Clear Sky Science · en
Bifidobacterium breve promotes growth and lipid alteration in Trichomonas vaginalis transiently through transcriptomic reprogramming
Why Tiny Vaginal Microbes Matter
The human vagina is home to a bustling community of microbes that quietly help keep infections at bay. Among these residents are friendly bacteria such as Bifidobacterium breve and an unwelcome parasite called Trichomonas vaginalis, which causes the world’s most common non-viral sexually transmitted infection. This study asks a surprisingly simple but important question: when these two microbes meet, do the “good” bacteria help protect us, or do they accidentally give the parasite a boost?
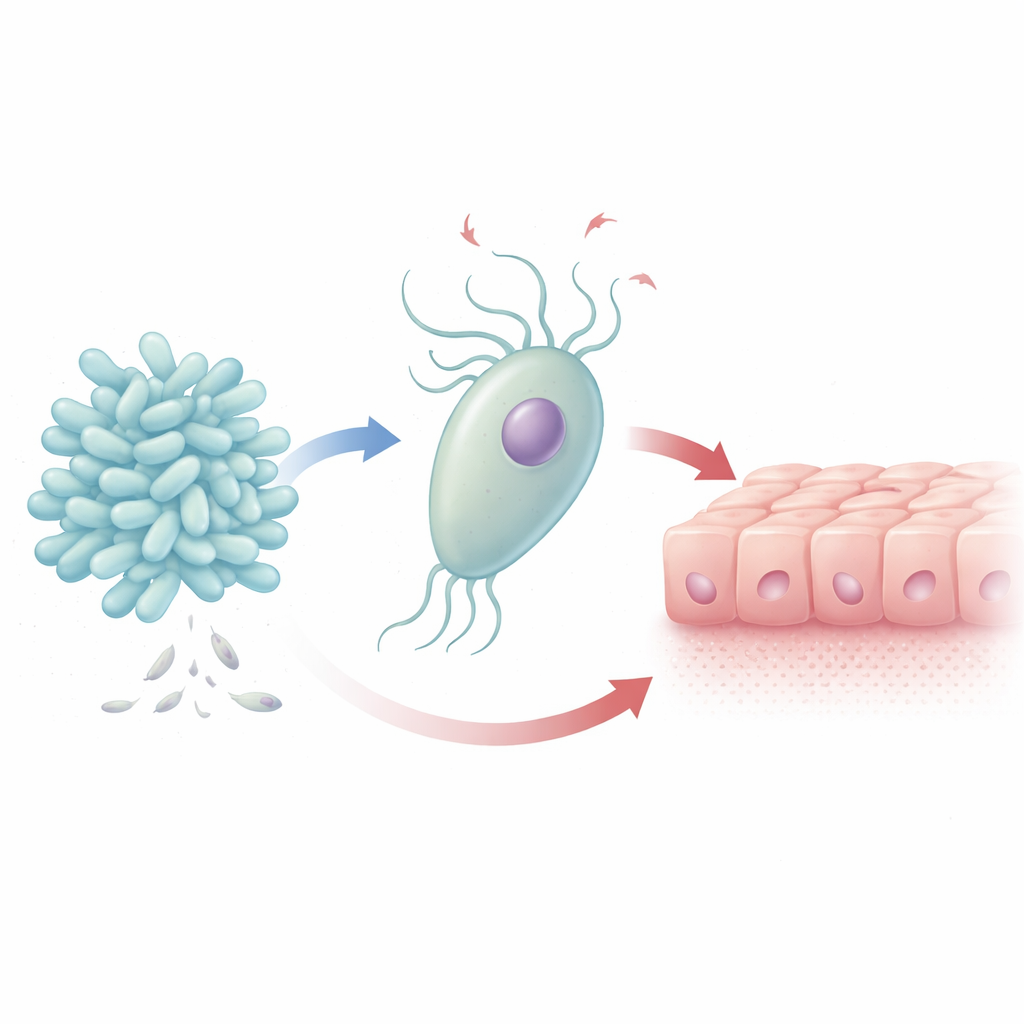
A Delicate Balance in the Vaginal Neighborhood
Women with healthy vaginal ecosystems usually have communities dominated by protective bacteria that make lactic acid and help maintain a low pH, discouraging invaders. B. breve is one such helpful species, while T. vaginalis is a flagellated protozoan linked not only to discomfort and inflammation but also to higher risks of HIV and persistent human papillomavirus infections. Clinical studies have noticed that women infected with T. vaginalis often have fewer B. breve, suggesting that parasite overgrowth goes hand in hand with shifts in the normal microbial balance. The authors set out to understand whether B. breve fights the parasite, ignores it, or, unexpectedly, supports it.
When “Good” Bacteria Meet a Parasite
In lab dishes, the researchers grew B. breve together with T. vaginalis under oxygen-free conditions that mimic the vaginal environment. Over just four hours, the parasite’s numbers rose by about 20 percent, while the bacterial population dropped by more than a quarter. Under the microscope, B. breve clustered tightly around the surface of T. vaginalis, hinting at direct physical contact between the two. When the parasite that had been exposed to B. breve was later added to human cervical cells (HeLa cells), the host cells did not show any extra protection: there was no reduction in the parasite’s ability to damage the cell layer.
Testing Host Protection and Inflammation
The team then explored two realistic scenarios: what happens if B. breve settles on vaginal cells before the parasite arrives, and what if both reach the cells at the same time? In both cases, B. breve failed to shield the epithelial cells from damage caused by T. vaginalis. Measures of cell coverage and shape remained essentially unchanged whether or not B. breve was present. When the scientists measured inflammatory signals, especially the cytokines IL-6 and IL-8, they found that B. breve sometimes even amplified these early responses. Rather than calming inflammation, simultaneous or prior exposure to B. breve tended to produce brief spikes in these signals, which could be a double-edged sword—supporting immune defense, but also potentially feeding parasite survival.
Rewiring the Parasite from the Inside
To understand how the parasite itself changes in the presence of B. breve, the researchers analyzed which genes turned on or off in T. vaginalis and what happened to its fat-related molecules. They found that genes involved in copying DNA, packaging chromosomes, and generating energy were switched on, consistent with faster parasite growth. Importantly, genes and pathways tied to fatty acid use—especially the handling of long-chain fats and short-chain fatty acids like propionate—were strongly activated. Detailed chemical measurements revealed that many fatty acids inside the parasite decreased after co-culture, suggesting that T. vaginalis was burning through these fats as fuel and building material. Together, the data point to a brief metabolic “reprogramming” in which B. breve contact nudges the parasite to consume more fats and divide more quickly.
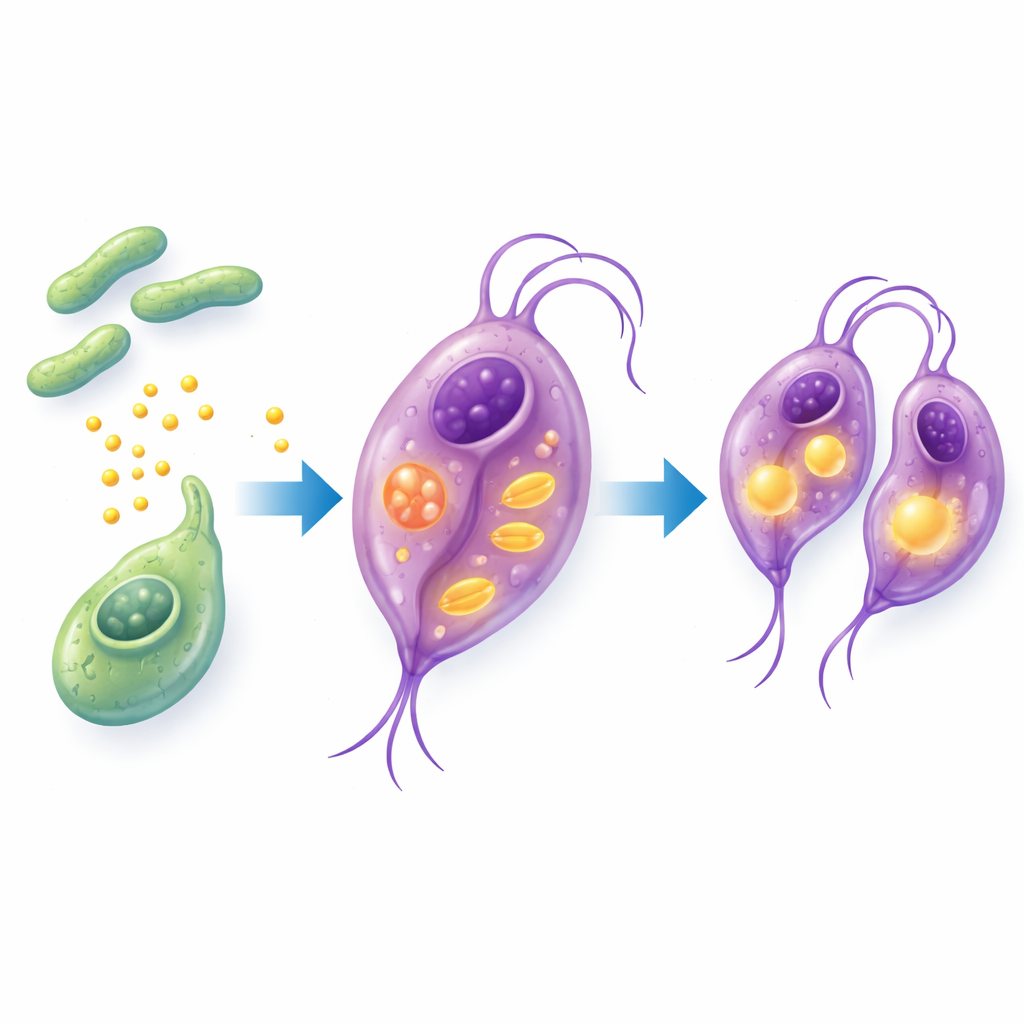
What This Means for Vaginal Health
Overall, the study shows that B. breve, despite its reputation as a probiotic ally, does not act as a simple bodyguard against T. vaginalis. In the short term, these friendly bacteria appear to stick to the parasite, spur its growth, and tweak its use of fats, without protecting human cells from damage. Over longer periods, B. breve may still help maintain overall microbial balance, but it does not directly counter this particular pathogen. For patients and clinicians, this work underscores that not all “good” bacteria are protective in every context and that the mix and timing of microbes in the vagina may signal when conditions are shifting toward parasite overgrowth, potentially offering new early-warning markers or targets for microbiome-based therapies.
Citation: Chen, PY., Yeh, YM., Chen, CH. et al. Bifidobacterium breve promotes growth and lipid alteration in Trichomonas vaginalis transiently through transcriptomic reprogramming. Sci Rep 16, 8997 (2026). https://doi.org/10.1038/s41598-026-38866-0
Keywords: vaginal microbiome, Trichomonas vaginalis, Bifidobacterium breve, fatty acid metabolism, probiotic–parasite interaction