Clear Sky Science · en
G-quadruplexes self-assembled from nucleotide monomers as stable prepolymer scaffolds in aqueous environments
A New Clue to How Life Got Started
How did lifeless chemistry on the early Earth turn into the first living systems made of long genetic molecules like RNA and DNA? This study explores a simple but powerful idea: that one of RNA’s building blocks can automatically line up and concentrate itself into orderly structures in water, creating a kind of molecular scaffolding that could have helped the first genetic polymers form without enzymes or modern biology.
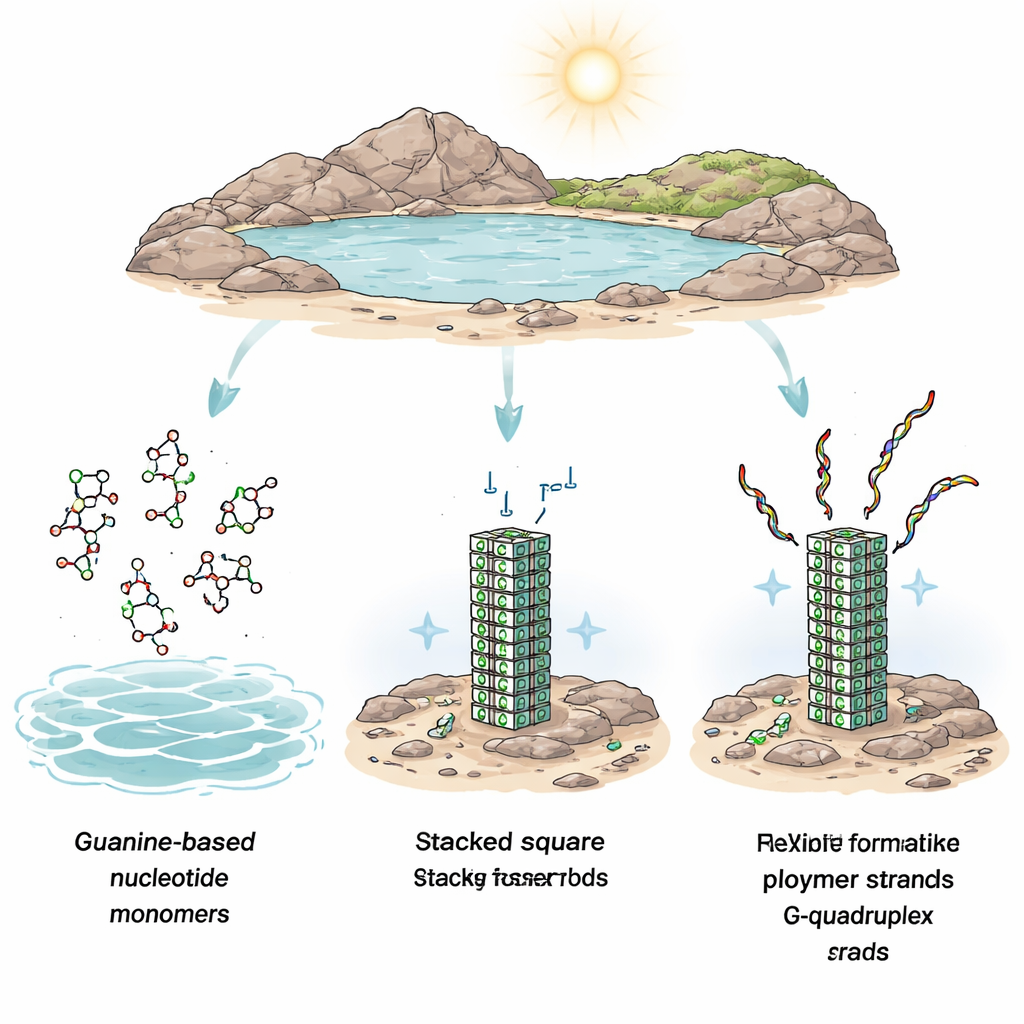
Building Blocks Looking for Order
Before life began, Earth’s ponds and pools were likely full of many different small organic molecules. For life to emerge, a few special ones—like nucleotides, the building blocks of RNA and DNA—had to be selected, brought together, and linked into long chains. That is harder than it sounds. Nucleotides are usually dilute in water and surrounded by countless unrelated molecules. On top of that, linking them into chains is energetically uphill and does not happen easily in ordinary water. Previous ideas suggested that repeated drying and re-wetting, or natural energy sources like heat and sunlight, could help push the chemistry forward, but they did not explain how specific building blocks could be picked out from the crowd.
Self-Organizing Stacks of One Special Nucleotide
The authors focus on one nucleotide in particular: a guanine-based unit called GMP. Guanine has a striking tendency to self-organize. When many guanine units are present together, they can form flat square groups of four, which then stack into long columns known as G-quadruplexes. Using high-resolution atomic force microscopy (AFM), the researchers dried solutions of GMP on a smooth mineral surface called mica and then imaged what formed in water. They saw long, wire-like filaments—G-quadruplexes—extending tens to hundreds of nanometers, even though the overall amount of GMP in solution was extremely low. These filaments were stable for hours in a salt solution containing potassium ions, and their repeating height pattern matched what is expected from a stack of guanine layers. In other words, without any enzymes or added catalysts, identical nucleotide units had found and organized each other into highly regular, concentrated structures.
Testing Stability with Different Salty Conditions
To probe how robust these self-assembled filaments were, the team changed the salt in the surrounding water. Potassium ions are known to favor G-quadruplex structures, while nickel ions interact more strongly and can disturb them. When the imaging solution was switched from potassium to nickel, many of the long filaments broke into shorter fragments or disappeared from the surface. This behavior shows that the filaments are held together mainly by non-permanent interactions—hydrogen bonds and stacking—rather than strong covalent bonds. The breakup pattern also confirms that the structures are indeed made from assembled GMP units instead of pre-formed contamination. A few segments survived longer, hinting that some arrangements may be especially stable and could have been favored over time in natural settings.
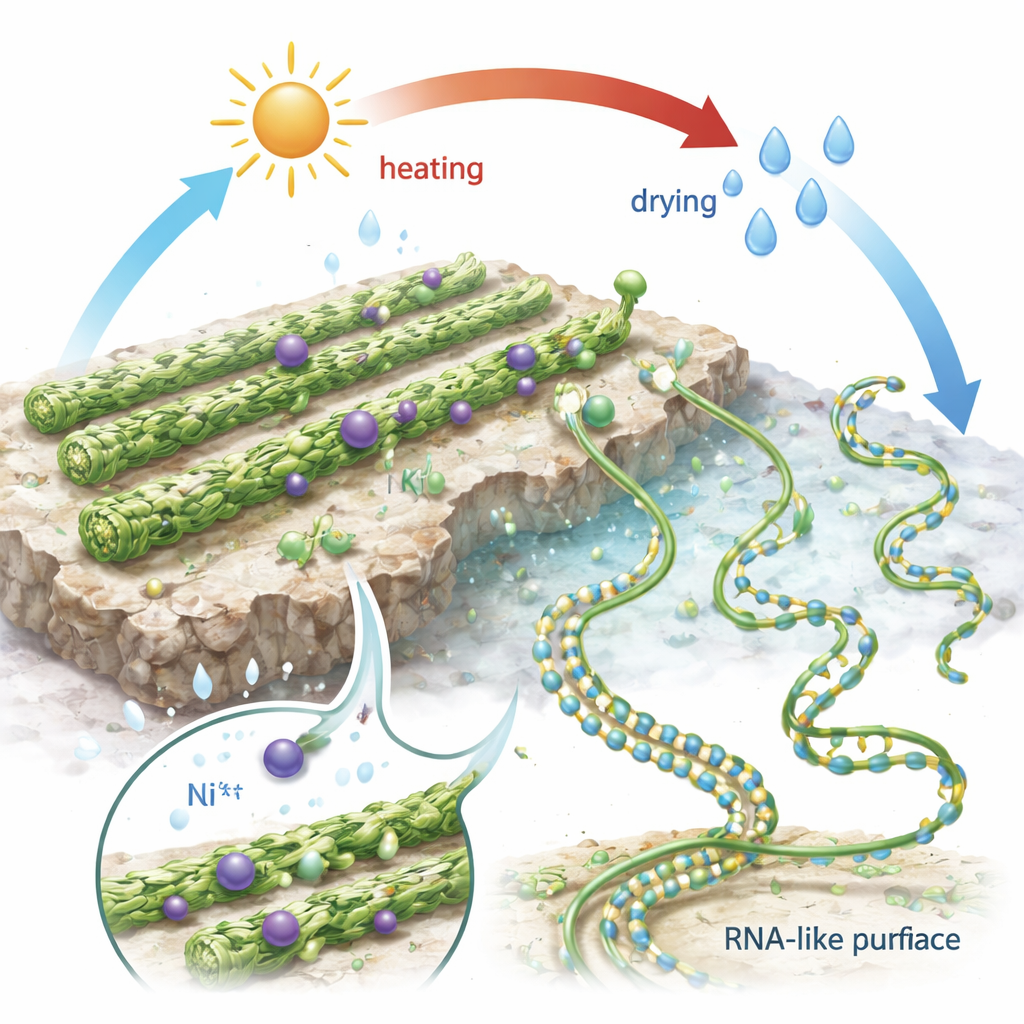
From Ordered Scaffolds to RNA-Like Chains
The crucial step toward life, however, is not just self-assembly but the creation of real polymers—chains where building blocks are joined by covalent bonds. To mimic conditions in early hot spring pools, the researchers subjected the GMP-coated surfaces to repeated cycles of heating to 80 °C and drying, followed by re-wetting. After three such cycles, AFM images revealed not only G-quadruplex filaments but also many much thinner, coiled strands spread across the surface. These new strands were often attached like tails to the thicker filaments, suggesting they grew from or were derived from them. Their height, length, and coiled appearance closely resembled known single-stranded RNA molecules. Unlike loosely held aggregates, these thin strands stayed stuck to the negatively charged surface even in solutions where simple GMP assemblies should fall apart, implying that their building blocks are now joined by covalent bonds. When the salt environment was again switched to one containing nickel, the thin strands did not vanish but instead folded into more compact, beaded shapes, just as real single-stranded RNA is known to do in the presence of certain metal ions.
What This Means for Life’s Beginnings
These experiments suggest a simple, physically driven path from scattered nucleotide building blocks to concentrated, structured assemblies, and then to RNA-like chains. Guanine-based units spontaneously form long G-quadruplex filaments that act as stable pre-polymer scaffolds on mineral surfaces in water, even at very low concentrations. Under cycles of heating and drying—conditions plausible in early hot spring pools—these scaffolds can partially transform into flexible, RNA-like polymers that remain stable in solution and behave much like genuine single-stranded RNA. Although the precise chemical linkages in these products are not yet fully identified, the work supports the idea that self-organized guanine assemblies could have provided both a selection step and a staging ground for the first genetic polymers, helping to bridge the gap between a messy prebiotic soup and the ordered molecules needed for life.
Citation: Eiby, S.H.J., Catley, T.E., Gamill, M.C. et al. G-quadruplexes self-assembled from nucleotide monomers as stable prepolymer scaffolds in aqueous environments. Sci Rep 16, 7644 (2026). https://doi.org/10.1038/s41598-026-38899-5
Keywords: origin of life, RNA world, G-quadruplex, prebiotic chemistry, nucleotides