Clear Sky Science · en
Determination of cardiac output, shunt-fraction, and active circulatory volume in children with hypoplastic left heart syndrome after the Norwood procedure with RV to PA-shunt.
Keeping a Fragile Newborn Heart in Balance
For babies born with hypoplastic left heart syndrome, the main pumping chamber that should send blood to the body is severely underdeveloped. Surgeons can reroute the circulation with a first-stage operation called the Norwood procedure, but the days after surgery are a tightrope walk: doctors must share a single working ventricle between the lungs and the rest of the body. This study explores how different amounts of oxygen in the breathing gas affect that delicate balance, and what really controls where the blood goes.
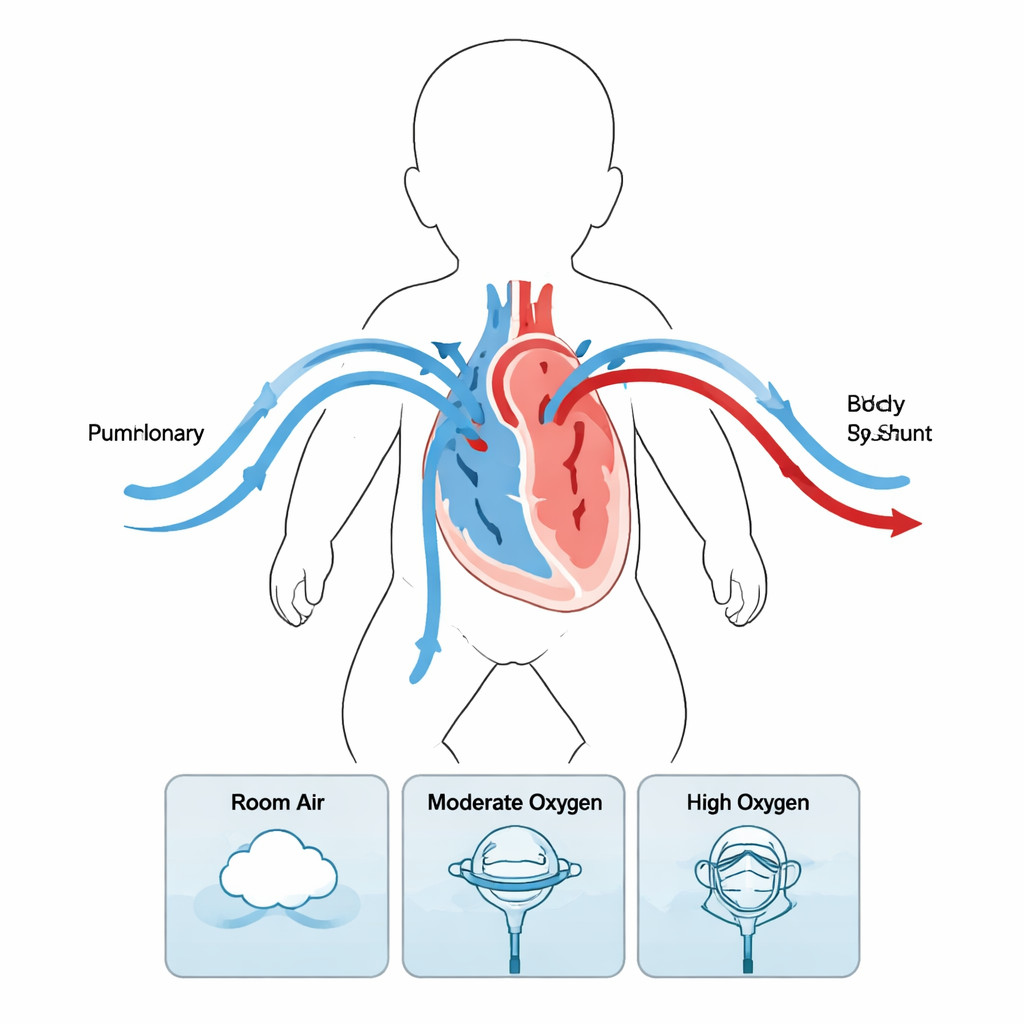
A Single Pump Doing Double Duty
In a typical heart, one ventricle sends blood to the lungs and the other sends blood to the body. In these newborns, one ventricle has to do both jobs. During the Norwood procedure, surgeons create a new pathway from the heart to the body and connect the right ventricle to the lung arteries through a small tube, or shunt. Blood leaving the heart therefore splits: some flows through the shunt to the lungs (pulmonary flow, Qp) and some goes out to the body (systemic flow, Qs). Getting the right Qp/Qs balance is critical. Too much to the lungs robs the body of oxygen-rich blood; too little threatens oxygen pickup in the lungs.
A New Way to Measure Tiny Hearts
Standard bedside signs like blood pressure and oxygen readings give only a rough picture of circulation in these fragile infants. Directly measuring oxygen use and blood flow, which is needed for classic calculations, is especially difficult and error-prone in small, critically ill babies. The researchers used an ultrasound dilution technique instead. They briefly injected warm saline into a venous line and measured tiny changes in how sound travels through the blood as it passes through an extracorporeal loop. From the shape of the resulting curves, specialized software could estimate total cardiac output, how much blood went to the lungs versus the body, and how much blood was actively circulating in the vessels at any moment.
Turning Up the Oxygen: What Really Changes
Sixteen newborns were studied soon after their Norwood operations while lightly sedated and on ventilators. The team measured blood flow and pressures at three oxygen levels in the breathing gas: roughly room air, moderate extra oxygen, and very high oxygen. As oxygen was increased, blood flow to the lungs remained essentially unchanged, but blood flow to the body fell. Because the lung flow stayed steady while body flow dropped, the calculated lung-to-body flow ratio (Qp/Qs) rose. Yet pressures driving blood through the lungs and the overall resistance across the lung circuit did not change in a meaningful way. Instead, the body’s blood vessels became tighter, raising systemic vascular resistance, and the babies’ effective circulating blood volume—which was already low compared with children with two functioning ventricles—fell even further.
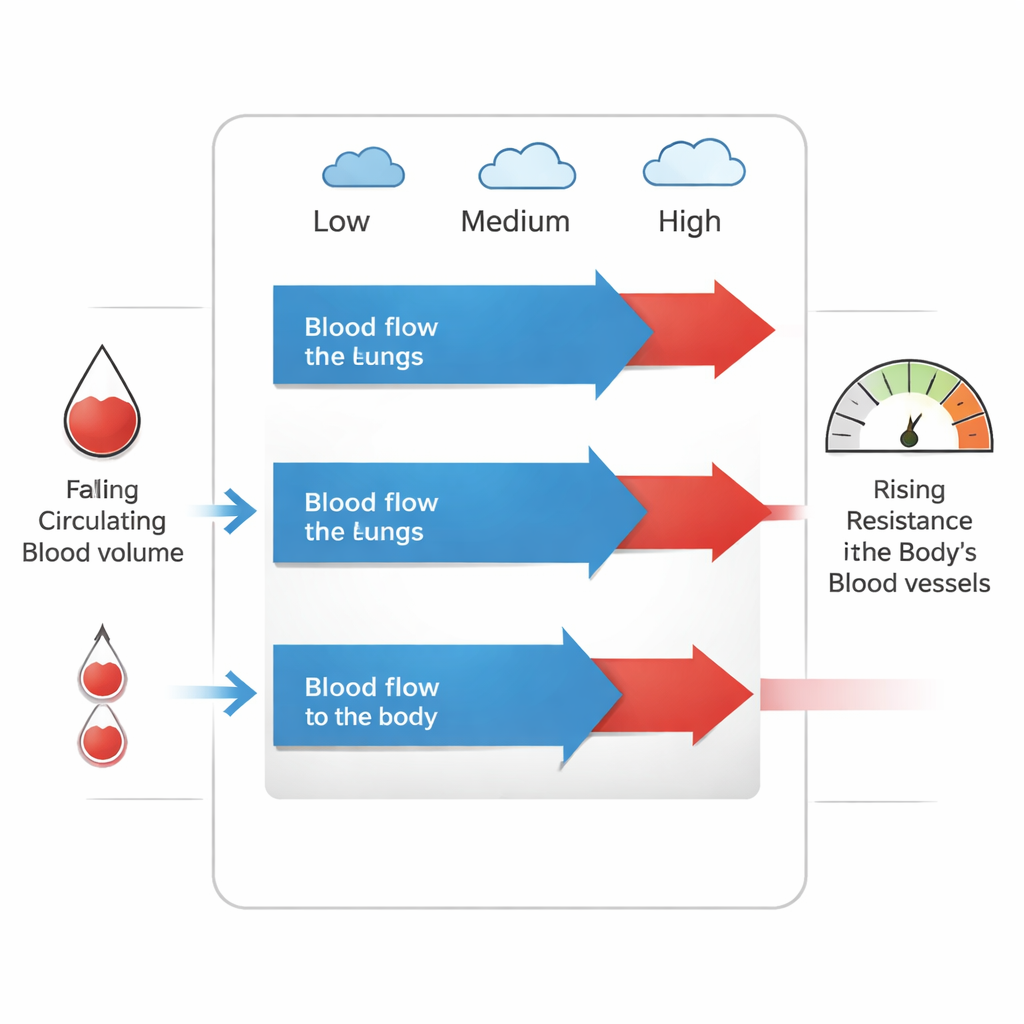
Why a Fixed Tube Matters More Than Oxygen
These patterns suggest that, early after surgery, the rigid shunt from the ventricle to the lung arteries acts as the main “valve” controlling lung blood flow. Because the shunt behaves like a fixed mechanical bottleneck, extra oxygen—normally a powerful lung vessel relaxer—has limited ability to boost flow to the lungs. Instead, higher oxygen appears to constrict the body’s blood vessels, slightly slow the heart rate, and worsen the relative shortage of circulating blood. The result is less blood reaching vital organs, even as oxygen readings in the arteries and veins rise because gas exchange in the lungs improves. Small shifts in blood volume, for example during infection or inflammation, may further tip the balance toward the lungs and away from the body.
What This Means for Treating These Newborns
For clinicians, the study implies that simply turning up the oxygen or using drugs that relax lung vessels may not be the most effective way to protect these babies. Since lung flow is largely set by the size and stiffness of the shunt, careful surgical choice of shunt size and postoperative strategies that expand circulating blood volume and gently lower the resistance in the body’s vessels may be more important for keeping organs well supplied. In plain terms, the findings argue that supporting the body’s side of the circulation—how much blood it gets and how hard the heart has to push against it—may matter more than trying to fine-tune lung blood flow with oxygen in the crucial days after the Norwood operation.
Citation: Aronsson, A., Sigurdsson, T.S. & Lindberg, L. Determination of cardiac output, shunt-fraction, and active circulatory volume in children with hypoplastic left heart syndrome after the Norwood procedure with RV to PA-shunt.. Sci Rep 16, 4748 (2026). https://doi.org/10.1038/s41598-026-38858-0
Keywords: hypoplastic left heart syndrome, Norwood procedure, single ventricle, neonatal hemodynamics, oxygen therapy