Clear Sky Science · en
Practical in-syringe mixing method for uniform particle delivery during embolization procedures
Keeping Tiny Treatment Beads on Target
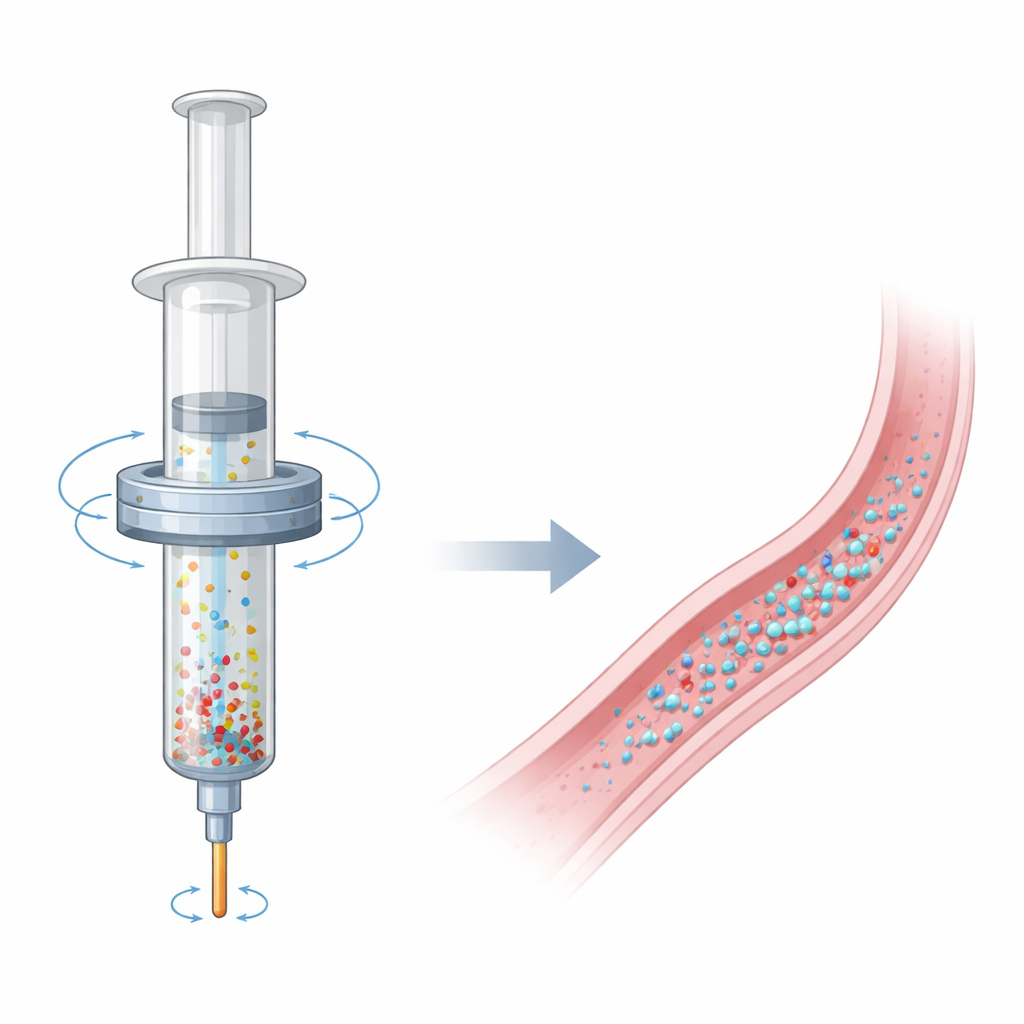
Many minimally invasive cancer and blood‑vessel treatments rely on microscopic beads that are injected through a syringe to intentionally block small vessels. For these procedures to work as intended, doctors need those beads to flow into the body in a smooth, even stream. In reality, the beads tend to sink or float inside the syringe, so patients may receive mostly liquid at first and a sudden burst of beads at the end. This study introduces a compact, battery‑powered way to gently stir the syringe from the outside, keeping the beads evenly spread out and helping doctors deliver more predictable, safer treatments.
Why Uneven Bead Flow Is a Hidden Problem
In embolization procedures, tiny particles are mixed with a contrast dye and saline so physicians can watch them move under X‑ray while they block blood flow to a tumor or abnormal vessel. Even when the mixture starts out well blended, the particles slowly separate from the liquid in less than a few minutes, settling toward the bottom of the syringe or floating to the top, depending on their density. Because these beads themselves are invisible to X‑rays, the doctor sees only the dye, not the true bead concentration. That means the injection can look normal on the screen while the actual dose of beads delivered over time is highly uneven—mostly liquid early on, followed by clumps of beads or even a large bolus at the end.
A Simple Ring That Turns the Syringe into a Stirrer
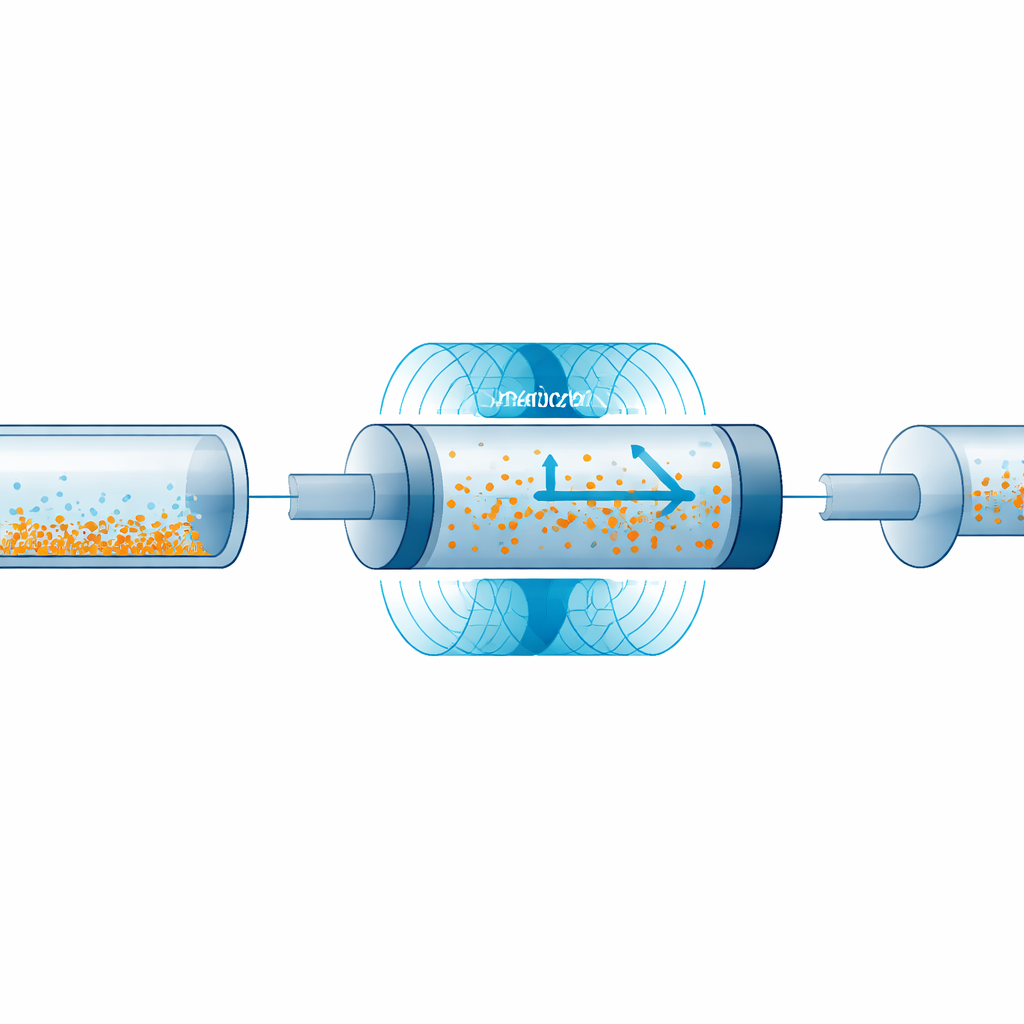
The researchers repurposed the stationary outer shell of a common stepper motor—the same kind of part used in 3D printers and robots—to create a rotating magnetic field around a standard plastic syringe. By removing the internal moving parts of the motor, they left a ring‑shaped "stator" with a central opening just wide enough for a syringe barrel. Inside the syringe, they placed a small rod‑ or impeller‑shaped magnetic agitator that spins when exposed to the changing magnetic field produced by the stator. When powered by a compact controller and AA batteries, the stator makes the hidden magnetic rod spin and reverse direction at programmed speeds, gently stirring the fluid and particles without any moving parts outside the syringe.
Testing How Well the Beads Stay Even
To see whether this in‑syringe mixing actually improves delivery, the team used large, clinically used hydrogel beads suspended in a water‑contrast mixture. They filmed the particles flowing through a custom observation channel connected to the syringe, using a microscope to count beads frame by frame as they exited during injections at different speeds. When the syringe was left still after initial shaking, longer waiting times before injection caused more beads to settle. That produced very uneven flow: modest bead output at the start, a long period of mostly liquid, and then a surge of densely packed beads at the end. Measured mathematically, nonuniformity grew as delay increased, and was especially severe at slow injection rates, where particles had plenty of time to settle mid‑procedure.
How Continuous Stirring Changes the Picture
When the magnetic agitator was activated inside the syringe, spinning throughout a two‑minute waiting period and during injection, bead delivery became much smoother. Under the worst‑case condition—slow injection after a long delay—the mixing system cut variations in bead concentration roughly fourfold at common injection speeds and at least twofold even at the slowest rate. The rod‑shaped agitator performed slightly better than the impeller, so it became the preferred design. The team also explored different spin speeds and how often the agitator reversed direction. Moderate speeds (around ten turns per second) with frequent reversals every quarter‑second produced the most even flow; very slow or very fast spinning, or always spinning in the same direction, tended to push beads away from the outlet or mix them only in part of the syringe. Short, high‑speed bursts separated by pauses could rapidly re‑suspend the entire contents with minimal disturbance to the injection.
From Lab Setup to Real‑World Use
Beyond the main experiments, the authors showed that the same approach can keep particles from rapidly settling in thin, water‑like liquids and can partially resuspend them in thick, syrupy contrast fluid. They discuss how factors such as particle size, density, and fluid thickness will shape the best mixing recipe in different applications, and note practical considerations like heat generation, syringe angle, and the small volume taken up by the agitator. Importantly, the system works with off‑the‑shelf motor parts and standard syringes, with no need for custom syringe bodies or complex mechanical drives, making it easier to integrate into clinical or research workflows.
What This Means for Patients and Practitioners
For a layperson, the key outcome is that this device turns a regular syringe into a self‑stirring injector that can deliver tiny treatment beads far more evenly over time. Instead of an unpredictable trickle of a few beads followed by a sudden rush, the patient is more likely to receive a steady, controlled stream. That should help doctors hit their targets more reliably, reduce the chance of stray blockages in healthy tissue, and make dosage more consistent from case to case. While further work is needed to tailor the mixing settings for different bead types and fluids, the study shows that a simple magnetic stirring ring could quietly solve a long‑standing, largely invisible problem in image‑guided treatments.
Citation: Ng, D.KH., Drangova, M. & Holdsworth, D.W. Practical in-syringe mixing method for uniform particle delivery during embolization procedures. Sci Rep 16, 9245 (2026). https://doi.org/10.1038/s41598-026-38823-x
Keywords: embolization, microspheres, syringe mixing, magnetic stirring, interventional radiology