Clear Sky Science · en
Computational study of carbon-doped TiO2(B) nanomaterials for improved dye-sensitized solar cells
Turning More Sunlight into Power
Solar panels that use colorful dyes instead of thick silicon slices promise flexible, low-cost power even in dim or indoor light. But their performance hinges on an invisible handshake: how tightly the dye molecules grip the surface of a transparent semiconductor and how easily electrons can jump across that boundary. This study uses advanced computer simulations to explore a new way to strengthen that handshake by tweaking a particular form of titanium dioxide with tiny amounts of carbon, with the goal of making dye‑sensitized solar cells more efficient and longer lasting.
Why This Special Solar Cell Matters
Dye-sensitized solar cells work a bit like artificial leaves. A thin layer of titanium dioxide acts as a scaffold coated with a light-absorbing dye. When sunlight hits the dye, electrons are excited and must quickly move into the titanium dioxide and then through the rest of the device to produce usable power. One of the most successful dyes, known as N719, has already helped these cells reach efficiencies of about 15%, but there is still plenty of room for improvement. A key challenge is to make the dye stick more firmly and in the right way to the surface so that electrons can travel smoothly and avoid recombining or getting lost.
A New Twist on Titanium Dioxide
The semiconductor studied here is a less familiar form of titanium dioxide called the bronze phase, or TiO2(B), which has shown promise in both solar cells and batteries. The researchers focused on an ultrathin sheet of this material and studied how a single N719 dye molecule attaches to one of its most reactive surfaces. Using quantum mechanical calculations, they tested several ways the dye could anchor itself through its carboxyl groups—chemical "hooks" that can latch onto titanium atoms in different patterns. They found seven stable arrangements, with the most favorable one using three of the dye’s four hooks at once, giving a particularly strong and compact attachment to the surface.
Making the Surface More Welcoming
To further improve the interface, the team explored what happens when some surface oxygen atoms in TiO2(B) are replaced by carbon—a strategy known as surface doping. Their simulations show that this subtle modification dramatically strengthens the attraction between the dye and the surface, increasing the adsorption energy by up to about 300% compared with the undoped material. In practical terms, the dye sits closer and more securely on the carbon-doped surface, allowing denser coverage. At the same time, the electronic structure of the material changes: new hybrid states appear at the boundary between dye and semiconductor, and the effective energy gap of the system narrows, which can help electrons move more easily under visible light.
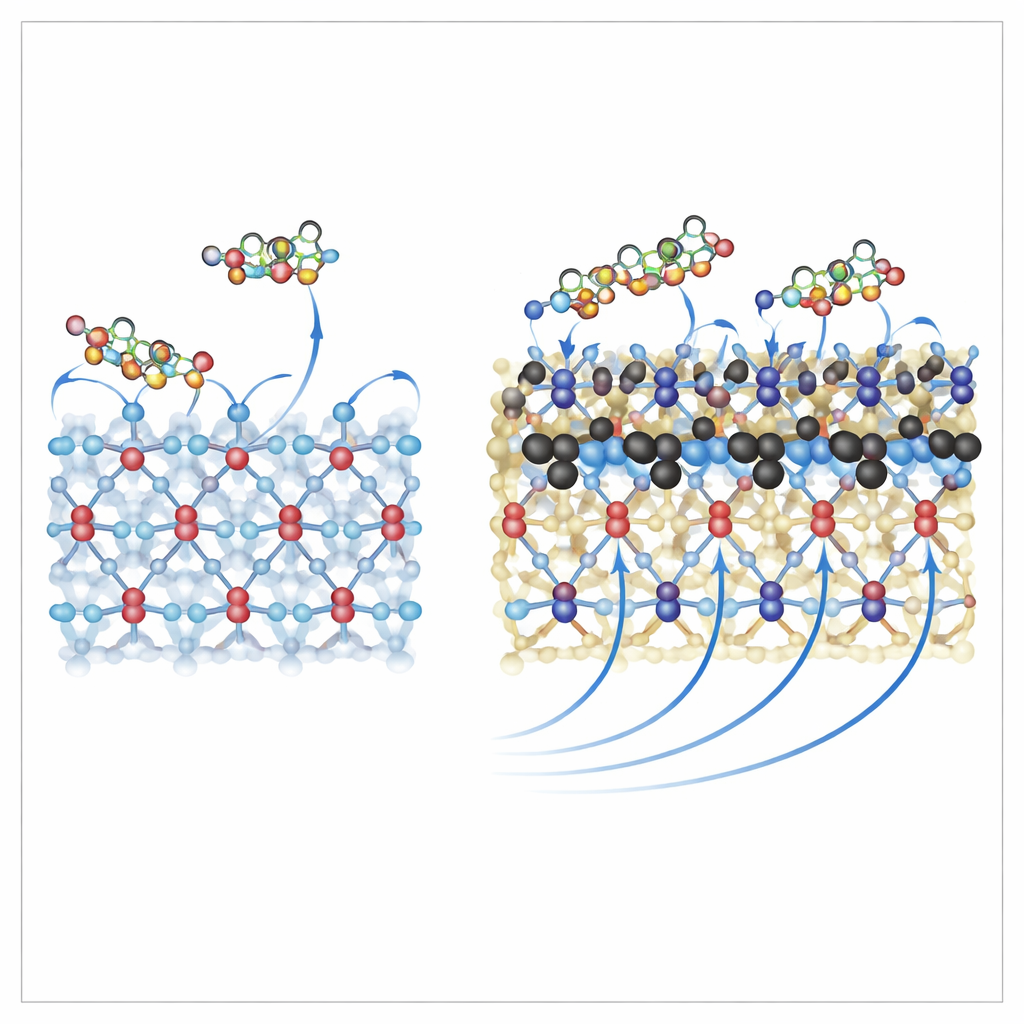
Helping Electrons Find the Fast Lane
The study also connects these atomic-scale changes to solar-cell performance. Carbon at the surface tends to lower the work function of TiO2(B), effectively raising the energy level from which electrons can be injected. The new states created by carbon act as stepping stones that link the dye’s excited electrons to the conduction band of the titanium dioxide, providing smoother pathways into the bulk of the material. Because electrons can be injected more efficiently and with fewer chances to recombine with positive charges or leak back to the dye–electrolyte interface, the cell should deliver higher current and potentially a slightly higher voltage under real operating conditions.
What This Means for Future Solar Devices
In summary, the simulations suggest that carefully placing carbon at the surface of TiO2(B) can make N719 dye bind more strongly, sit closer, and exchange electrons more effectively with the semiconductor, all without disrupting the beneficial overall character of the material. While the work is theoretical, it offers concrete design rules for chemists and materials scientists: target specific surface sites for carbon substitution and favor dye arrangements that use three anchoring groups. If confirmed in the lab, these insights could guide the fabrication of dye-sensitized solar cells that are more efficient and more stable over time, helping this flexible solar technology move closer to widespread practical use.
Citation: Heffner, H., Marchetti, J.M., Faccio, R. et al. Computational study of carbon-doped TiO2(B) nanomaterials for improved dye-sensitized solar cells. Sci Rep 16, 8180 (2026). https://doi.org/10.1038/s41598-026-38897-7
Keywords: dye-sensitized solar cells, titanium dioxide, surface doping, solar energy materials, density functional theory