Clear Sky Science · en
Cadmium induces ferroptosis in mouse spermatocytes by activating the ROS–TCA pathway
Why a Common Metal Matters for Men's Health
Cadmium is a soft metal used in batteries, pigments, and other industrial products. It can build up in soil, water, and food, quietly entering our bodies over time. Many studies have linked cadmium exposure to lower sperm counts and poorer sperm quality in men, but the exact way it injures the cells that make sperm has remained murky. This study zooms in on that problem, uncovering how cadmium pushes mouse sperm‑forming cells down a specific path of cell death tied to iron and a stressed energy system inside their mitochondria, the cell’s power plants.
From Pollution to Vulnerable Sperm Cells
Cadmium is a well-known environmental troublemaker for the male reproductive system. Men exposed at work or living in polluted regions often show reduced sperm number, movement, and normal shape, and even DNA damage in their sperm. Animal studies echo these findings, showing shrunken testes, leaky protective barriers, low testosterone, and widespread loss of germ cells. The authors previously observed that cadmium could trigger an unusual, iron-dependent form of cell death called ferroptosis in testicular tissue. In the present work, they focused on mouse spermatocytes—cells in the middle of becoming sperm—to see exactly how cadmium harms them and whether this iron-linked death route is involved.
Iron Overload and a Special Kind of Cell Death
To investigate, the team grew a mouse spermatocyte cell line in dishes and exposed the cells to cadmium, with or without drugs known to block ferroptosis. Cadmium cut cell survival roughly in half and slowed cell division. Inside the cells, levels of free iron and a marker of fat damage called malondialdehyde rose sharply, while a key protective enzyme, GPX4, dropped. Other proteins involved in iron storage and export increased, signaling that iron handling was badly disturbed. When the researchers added ferroptosis blockers—an iron-chelating compound and a lipid-protecting molecule—many of these harmful changes eased and cell survival improved. Together, these shifts match the signature of ferroptosis, indicating that cadmium is not just killing cells in a vague way, but by driving them into this specific iron- and lipid-driven death program.
Mitochondria Under Siege
The story deepened when the scientists turned their attention to mitochondria, the energy centers that also generate reactive oxygen species, or ROS, as by‑products. Under the microscope, healthy sperm cells showed long, threadlike mitochondria, but cadmium-exposed cells displayed short, fragmented ones, a sign of structural damage. Measurements confirmed that mitochondrial ROS spiked, while the electrical charge across the mitochondrial membrane—a key indicator of function—fell. The cells responded by cranking up internal defense systems, including proteins governed by the stress sensor Nrf2, yet these defenses were not enough. Importantly, the ferroptosis-blocking drugs reduced mitochondrial ROS and partially restored mitochondrial health, linking the damaged power plants directly to the iron-dependent death pathway.
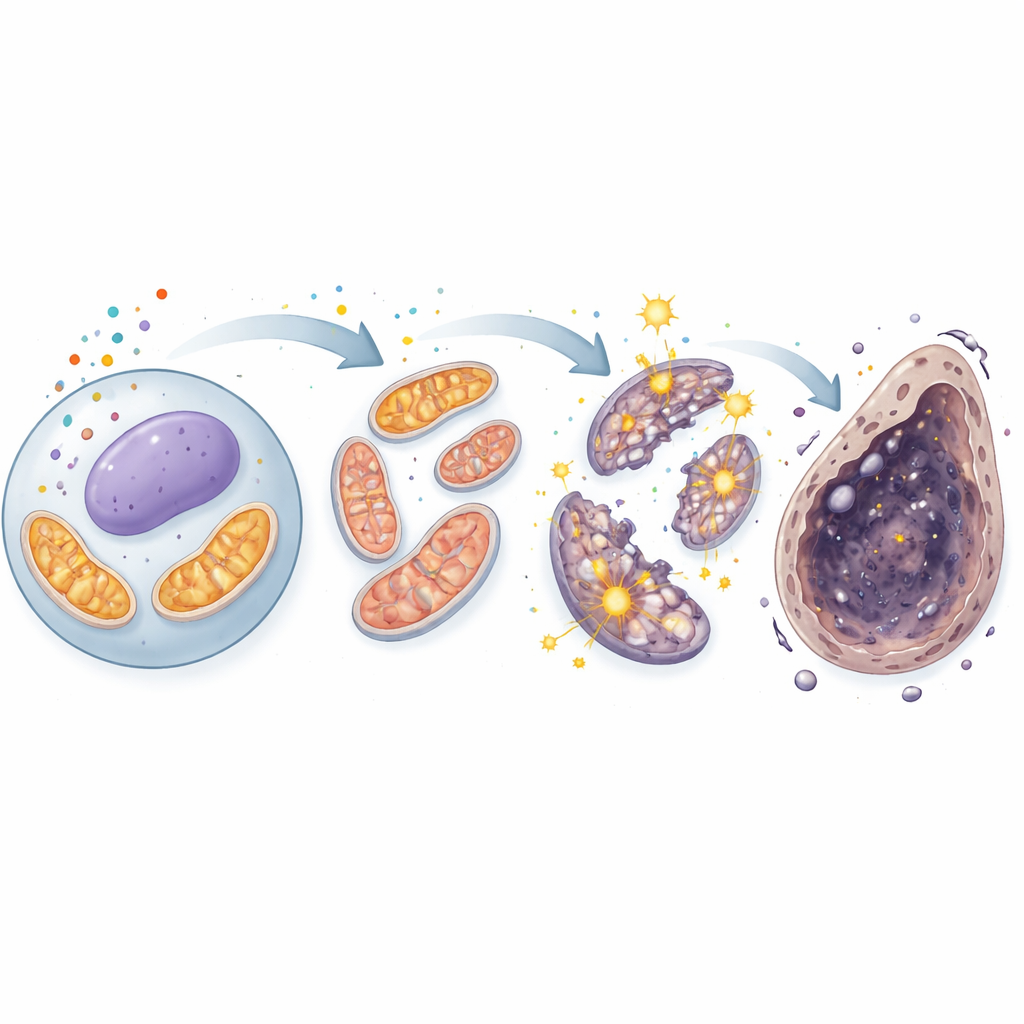
A Vicious Cycle in the Cell's Power Plant
Using a combination of gene activity profiling and metabolite measurements, the researchers saw that cadmium did more than simply injure mitochondria—it seemed to hijack their core energy circuit, the tricarboxylic acid (TCA) cycle. Levels of two central TCA intermediates, alpha‑ketoglutarate and succinate, rose significantly, and the activity of pyruvate carboxylase, an enzyme that feeds fresh fuel into the cycle, was boosted. This suggests that the TCA cycle became paradoxically overactive in damaged cells, forcing more electrons through the respiratory chain and leaking more ROS in the process. That extra ROS then fed back into iron chemistry and membrane damage, reinforcing ferroptosis. Again, ferroptosis inhibitors dialed back these metabolic distortions, underscoring the tight link between cadmium, mitochondrial overdrive, and iron-dependent cell death.
What This Means for Fertility and Future Research
In plain terms, the study shows that cadmium attacks sperm-forming cells by directly harming their mitochondria and throwing their energy cycle into harmful overdrive. This combination floods the cells with reactive oxygen molecules and disrupts iron balance, pushing them into ferroptosis. Because spermatocytes are essential for producing healthy sperm, their loss can help explain why cadmium exposure is so strongly tied to reduced male fertility. While these experiments were done in mouse cells in a dish, they provide a clear mechanistic map that future animal and human studies can test. The work also highlights potential protective strategies: drugs or nutrients that calm mitochondrial stress, fine‑tune the TCA cycle, or prevent iron-driven membrane damage might help shield the male reproductive system from environmental metals like cadmium.
Citation: Xiong, L., Yi, L., Zeng, X. et al. Cadmium induces ferroptosis in mouse spermatocytes by activating the ROS–TCA pathway. Sci Rep 16, 8449 (2026). https://doi.org/10.1038/s41598-026-38827-7
Keywords: cadmium toxicity, male fertility, mitochondrial stress, ferroptosis, reactive oxygen species