Clear Sky Science · en
DeepRetro discovers retrosynthetic pathways through iterative large language model reasoning
Why Smarter Chemistry Matters
Many of today’s most important medicines and materials begin as intricate, hard-to-make molecules. Planning how to build these molecules in the lab is a bit like figuring out the best way to dismantle and then rebuild an elaborate piece of machinery from spare parts. That planning step, called synthesis design, is often a major bottleneck for drug discovery and advanced materials. This paper introduces DeepRetro, a new open-source system that uses large language models—the same class of AI behind modern chatbots—together with traditional chemistry software and human expertise to design realistic step-by-step recipes for making very complex molecules.
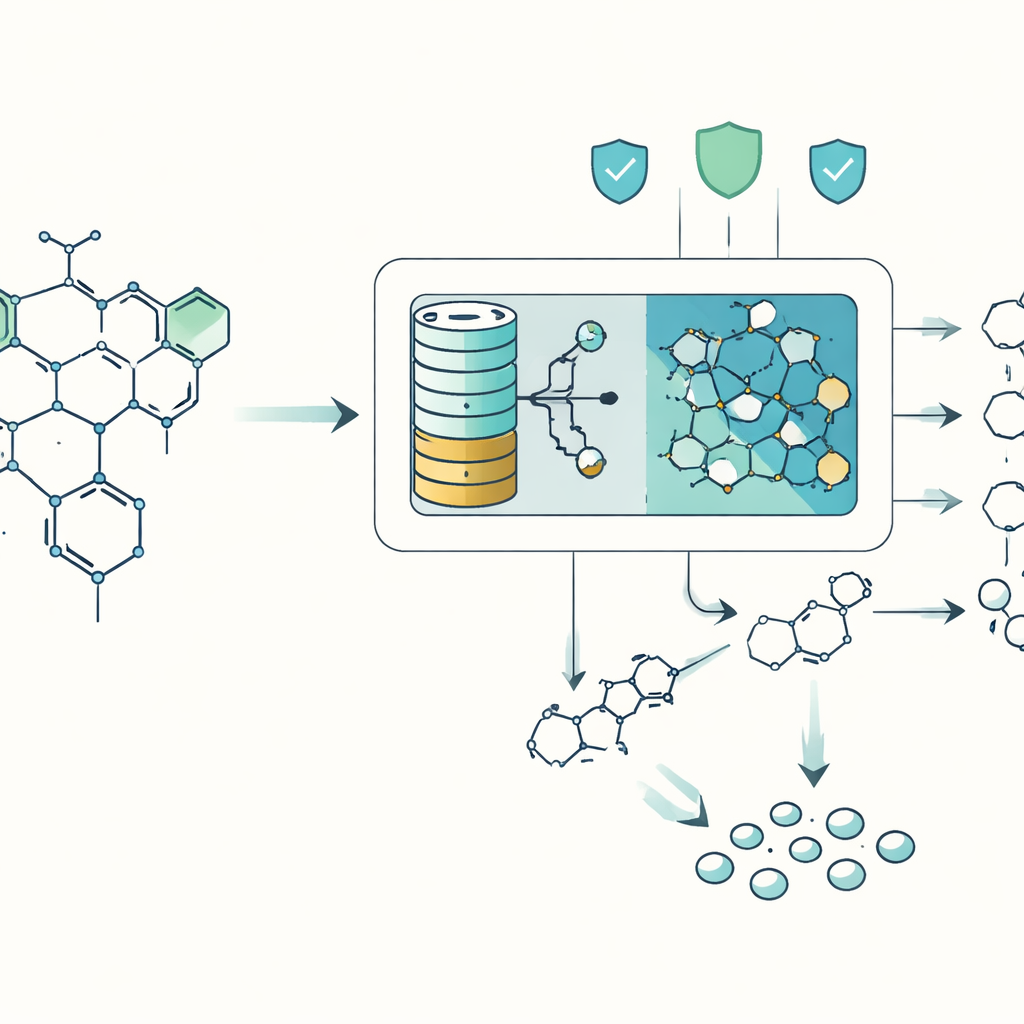
Breaking Big Molecules into Manageable Pieces
Chemists usually plan a synthesis by working backward from the target molecule, mentally “snapping” it apart into simpler pieces that can be bought or made. Computers have been helping with this task for decades, but existing tools struggle when molecules become too tangled, exotic, or unlike anything in their reaction databases. DeepRetro tackles this by combining two worlds: fast, rule-based engines that apply known reaction patterns, and a language-model brain that can suggest unusual yet chemically sensible ways to break a molecule apart. Instead of asking the AI to dream up a full recipe in one go, DeepRetro asks it for only one backwards step at a time and then checks each suggestion carefully.
Keeping the AI Honest
A key problem with large language models is that they can “hallucinate”–they may confidently propose steps that violate basic chemistry. DeepRetro wraps the AI in several layers of automated checking. Every proposed intermediate molecule is tested for simple correctness (for example, whether atoms have the right number of bonds), for likely stability, and for internal consistency with the rest of the reaction. Suggestions that fail these tests are rejected. For those that pass, the system then calls a more traditional search engine to see whether known chemistry can connect these building blocks back to real, purchasable starting materials. Chemists can also intervene at any point via a graphical interface: they can edit structures, rerun only a portion of a pathway, or add common protective handles that make multi-step chemistry practical.
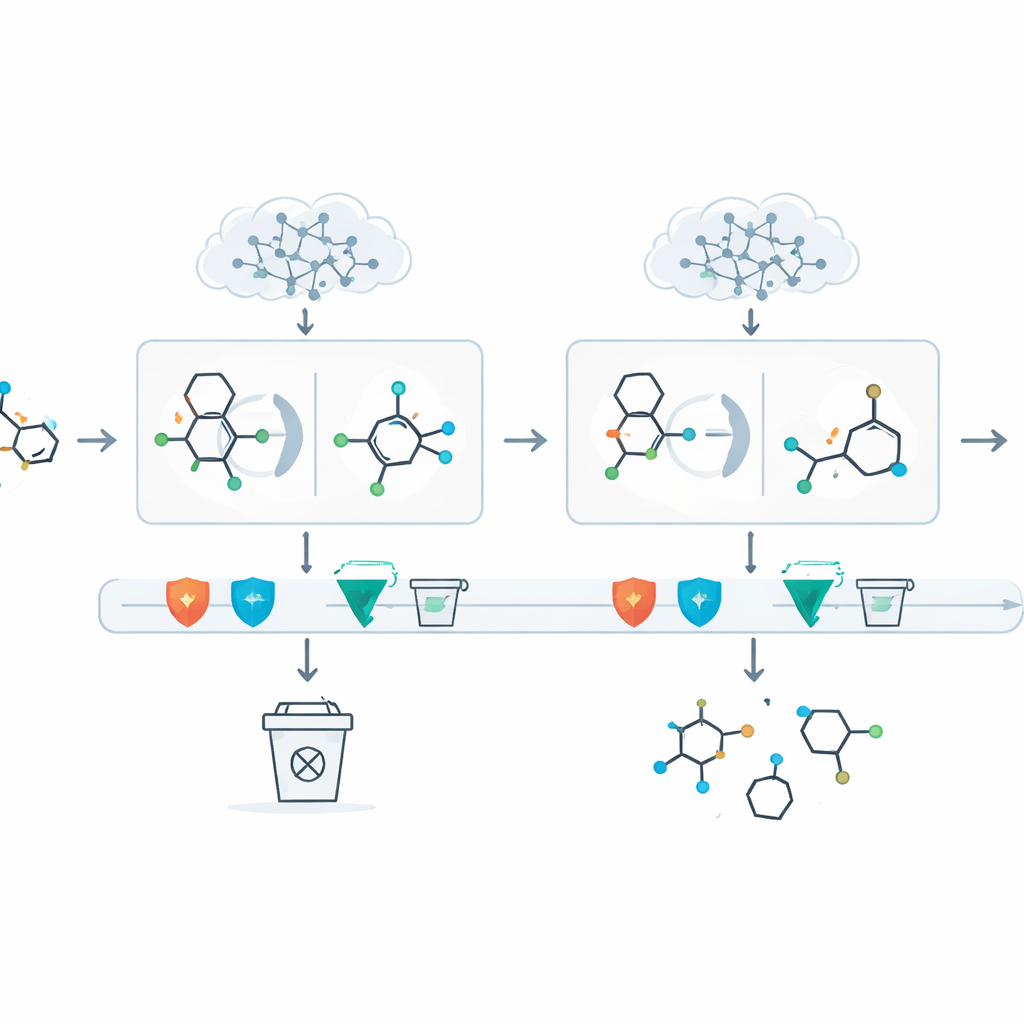
Putting the System to the Test
To see how well DeepRetro works, the authors evaluated it on standard benchmark collections of reactions from patent databases. For single-step predictions—guessing which reactants could make a given product—the system matched or exceeded strong existing tools on several measures, especially in correctly identifying the main precursor even when minor ingredients differed. For multi-step planning, DeepRetro solved nearly all targets in two demanding test sets, including a collection of particularly tricky drug-like molecules, outperforming earlier state-of-the-art methods. Importantly, these tests were run in a fully automatic mode, without human corrections, demonstrating that the framework is robust even before expert chemists step in.
Real-World Case Stories
Benchmarks alone can miss what chemists really care about: does a proposed route look like something a skilled practitioner might actually try in the lab? The authors therefore studied five famous, highly complex natural products, including the antibiotics erythromycin B and discodermolide, and the alkaloid reserpine. For each case, DeepRetro worked alongside human chemists in an iterative loop. The AI suggested disconnections and route fragments; chemists pruned out dubious ideas, fixed subtle stereochemical issues, and occasionally nudged the system with a key intermediate. In two cases, DeepRetro produced full synthetic plans whose overall strategy did not match anything the authors could find in the literature, even though the individual reactions were known. This suggests that the system can recombine familiar chemistry into genuinely new global routes.
Promise, Limits, and What Comes Next
DeepRetro shows that large language models can be more than clever text generators; when tightly supervised and combined with established tools, they can help navigate the enormous search space of possible chemical syntheses. The framework still has limits: general-purpose language models often propose unstable or unrealistic intermediates, and fully automatic solutions to the very hardest molecules remain out of reach without human oversight. Nonetheless, DeepRetro’s strong performance on standard tests, its success on challenging case studies, and its open-source release make it a practical template for future AI-assisted scientific discovery. For non-specialists, the takeaway is that AI is moving from merely predicting molecular properties to co-designing entirely new lab recipes, with the potential to speed up the creation of drugs and materials in the years ahead.
Citation: Sathyanarayana, S.V., Hiremath, S.D., Rahil Kirankumar, S. et al. DeepRetro discovers retrosynthetic pathways through iterative large language model reasoning. Sci Rep 16, 8448 (2026). https://doi.org/10.1038/s41598-026-38821-z
Keywords: retrosynthesis, large language models, organic synthesis planning, drug discovery, computational chemistry