Clear Sky Science · en
RHA/TiO2-[bip]-NH2+NO3− as an efficient catalyst for the solvent-free synthesis of 1,8-dioxo-decahydroacridine and 2,3-dihydroquinazolin-4(1H)-one derivatives
Turning Farm Waste into Helpful Chemistry
Chemists are constantly looking for cleaner, faster ways to make the complex ring-shaped molecules that appear in many medicines. This study shows how an everyday agricultural by-product—rice husk ash—can be transformed into the backbone of a new solid catalyst that drives such reactions quickly, without using any solvent, and can be reused several times. For readers interested in green technology and drug discovery, it offers a glimpse of how discarded plant material can be upgraded into a high‑performance tool for building bioactive compounds.
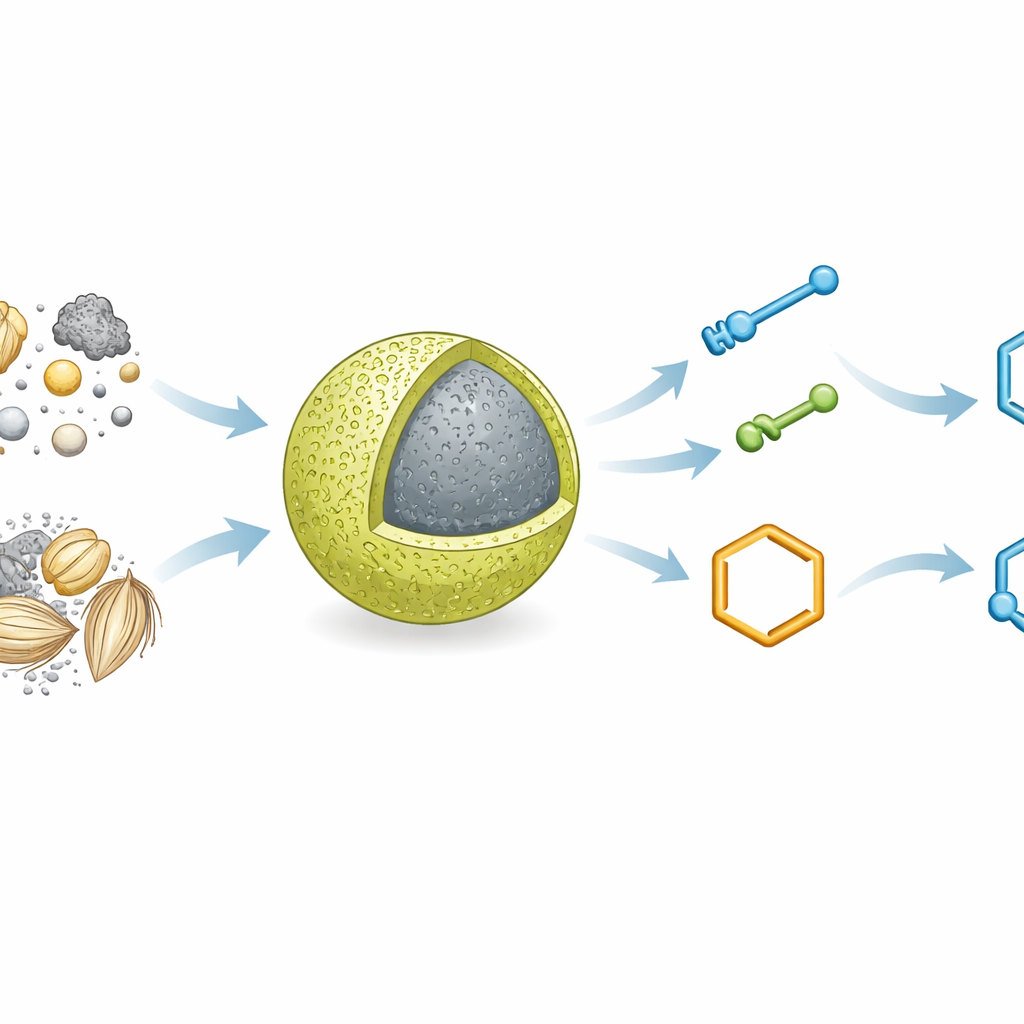
Why These Ring Molecules Matter
The team focused on two families of nitrogen‑containing ring systems: 1,8‑dioxo‑decahydroacridines and 2,3‑dihydroquinazolin‑4(1H)‑ones. Although their names sound forbidding, these structures underpin many experimental and approved drugs, with activities ranging from anticancer and antibacterial to antioxidant and cardiovascular effects. Because more than three‑quarters of small‑molecule medicines contain nitrogen, efficient routes to such ring frameworks are highly prized. The researchers used multi‑component reactions, where three or more simple ingredients join together in a single pot, an approach that saves time, reduces waste, and fits well with the principles of green chemistry.
Building a Catalyst from Ash and Oxide
The catalyst at the heart of this work is a carefully engineered hybrid material. The scientists first prepared a nanoporous composite of rice husk ash, rich in silica, and titanium dioxide (TiO₂) nanoparticles. They then chemically attached a bridge‑like organic fragment that carries acidic ionic groups, creating a solid with mild but well‑defined acidity. This final material, referred to in the paper by a long formula, behaves like an immobilized ionic liquid: it has the tunable reactivity of liquid acids but is fixed on a solid support. A battery of techniques—including infrared spectroscopy, X‑ray diffraction, electron microscopy, surface analysis, and thermal measurements—confirmed that the structure is stable, the components are well mixed at the nanoscale, and the ionic groups are firmly anchored to the ash–oxide framework.
Fast, Solvent‑Free Reactions
With the catalyst in hand, the team tested it in one‑pot syntheses of the two target ring systems under solvent‑free conditions. For the dihydroquinazolinone products, a mixture of an isatoic anhydride, an aldehyde, and ammonium acetate was heated with a small amount of the catalyst. Under optimized conditions, the desired products formed in as little as five minutes, often in virtually quantitative yield. A similar three‑component protocol—from dimedone or related diketones, aldehydes, and ammonium acetate—produced the acridinedione family in around ten minutes at slightly higher temperature. Both electron‑rich and electron‑poor aldehydes worked well, and calculated turnover numbers and frequencies showed that each acidic site on the material participates in many successful reaction cycles.
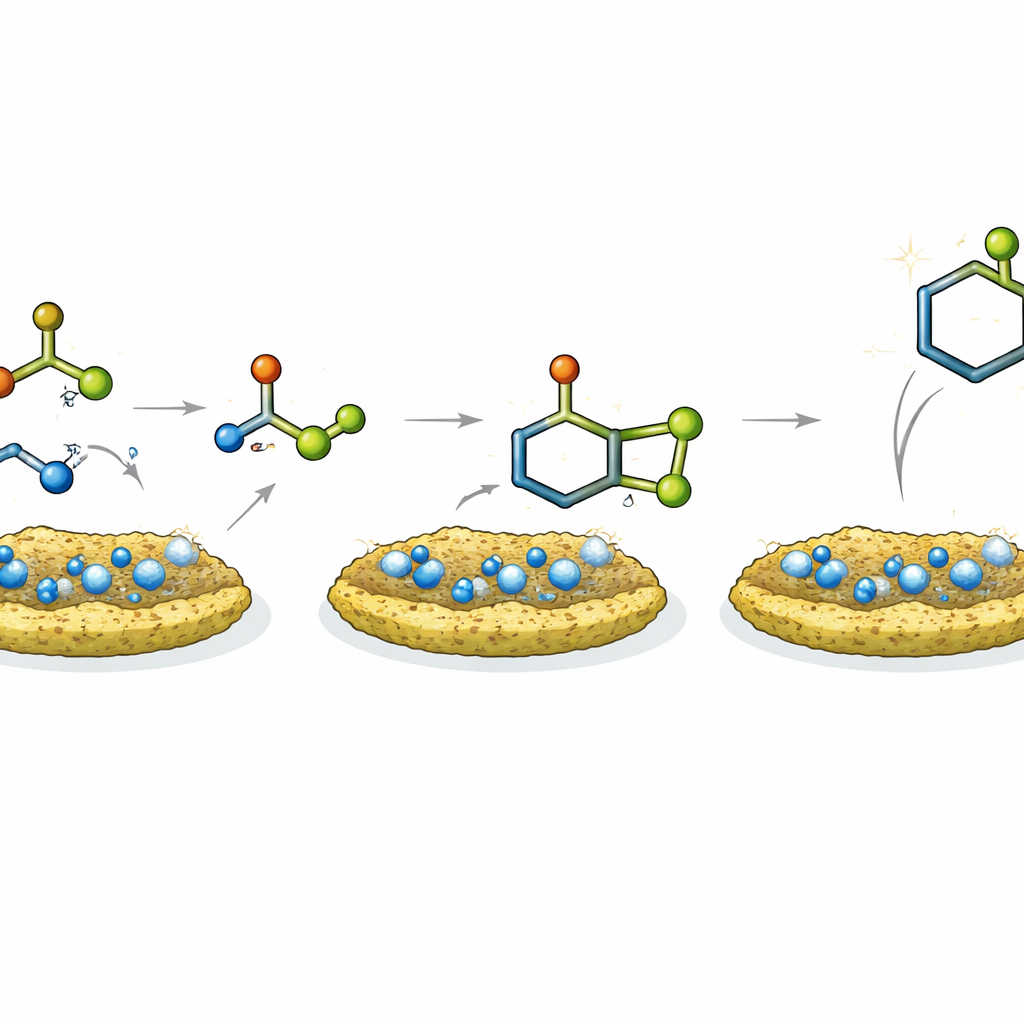
How the Catalyst Does Its Job
Mechanistic experiments and comparisons with other catalysts suggest that the mild acidity and nanostructured surface act together. The acidic sites on the solid activate the carbon–oxygen bonds of the starting materials, making them more susceptible to attack by nitrogen‑bearing partners, while the porous ash–TiO₂ scaffold concentrates the reactants and brings them together. For the quinazolinone route, the catalyst helps first convert isatoic anhydride into an aminobenzamide, then promotes combination with an aldehyde and final ring closure. In the acridinedione pathway, it assists a condensation step, formation of a reactive enamine, and a subsequent addition that closes the ring. A small study of reaction rates with differently substituted aldehydes supports the idea that both electronic effects and the multi‑step nature of the process control the speed.
Lasting Performance and Greener Prospects
Beyond speed and yield, the material’s durability is crucial for sustainable use. The researchers showed that the catalyst can be filtered off after each reaction, washed, and reused at least five times with only modest loss of activity. Structural and elemental analyses before and after recycling revealed no significant changes, indicating that the ionic groups stay in place and the ash–TiO₂ scaffold remains intact. Overall, the work demonstrates that a solid built from rice husk ash and titanium dioxide can match or surpass more traditional acid catalysts while avoiding corrosive conditions and excess solvent. For non‑specialists, the key takeaway is that agricultural waste can be redesigned into a robust, reusable tool that helps chemists assemble valuable, drug‑like molecules in a cleaner and more efficient way.
Citation: Aloueian, F., Shirini, F., Gholinejad, M. et al. RHA/TiO2-[bip]-NH2+NO3− as an efficient catalyst for the solvent-free synthesis of 1,8-dioxo-decahydroacridine and 2,3-dihydroquinazolin-4(1H)-one derivatives. Sci Rep 16, 8190 (2026). https://doi.org/10.1038/s41598-026-38867-z
Keywords: green catalysis, rice husk ash, multicomponent reactions, heterocyclic synthesis, ionic liquid catalyst