Clear Sky Science · en
Dual-functional amino-carboxyl co-modified Fe3O4 nanoparticles for synergistic selective adsorption of lead and cadmium ions from aqueous solutionss
Why Cleaning Up Toxic Metals Matters
Lead and cadmium are two heavy metals that can quietly build up in rivers, lakes, and even drinking water, damaging the brain, kidneys, and bones at very low levels. Getting them out of water is difficult, especially when many different metals are present at once. This study describes a new kind of tiny magnetic particle that can be sprinkled into contaminated water to latch onto lead and cadmium, then be pulled back out with a magnet, offering a smarter and more selective way to protect people and ecosystems.
Tiny Magnets with a Smart Coating
At the heart of this work are nanoparticles of magnetite, a form of iron oxide that behaves like a tiny magnet. The researchers coated these particles with a thin organic layer carrying two types of chemical groups: amino groups and carboxyl groups. Each group grabs metal ions in a slightly different way. By combining both on the same surface, the team aimed to create a “dual‑functional” magnet that could recognize and hold onto lead and cadmium more strongly and selectively than simpler, single‑coated particles. The particles remain strongly magnetic, so once they have trapped the metals, they can be rapidly removed from water using a modest external magnetic field.
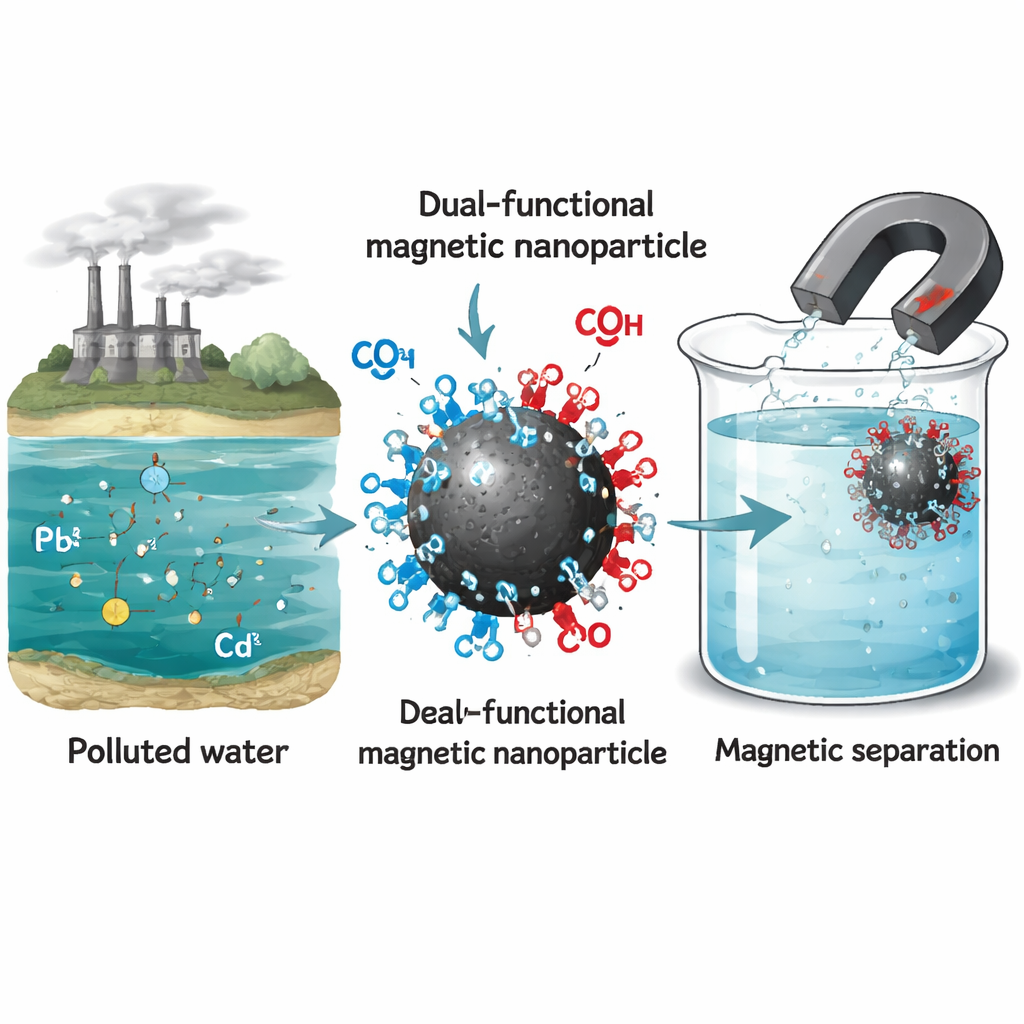
How the New Particles Outperform Older Designs
To test their design, the authors carefully compared three materials: uncoated magnetite, magnetite coated only with amino groups, and magnetite coated only with carboxyl groups, against their new dual‑coated version. In single‑metal tests, the dual‑functional particles held up to about 125 milligrams of lead and 99 milligrams of cadmium per gram of material—roughly 20–35% more than the single‑coated versions and several times more than bare magnetite. Even more importantly, when lead and cadmium were present together, the new particles clearly preferred lead. Depending on conditions, lead was taken up between about three and five times more strongly than cadmium, a big advantage for real wastewaters where many metals compete for the same sites.
Peeking into the Binding Trick
Why does this dual coating work so well? The answer lies in how lead and cadmium interact with the different surface groups. Lead, which is large and relatively “soft” in chemical terms, binds very strongly when it can connect at once to a nitrogen atom from an amino group and an oxygen atom from a carboxyl group, forming a stable ring‑like structure on the surface. Cadmium, with slightly different size and preferences, benefits less from this two‑point grip. Using computer chemistry calculations alongside experiments, the team showed that these mixed binding sites give lead a sizable extra stabilization compared with any single site alone. Measurements of how fast and how completely metals were taken up matched a picture where chemical bonding, rather than simple sticking to the surface, controls the process.
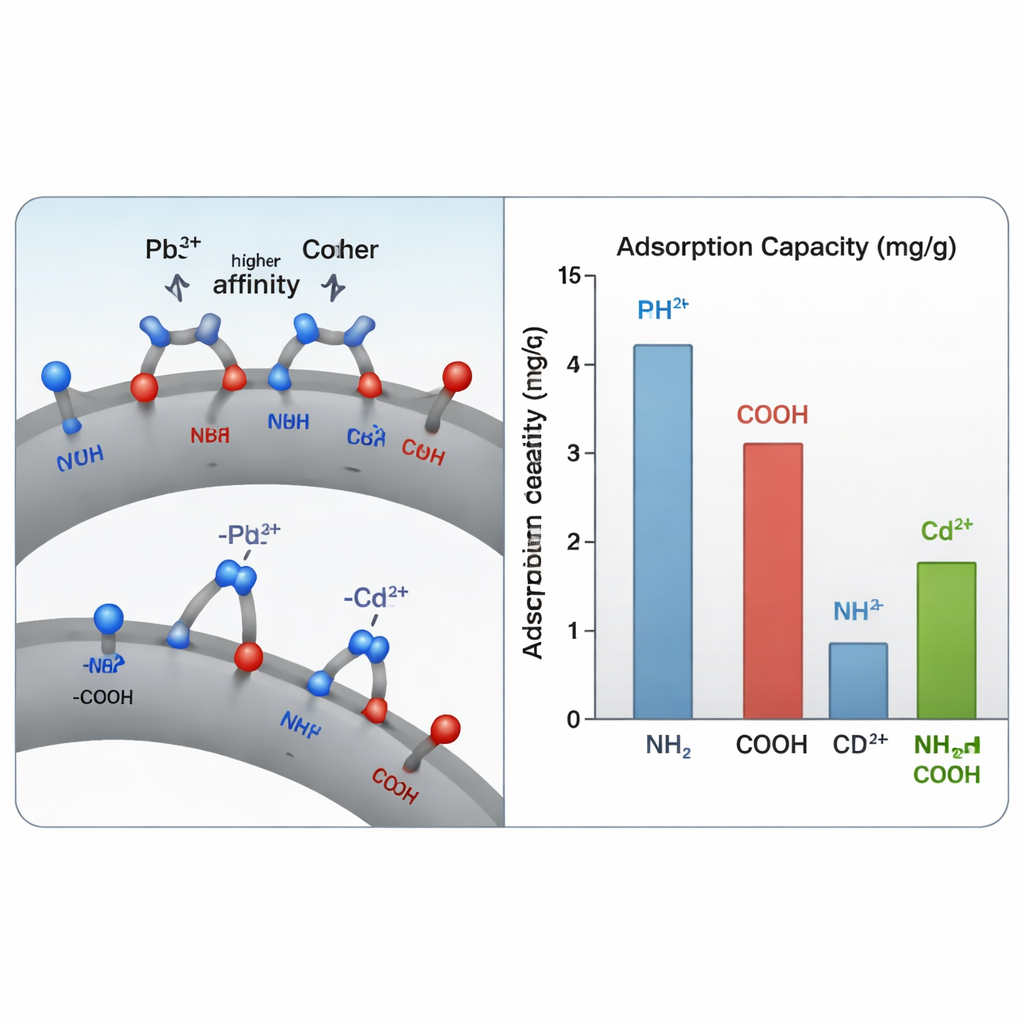
From Lab Tests to Real‑World Conditions
The authors also explored how practical these particles might be outside the lab. They found that the material works best in slightly acidic to near‑neutral water, a common range for natural and industrial waters. Under these conditions, modest doses of the particles removed nearly all of the lead and close to 90% of the cadmium within about two hours. Common background ions such as sodium, potassium, calcium, and magnesium caused only limited interference, and even in mixtures containing several different heavy metals at once, lead remained the preferred target. After use, the particles could be regenerated by washing with dilute acid, retaining more than 85% of their original capacity after five cycles while still responding quickly to a magnet.
What This Means for Safer Water
For non‑specialists, the main message is that it is now possible to build tiny, magnetically retrievable “sponges” that do more than just soak up pollutants—they can be tuned to favor the most dangerous metals, like lead, even in complex mixtures. By combining two simple chemical hooks on the same magnetic core, this study delivers a reusable material that grabs lead and cadmium efficiently, lets engineers separate it from water in minutes with a magnet, and holds up over multiple cleaning cycles. While further tests in real industrial effluents and long‑term safety studies are still needed, these dual‑functional nanoparticles point toward more selective, energy‑efficient water treatment systems that directly target some of the most harmful metal contaminants.
Citation: Yang, M., Dang, S., Gao, L. et al. Dual-functional amino-carboxyl co-modified Fe3O4 nanoparticles for synergistic selective adsorption of lead and cadmium ions from aqueous solutionss. Sci Rep 16, 7676 (2026). https://doi.org/10.1038/s41598-026-38809-9
Keywords: heavy metal pollution, magnetic nanoparticles, water purification, lead removal, nanotechnology in remediation