Clear Sky Science · en
Local infrared stimulation modulates spontaneous cortical slow wave dynamics in anesthetized rats
Why warming tiny spots in the brain matters
Most of us know that deep sleep is vital for feeling rested and for forming memories, but the brain rhythms that underlie this state are still being unraveled. This study explores an unusual tool: near‑infrared light gently heating a pinpoint region of the rat cortex. By locally warming the tissue and listening in on brain waves and nerve cell chatter, the researchers show that it is possible to fine‑tune the slow, rolling waves that dominate deep sleep–like states, offering clues to how temperature and local circuits help shape our sleeping brains.
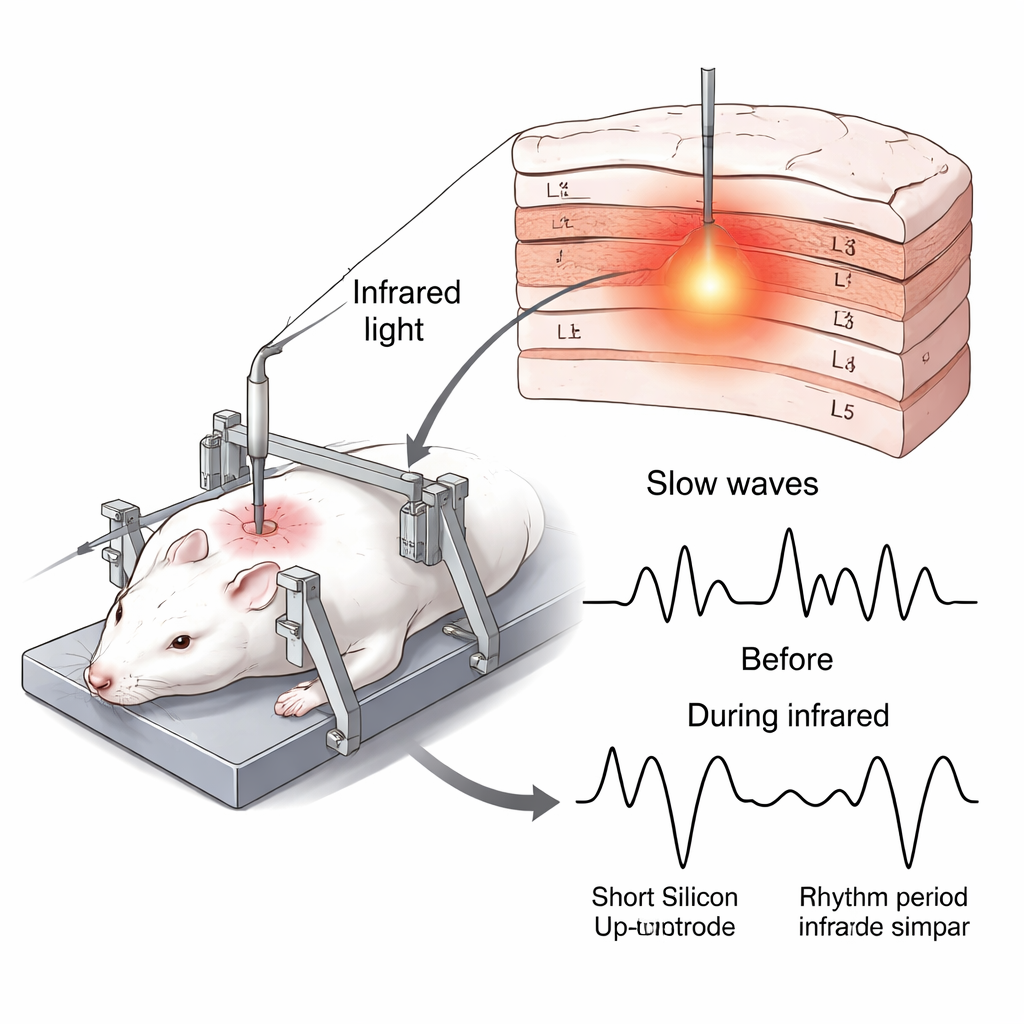
Slow rolling waves in the sleeping brain
During deep sleep and under some forms of anesthesia, vast networks of brain cells rise and fall together in a slow rhythm about once per second. In these cycles, neurons alternate between active “up‑states,” when many cells fire, and quiet “down‑states,” when activity nearly stops. These slow waves are thought to support memory consolidation, reset synaptic connections, and even help flush waste products from the brain. Earlier research showed that broadly cooling or warming the cortex can change these rhythms, but those methods affected large regions at once, making it hard to see how small, local patches of cortex contribute.
A tiny light source that doubles as a listener
To probe slow waves more precisely, the authors used a silicon‑based “optrode” — a hair‑thin probe that both delivers near‑infrared (NIR) light and records electrical signals. Inserted about 1.2 millimeters into the neocortex of anesthetized rats, the probe’s sharp tip acted as a waveguide, spreading NIR light into a small volume of tissue and raising its temperature by roughly 4–5 degrees Celsius within about a millimeter of the tip. At the same time, a line of 12 microscopic electrodes along the shaft captured local field potentials (overall brain waves) and multi‑unit activity (combined firing of nearby neurons) across superficial and deep layers in two cortical regions: a higher‑order parietal association area and a primary touch‑processing area.
Shorter bursts of activity, longer pauses
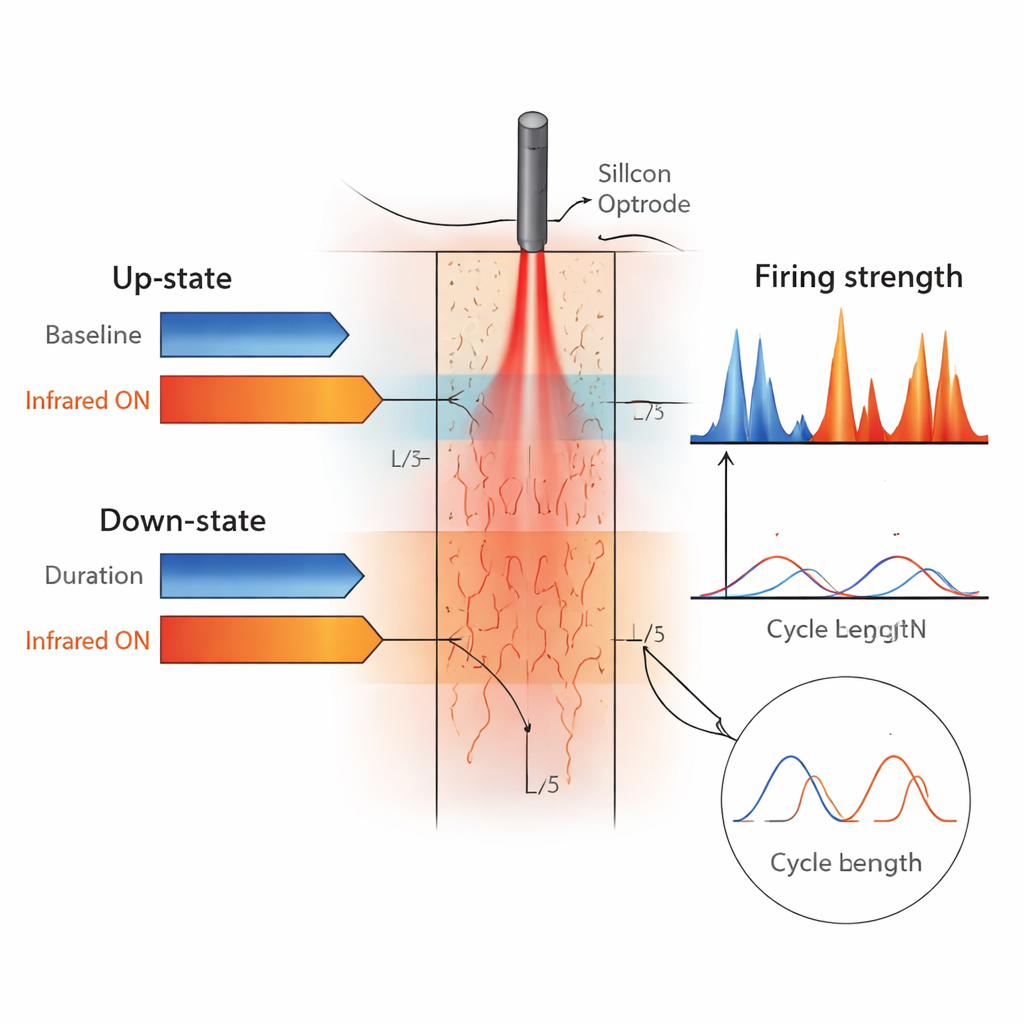
When the light was switched on for a few minutes at a time, the slow waves changed in a consistent but subtle way. The active up‑states became shorter, while the quiet down‑states grew longer, even though the overall cycle time of one up‑plus‑down sequence stayed about the same. In other words, the rhythm’s tempo barely changed, but its internal balance shifted so that neurons spent less of each cycle firing and more time in silence. At the same time, the strength of population firing during up‑states increased, and the transitions into and out of these states became steeper, suggesting that neurons were being recruited and shut off more synchronously. These effects appeared in both superficial and deep layers and repeated reliably across trials, then faded quickly once the light — and the extra warmth — were removed.
Local brain area, local response
The impact of heating on the larger‑scale brain waves depended on where the probe was placed. In the parietal association cortex, near‑infrared stimulation tended to boost the size and low‑frequency power of slow waves, hinting at stronger, more synchronized network activity. In the primary somatosensory cortex, the opposite trend often appeared: slow‑wave amplitudes and related spectral power tended to decline. The authors suggest several reasons for this contrast, including differences in cortical thickness and layering, the exact depth of the probe tip, and even the size of the surgical window over the brain, which can alter baseline cortical temperature. Despite these regional nuances, the basic pattern — briefer up‑states, longer down‑states, and sharper bursts of population firing — was robust.
What this tells us about sleep and brain control
To a non‑specialist, these findings show that gentle, highly localized warming with infrared light can nudge deep‑sleep‑like brain rhythms without disrupting them entirely. The technique acts a bit like a fine‑tuning knob: it does not speed up or slow down the beat, but it changes how long the brain spends in active versus quiet phases and how tightly neurons fire together. Because slow waves are linked to memory processing, synaptic reset, and brain “clean‑up,” understanding how temperature and local circuits shape them could eventually inform new approaches to modulating sleep, anesthesia depth, or abnormal brain rhythms — all using a minimally invasive optical tool that both shines light and listens to the brain at the same time.
Citation: Szabó, Á., Fiáth, R., Horváth, Á.C. et al. Local infrared stimulation modulates spontaneous cortical slow wave dynamics in anesthetized rats. Sci Rep 16, 7446 (2026). https://doi.org/10.1038/s41598-026-38781-4
Keywords: slow wave sleep, infrared neuromodulation, cortical temperature, neural oscillations, anesthesia