Clear Sky Science · en
Single-cell mass-density measurements using microchannel gradient centrifugation
Why weighing tiny cells matters
Every living cell is more than just a bag of molecules; its weight and compactness tell a story about its health. Subtle changes in how densely packed a cell is can signal whether it is growing, dying, fighting infection, or turning cancerous. Yet, despite this promise, measuring the mass density of thousands of individual cells has remained slow, technically demanding, and expensive. This article presents a new, much simpler way to "weigh" single cells by how they float or sink in a controlled liquid inside a hair‑thin glass channel spun in a centrifuge.
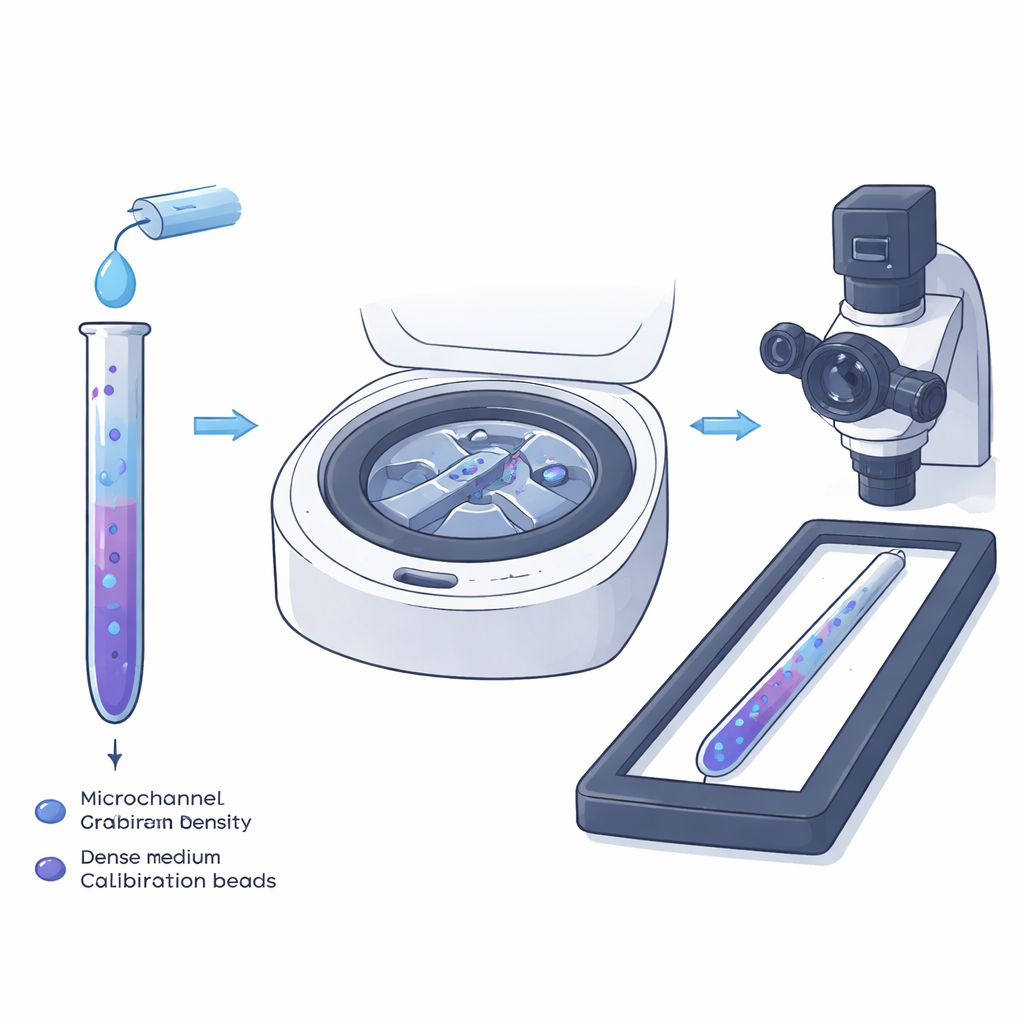
A new twist on an old lab trick
Traditional density‑gradient centrifugation has long been used in test tubes to separate mixtures of cells: when spun, cells settle at the point in a layered liquid where their own density matches that of the surrounding fluid. The authors miniaturize this idea into a narrow microchannel so that individual cells, not just layers, can be measured directly under a microscope. They fill the channel first with a light liquid containing the cells, then with a heavier liquid containing tiny plastic particles whose densities are precisely known. When these two liquids meet and flow in the tiny channel, they naturally form a smooth, one‑dimensional density gradient along its length.
Creating a smooth slope of density
Inside such thin channels, fluid flows slowly and smoothly, without turbulence. Under these conditions, the parabolic flow profile mixes the light and heavy liquids just enough to create a gradual transition, rather than a sharp boundary, between them. The team studied this process both experimentally using a fluorescent dye and through computer simulations. They found that a nearly linear density gradient extending several millimeters can be formed within seconds. Channel height turned out to be crucial: shallow channels keep the gradient stable and prevent gravity‑driven sloshing of the heavier liquid, which would blur the relationship between position and density and introduce errors in the final cell measurements.
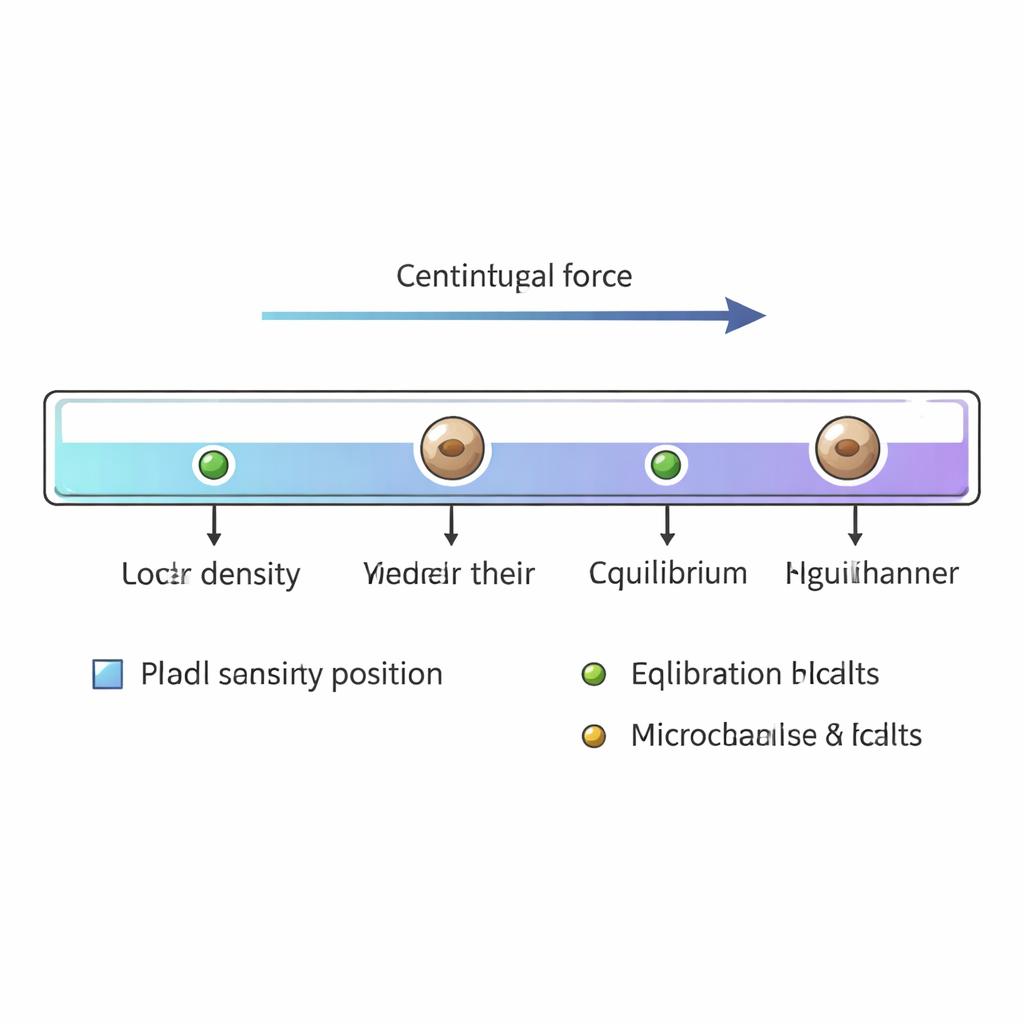
Spinning cells to their balance point
Once the channel is filled, its ends are sealed and it is placed in a small centrifuge. Spun at about 12,000 revolutions per minute, cells and calibration beads move along the channel until the outward force due to spinning is exactly balanced by buoyancy at their particular density. Larger than about three micrometers in diameter, yeast cells reach this equilibrium in under 20 seconds. After roughly a minute and a half of spinning, the channel is removed and scanned under a standard microscope. The researchers record the positions of thousands of single yeast cells and the reference beads, then convert each position along the gradient into a mass‑density value using the known densities of the beads as anchor points.
Reading cell health from tiny differences
Using this approach, the authors measured the densities of more than 20,000 yeast cells across multiple channels. The typical measurement uncertainty for a single cell was about 3.3 kilograms per cubic meter—small enough to resolve real biological differences, which were roughly twice as large in their samples. Over many hours, they observed that the main yeast population kept a stable density, while a second, denser and slightly smaller population gradually appeared. This denser group likely consisted of dead or damaged cells that had taken up the heavy fluid, making them more compact. The measured values matched well with results from far more complex and slower techniques such as suspended microchannel resonators, optical methods, and magnetic levitation.
From lab prototype to practical biomarker
The study shows that a simple combination of glass microchannels, off‑the‑shelf centrifuges, and standard microscopes can deliver high‑throughput, single‑cell density measurements at rates of roughly 16,000 cells per hour. While not yet sensitive enough to catch the tiniest drug‑induced changes, it is already powerful enough to distinguish different cell types or living from dead cells based on how tightly packed they are. By making precise cell "weighing" more accessible and affordable, this microchannel gradient method could help turn cell mass density into a routine biomarker for monitoring disease, evaluating therapies, and exploring how cells regulate their internal makeup.
Citation: Soller, R., Augustsson, P. & Barnkob, R. Single-cell mass-density measurements using microchannel gradient centrifugation. Sci Rep 16, 6501 (2026). https://doi.org/10.1038/s41598-026-38872-2
Keywords: single-cell density, microfluidics, centrifugation, yeast cells, cell biomarkers