’s natural hydroxyls, forming a thin, durable organic layer. The epoxy parts remain intact and protrude from the surface as tiny reactive rings. A suite of techniques confirmed this transformation: infrared spectra showed new carbon–hydrogen and epoxy-related vibrations; electron microscope images revealed that the originally compact plate-like clay became more open and porous; thermal analysis detected extra mass loss at moderate temperatures due to the newly attached organic layer; and surface spectroscopy showed much more carbon on the modified clay while preserving the underlying mineral framework.
Docking the Enzyme on the Clay
When catalase was mixed with this epoxy-bearing kaolinite, the enzyme quickly attached to the surface. Within the first hour, most of the binding sites were already occupied, and equilibrium was essentially reached in four hours. The modified clay could hold about 300 milligrams of catalase per gram of support—far higher than previous kaolinite-based systems. The team found that neutral pH and moderate temperatures (around room to body temperature) were best for loading, reflecting a balance between enzyme stability and the reactivity of amino groups on the protein with the epoxy rings on the surface. On the molecular level, nucleophilic groups on catalase attack the strained epoxy rings, forming multiple strong bonds. This multipoint attachment limits harmful motion while keeping the active center accessible. 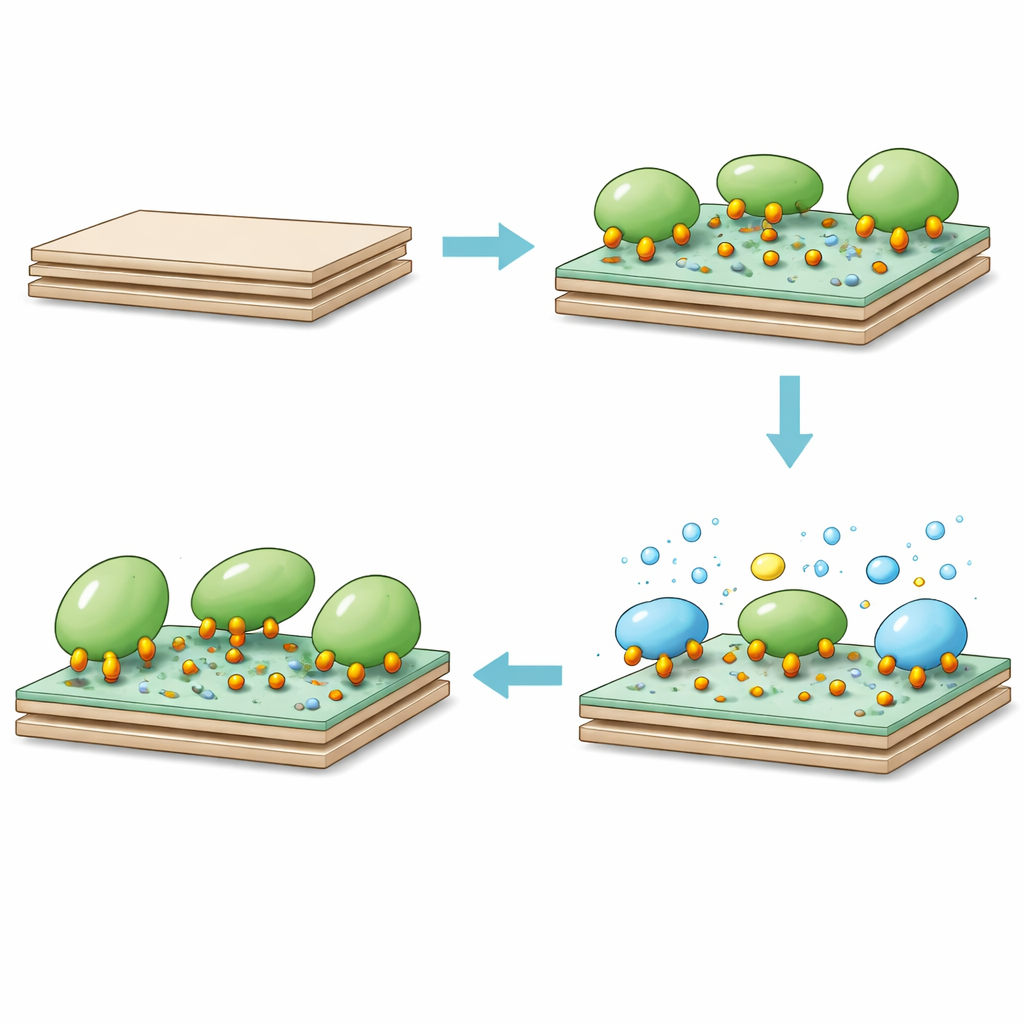
Faster, Tougher, and Longer-Lasting Action
Attaching catalase to the modified clay changed how it behaved toward hydrogen peroxide. The immobilized enzyme showed a much lower apparent "demand" for substrate than the free enzyme, meaning it could work efficiently even when peroxide levels were modest. Although the maximum reaction rate dropped somewhat—likely because diffusion through the solid layer and reduced enzyme flexibility slow things down—the overall catalytic efficiency actually rose by about 80 percent. Just as important, the immobilized catalase withstood repeated use and long storage far better than the free form. It retained high activity after many reaction cycles and kept a much larger fraction of its initial power after weeks in cold storage. The clay support itself could also be regenerated and reused multiple times for fresh enzyme loading with only gradual loss of capacity.
Why This Matters for Everyday Life
In simple terms, the study turns a familiar, low-cost clay into a smart, reusable holder for a natural detoxifying enzyme. By carefully engineering the clay’s surface with a thin epoxy-rich layer, the researchers created a platform that grabs catalase firmly, helps it recognize its target more easily, and protects it from damage. This means we can potentially clean peroxide-laden industrial streams, support advanced oxidation processes, or design safer food and pharmaceutical treatments using smaller amounts of enzyme over longer periods. The work shows how tuning the interface between minerals and proteins can unlock new, durable tools for greener technologies.
Citation: Erol, K., Veyisoğlu, A., Tatar, D. et al. Enhanced kinetic performance and stability of catalase immobilized on epoxy-functionalized kaolinite. Sci Rep 16, 8196 (2026). https://doi.org/10.1038/s41598-026-38910-z
Keywords: enzyme immobilization, catalase, kaolinite clay, wastewater treatment, epoxy-functional surface