Clear Sky Science · en
Randomized pilot study of camrelizumab with or without autologous cytokine-induced killer cells in refractory clear cell renal cell carcinoma
New Hopes for Hard-to-Treat Kidney Cancer
For people with advanced kidney cancer that has stopped responding to standard drugs, treatment options are limited and frighteningly few. This study explores a promising way to boost the body’s own defenses by pairing a modern immunotherapy drug with a personalized infusion of activated immune cells, offering a glimpse of what future care for these patients might look like.
Why This Cancer Is So Difficult
Clear cell renal cell carcinoma is the most common type of kidney cancer. Many patients now live longer thanks to drugs that cut off the tumor’s blood supply and to immune checkpoint drugs that help the immune system recognize cancer. Yet some tumors eventually find ways to evade these treatments and keep growing. At that stage, doctors urgently need options that can push the immune system even harder, without causing unacceptable side effects.
Using Patients’ Own Cells as Reinforcements
In this trial, researchers tested whether combining the checkpoint drug camrelizumab with a patient’s own “trained” immune cells could improve outcomes. Camrelizumab works by blocking a molecular brake called PD‑1, which tumors use to shut down attacking T cells. The added treatment, called cytokine-induced killer (CIK) cell therapy, is made by collecting a patient’s blood, growing and activating certain white blood cells in the lab, and then infusing these energized cells back into the bloodstream. The idea is that camrelizumab releases the brakes while CIK cells act as fresh troops, entering the tumor and helping to turn a “cold,” suppressive environment into a “hot,” attack-ready one. 
What the Small Trial Found
The study enrolled 21 people with advanced clear cell kidney cancer whose disease had worsened despite earlier treatments. Patients were randomly assigned to receive either camrelizumab alone or camrelizumab plus repeated CIK cell infusions. Because the study stopped early—only about one-third of the planned participants could be enrolled—its results are considered exploratory rather than definitive. Even so, the numbers were encouraging: more than half of the patients who received the combination saw their tumors shrink meaningfully, compared with about four in ten in the drug-only group. One man in the combination group had all signs of active cancer disappear on specialized scans and remained alive with a good quality of life long after his treatment had to be stopped due to lung inflammation.
Signals of Longer Control, With Caveats
When the researchers looked at how long the cancer stayed in check and how long patients lived, those who received both camrelizumab and CIK cells appeared to do better. On average, people in the combination group went more than two years before their disease clearly worsened, compared with less than a year for those on camrelizumab alone. Overall survival also looked somewhat better in the combination group, but the study was too small to say for sure that these differences were not due to chance. The side effects seen—such as small skin blood-vessel growths, thyroid changes, fatigue, and rare lung inflammation—were mostly mild to moderate and were consistent with what doctors already know about camrelizumab; adding CIK cells did not noticeably increase treatment risks. 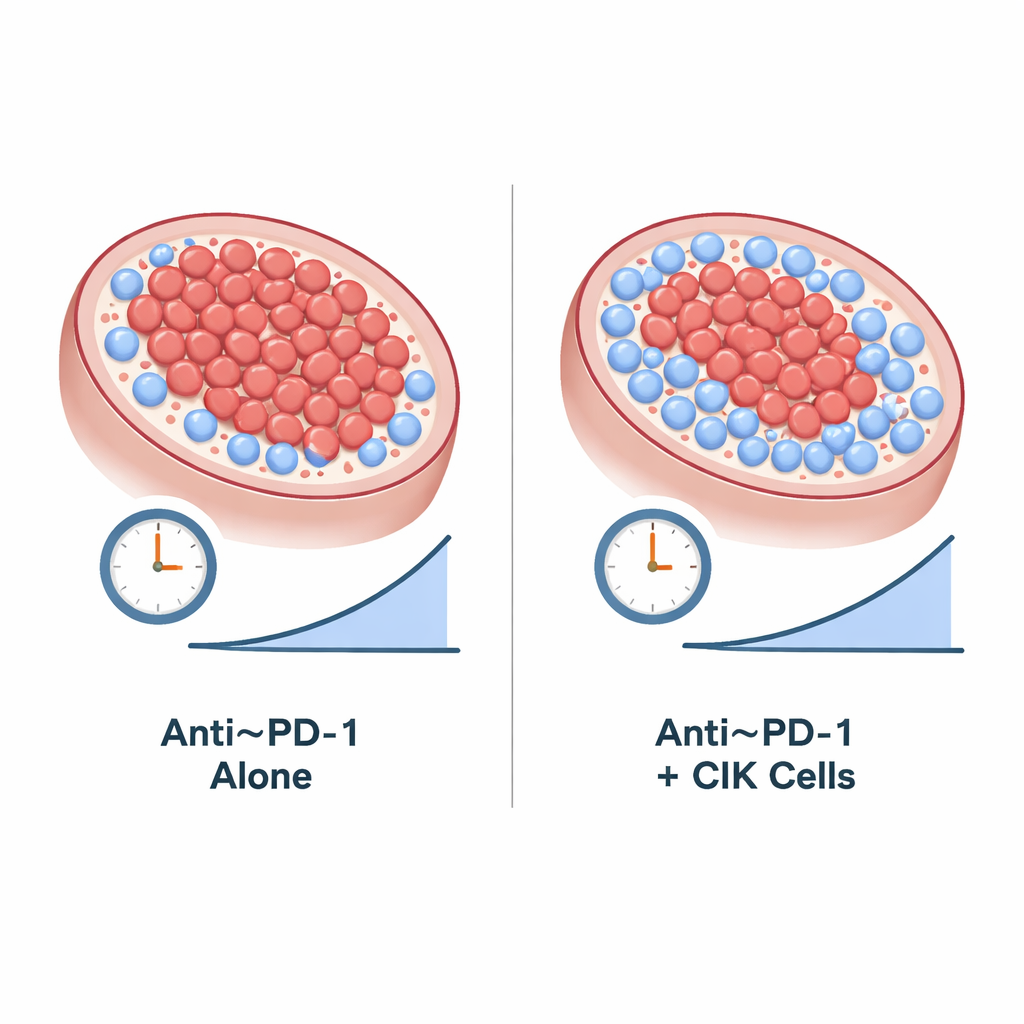
Clues from the Immune System
The team also took blood samples from some patients to search for early signs of who might benefit most from this approach. They focused on CD8 T cells, a key class of “killer” immune cells, and measured how many showed the PD‑1 marker before treatment. Patients whose killer cells started out with higher PD‑1 levels tended to respond better, and the share of PD‑1–positive cells generally dropped after therapy began—patterns that may reflect exhausted immune cells being revived. Because only a small number of patients were tested, these hints need to be confirmed in larger, more detailed studies.
What This Means for Patients
For now, this combined approach should be viewed as an experimental strategy rather than a new standard of care. The trial was too small to prove that adding CIK cells to camrelizumab truly extends life, and today’s first-line treatments for kidney cancer already pair immune drugs with blood-vessel–targeting pills. Still, the study shows that collecting, expanding, and reinfusing patients’ own immune cells alongside a checkpoint drug is feasible, appears safe, and may offer extra benefit for those whose disease has resisted other options. Larger, carefully designed trials will be needed to determine whether this “double-boost” immunotherapy can become a reliable new weapon against advanced kidney cancer.
Citation: Li, S., Qin, J., Sun, Q. et al. Randomized pilot study of camrelizumab with or without autologous cytokine-induced killer cells in refractory clear cell renal cell carcinoma. Sci Rep 16, 7768 (2026). https://doi.org/10.1038/s41598-026-38881-1
Keywords: kidney cancer, immunotherapy, checkpoint inhibitor, cell therapy, camrelizumab