Clear Sky Science · en
Residue-level determinants of the thermal stability of the extremophilic Ts2631 endolysin
Why heat-loving viruses matter for future antibiotics
As antibiotic resistance rises, scientists are racing to find new ways to kill dangerous bacteria. One promising approach borrows weapons from viruses that infect bacteria, called bacteriophages. These viruses produce enzymes, known as endolysins, that punch holes in bacterial cell walls. The study discussed here focuses on a remarkably heat-resistant endolysin, Ts2631, from a virus that lives in Icelandic hot springs. By uncovering which individual building blocks of this protein make it so stable at near-boiling temperatures, researchers hope to design tougher, longer-lasting antibacterial enzymes for medicine and biotechnology. 
A viral tool forged in boiling hot springs
Ts2631 comes from a bacteriophage that infects the heat-loving bacterium Thermus scotoductus, which thrives in water reaching well above 60 °C. The Ts2631 enzyme cuts a specific bond in peptidoglycan, the tough mesh that surrounds bacterial cells. What makes Ts2631 stand out is its extreme thermostability: it remains folded until roughly 100–105 °C, depending on the solution. This is far above the temperatures where most proteins fall apart. The authors compared Ts2631 to its best-studied cousin from a moderate-temperature virus, the T7 lysozyme, to figure out what structural tricks nature has used to keep Ts2631 intact under such harsh conditions.
Shorter loops and a different mix of building blocks
Proteins are chains of 20 types of amino acids, folded into intricate shapes. When the researchers overlaid the 3D structures of Ts2631 and T7 lysozyme, they found that the overall architecture is very similar: both have a compact core of helices and strands. The key difference lies in the flexible loop regions that connect these elements. In Ts2631, these loops are shorter and show lower flexibility, while T7 lysozyme has longer, floppier loops. Ts2631 also carries more of certain amino acids often linked to heat resistance: the rigid proline; aromatic residues such as tyrosine and tryptophan; and positively charged arginine. At the same time, it has fewer serine, aspartic acid and glutamic acid residues, which can make proteins more vulnerable at high temperatures. This pattern holds not only against T7 lysozyme but also when Ts2631 is compared to hundreds of thousands of viral and many millions of bacterial proteins.
Swapping single amino acids to test their importance
To move from correlations to cause-and-effect, the team systematically replaced 55 individual amino acids in Ts2631 and measured how each mutation affected the melting temperature (Tm) and the enzyme’s ability to break down bacterial cell walls. Some changes around the zinc-containing active site were especially damaging. When they altered three zinc-coordinating residues (H30, H131 and C139), the protein’s Tm dropped by up to about 20 °C, and activity was lost. Two conserved tyrosines (Y60 and Y69) also contributed to stability. Surprisingly, removing a disulfide bond—a type of covalent link between cysteines C80 and C90—actually raised the melting temperature, even though it destroyed activity. This shows that being very stable in a test tube does not always mean a protein will function better in real conditions. 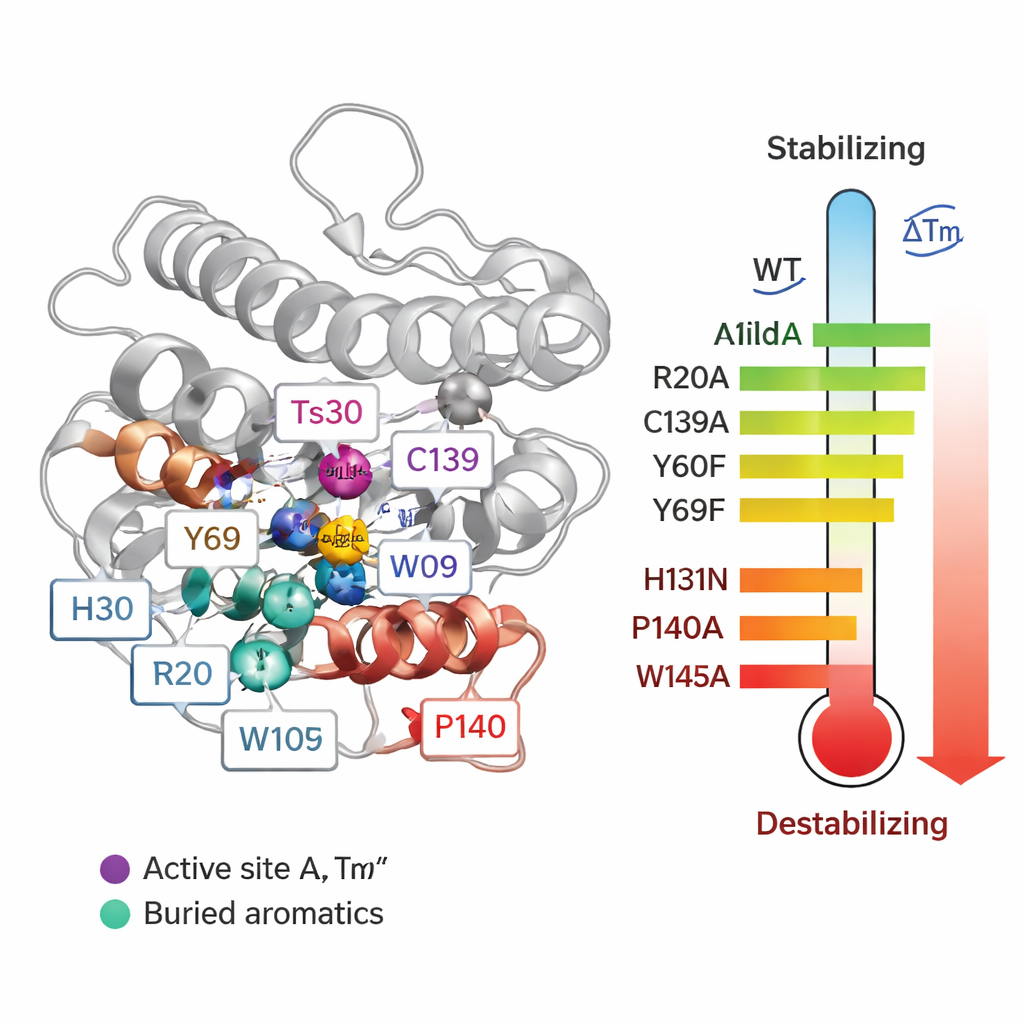
Hidden aromatic “anchors” and a key grip on the bacterial wall
The most striking results came from mutating proline and tryptophan residues. Changing a single proline (P140), located right next to the zinc-binding C139, reduced the Tm by over 21 °C and weakened activity after heating, because the mutation disrupted a special bond geometry needed for proper metal binding. Several deeply buried tryptophans (W102, W109 and especially W145) also proved critical: replacing them with alanine caused Tm drops of 14–24 °C and made the enzyme far more sensitive to heat during activity tests. These aromatic residues act like internal anchors that lock the protein’s core in place. Among arginines, most had little effect on stability, but R20 turned out to be essential for gripping peptidoglycan. When R20 was replaced, the enzyme bound the bacterial cell wall poorly and lost activity upon heating, revealing that some residues contribute more to function than to sheer thermal robustness.
From hot-spring enzymes to tougher antibacterials
Taken together, the study shows that Ts2631’s extraordinary heat resistance rests on a handful of well-placed amino acids rather than a uniform strengthening of the entire protein. Zinc-binding residues, a conserved proline near the active site, and buried aromatic side chains form a stabilizing core, while some classical stabilizing features, like disulfide bonds or salt bridges, matter less here than expected. For the broader field, this work suggests that mesophilic (moderate-temperature) endolysins could be made more durable by carefully swapping selected positions to tryptophan or proline without sacrificing antibacterial activity. In other words, by learning how nature builds enzymes that shrug off near-boiling temperatures, scientists gain a practical blueprint for engineering more reliable, long-lived protein-based antibiotics.
Citation: Cieminska, K., Kaczorowska, AK., Kozlowski, L.P. et al. Residue-level determinants of the thermal stability of the extremophilic Ts2631 endolysin. Sci Rep 16, 7630 (2026). https://doi.org/10.1038/s41598-026-38786-z
Keywords: thermostable endolysin, bacteriophage enzymes, protein thermostability, antibiotic resistance, protein engineering